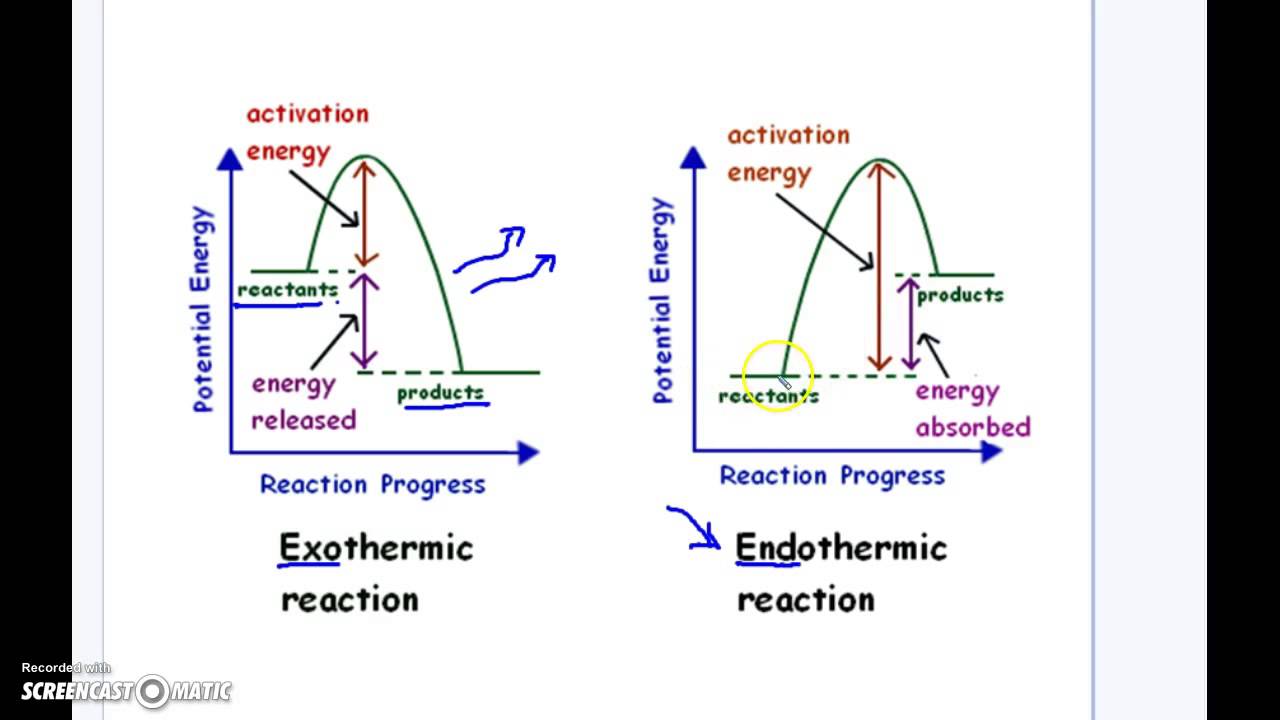
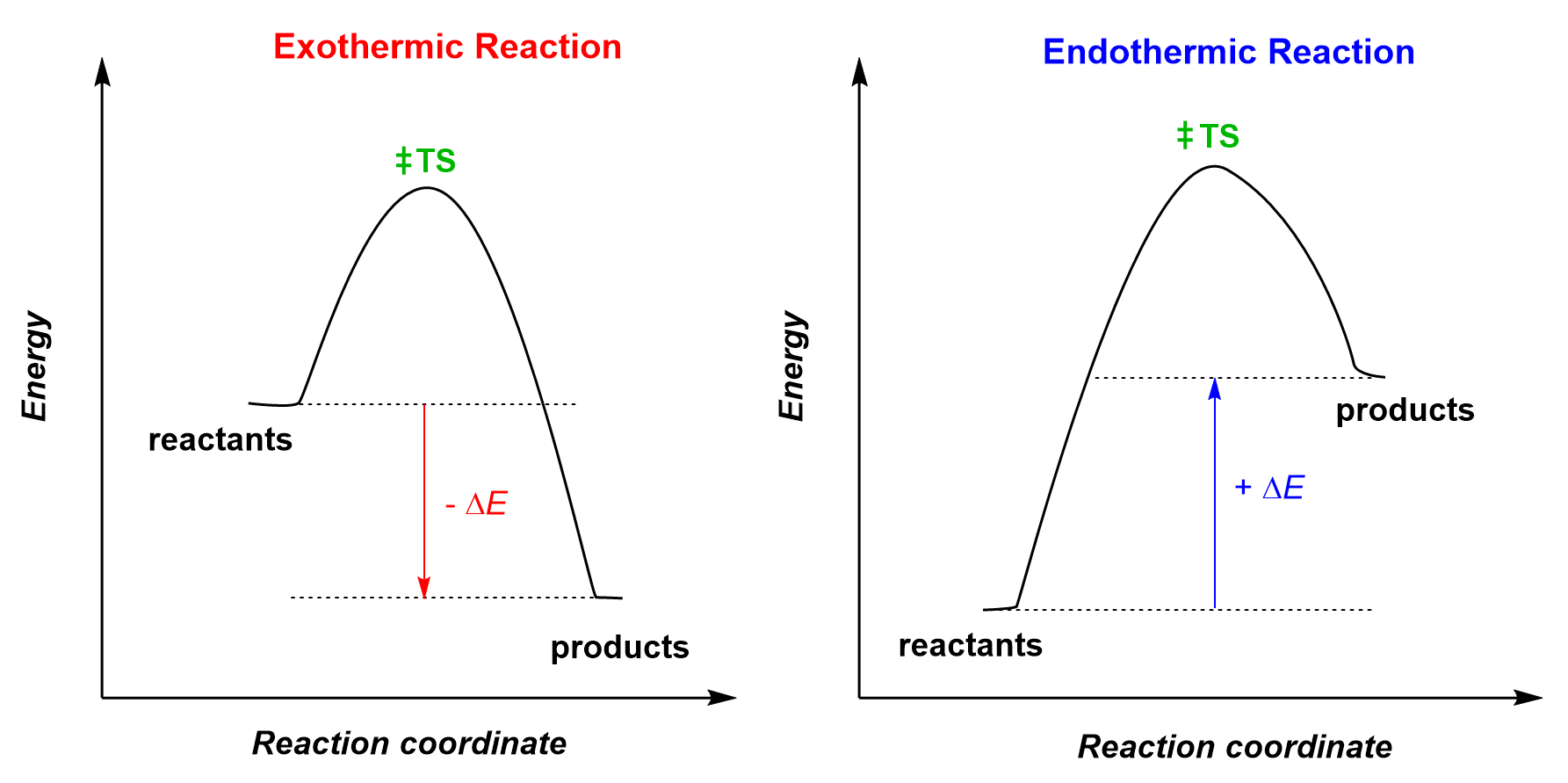
Graph Of Exothermic And Endothermic Reaction
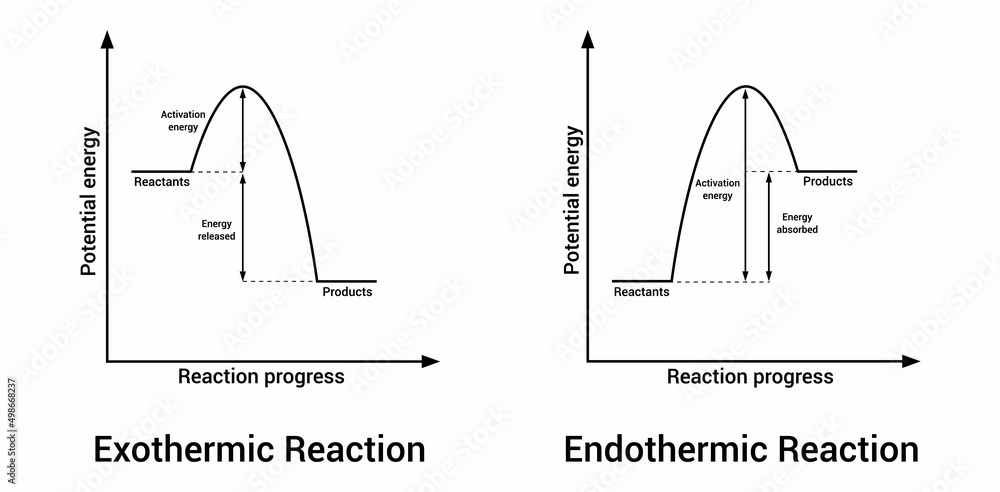
Ever have one of those days where you just feel like you're radiating energy? Or maybe the opposite, a day where you're completely drained, like a deflated balloon? Well, believe it or not, the world of tiny particles, the stuff everything is made of, has its own version of these feelings. We're talking about chemical reactions, those invisible dances happening all around us, even when we're just chilling on the couch.
Now, imagine these little particle parties. Some of them are like a super enthusiastic group of friends who just can't contain their excitement. They're practically buzzing, and when they get together, they throw off so much energy, it's like a tiny, microscopic fireworks show. This is what we call an exothermic reaction. Think of it as a reaction that’s giving off heat, like a warm hug or a cozy campfire on a chilly night. It's the kind of reaction that makes your hands feel warm when you rub them together super fast, but on a much tinier, more molecular level.
The funny thing is, we often don't even notice these little bursts of heat. Our homes are pretty good at insulating us from the constant ebb and flow of tiny energy releases. But some exothermic reactions are hard to miss! Think about the satisfying whoosh and warmth you feel when you strike a match. That's a speedy exothermic reaction at play, turning that little stick into a tiny inferno. Or, remember those hand warmers you can buy in the winter? You crack them open, and poof, instant warmth! That's another clever exothermic reaction, carefully designed to give you a little portable sunshine. It's like the universe is saying, "Here, have a little bit of warmth, you deserve it!"
Must Read
On the flip side, there are reactions that are the opposite of enthusiastic. They're more like introverts, needing a little nudge, a little encouragement to get going. These are the endothermic reactions. Instead of giving off energy, they actually take it in. Imagine a plant soaking up sunshine to grow. That's kind of like an endothermic reaction. It needs that energy from its surroundings to do its thing. Without that input, it just won't happen.
These are the reactions that might make you feel a little bit cooler. Think about melting an ice cube. As the ice melts, it absorbs heat from the air around it, making the air feel a tiny bit colder. It’s like the ice cube is saying, “Hey, can I borrow some of your warmth? I’ve got some melting to do!” Or, have you ever used an instant cold pack for a sore muscle? You snap it, and it gets really cold, right? That’s an endothermic reaction in action, sucking the heat right out of the immediate area. It’s a bit of a selfless act, really, taking on that warmth so you can feel better. It’s the universe offering a cool compress for your troubles.
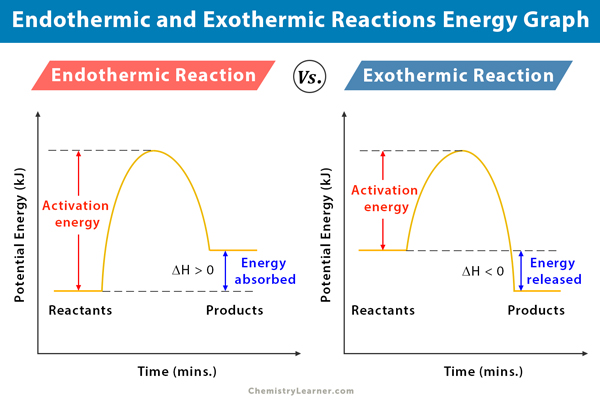
Now, imagine we could draw a little picture of what's happening with these reactions. That's where the idea of a graph comes in, and it’s not as scary as it sounds. Think of it like a storybook illustration for our tiny particle parties. On one side, we have the "before" picture – all the little bits and pieces that are about to have their reaction. On the other side, we have the "after" picture – what they've turned into. And in between, we see how their energy levels have changed.
For our energetic exothermic reactions, the graph looks like a little slide. The starting point, with all the ingredients ready to react, is up high, full of potential energy. Then, whoosh, they do their thing, release a bunch of energy, and end up at a lower energy level. It’s like starting at the top of a playground slide, full of anticipation, and then sliding down, feeling the exhilaration. The energy that was "stored up" in the ingredients is now released as heat or light. It's a satisfying descent, a feeling of release.

Now, for our energy-gulping endothermic reactions, the graph looks more like a climb. The starting ingredients are at a lower energy level. They need a boost, a helping hand of energy from their surroundings to get going. So, they climb up to a higher energy level in their "after" state, having absorbed all that lovely energy. It’s like starting at the bottom of a hill, needing to gather strength and effort to reach the summit. It’s a journey that requires input, a deliberate effort to gain something more.
So, the next time you feel a warmth spread through your hands, or a chill creep over your skin, you can think about these fascinating graphs and the tiny, energetic dances happening all around you. Whether it's a cozy fire or a refreshing cold pack, it's all part of the amazing, and sometimes surprisingly emotional, world of chemistry. It’s a constant conversation between matter and energy, a story told in warmth and coolness, and we get to be the audience to this incredible show.