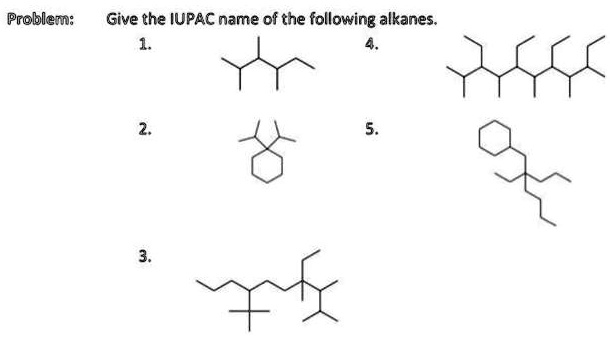
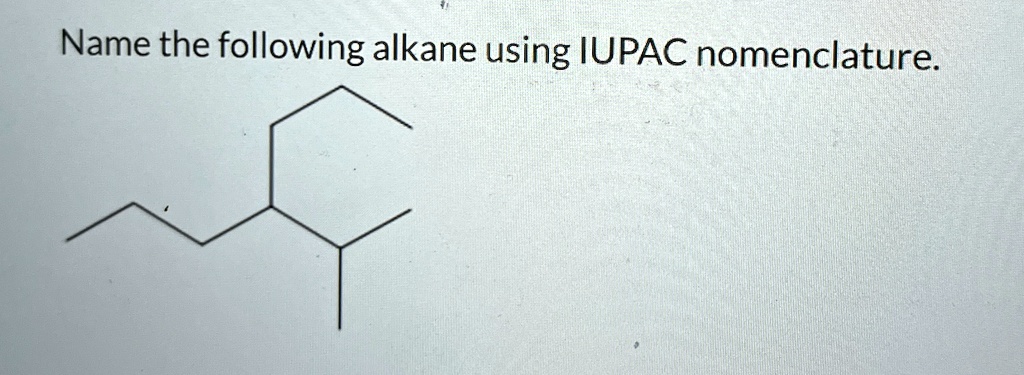
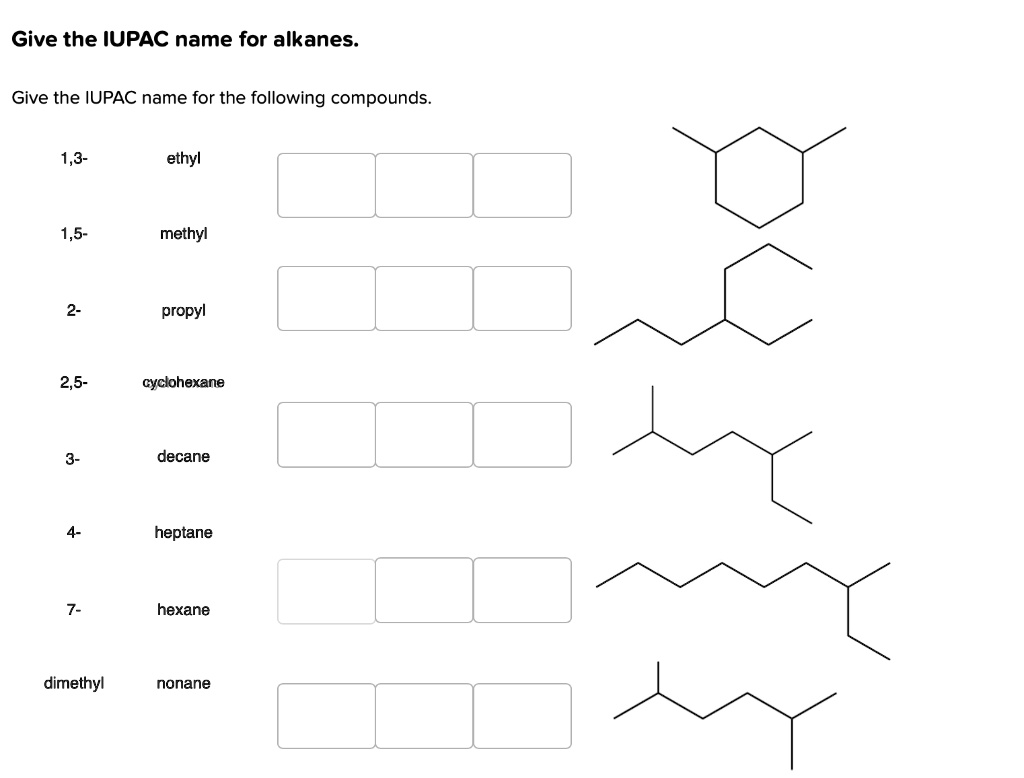
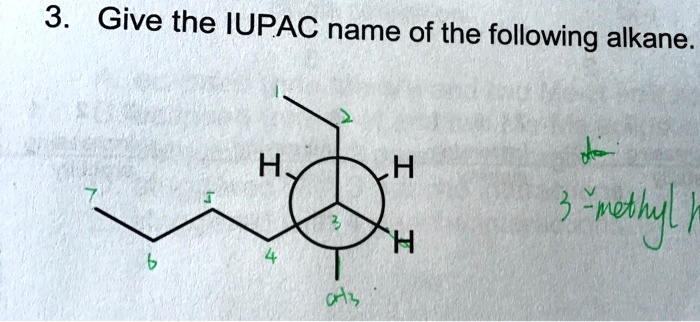
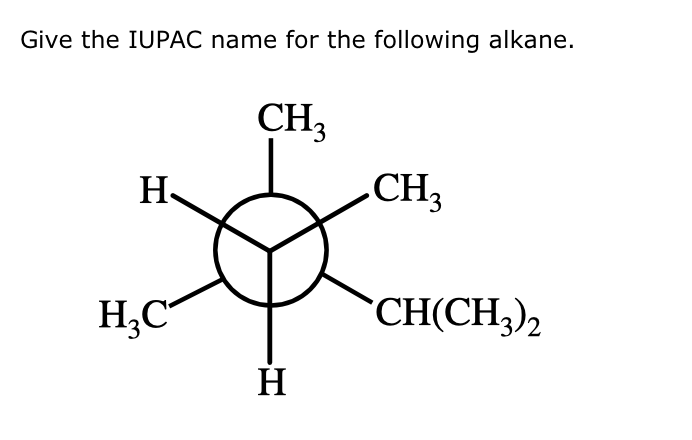
Give The Iupac Name For The Following Alkane
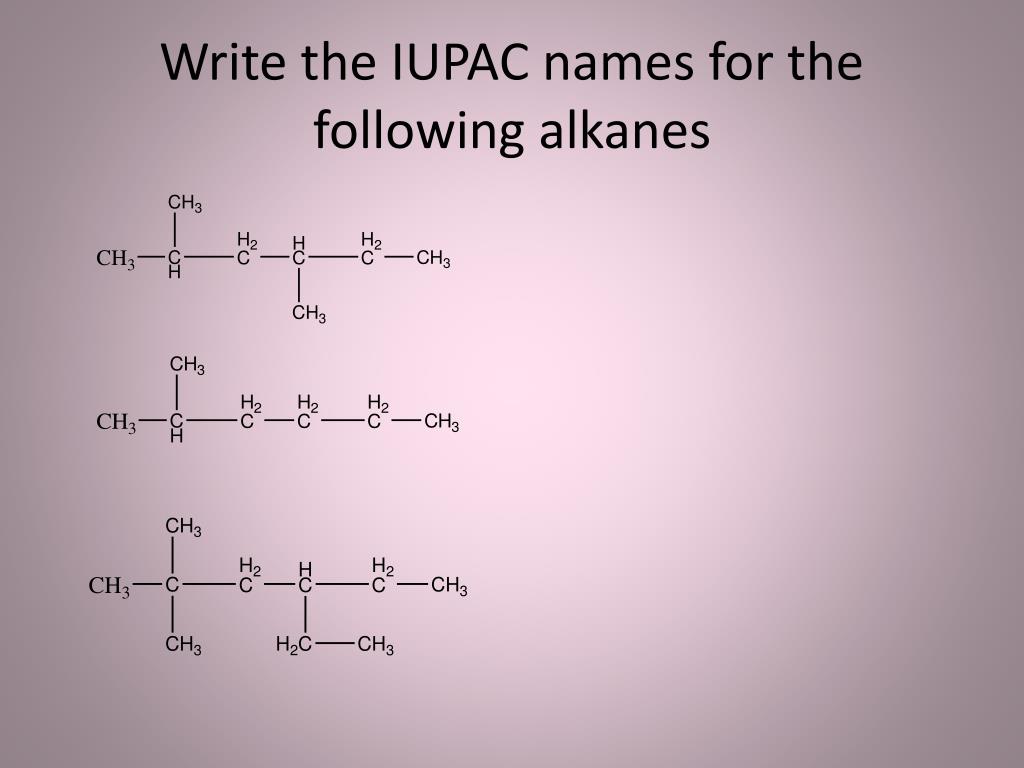
Ever looked at a complex chemical formula and thought, "Wow, that looks like a secret code!" Well, you're not far off! But here's the fun part: learning how to crack that code, especially for the building blocks of organic chemistry – alkanes – can be incredibly satisfying and surprisingly useful. Think of it like learning to read a map or speak a new language. Once you know the rules, you can navigate the vast world of molecules with confidence. And guess what? The International Union of Pure and Applied Chemistry (IUPAC) has created a system that makes this decoding process logical and, dare we say, a little bit fun!
So, why bother with these fancy names? For chemists, it's the universal language of molecules. Imagine trying to order a specific ingredient in a bustling international market without a common tongue. Chaos! IUPAC nomenclature provides that common language, ensuring that no matter where you are in the world, a scientist asking for a particular alkane will get exactly what they intended. This clarity prevents costly mistakes, speeds up research, and allows for seamless collaboration. Beyond the lab, understanding how molecules are named helps us appreciate the complex world around us, from the fuels that power our cars to the plastics in our everyday objects, and even the very building blocks of life.
Now, let's dive into the exciting challenge of naming an alkane. An alkane is a hydrocarbon, meaning it's made up solely of carbon and hydrogen atoms. They form a "skeleton" of carbon atoms linked together, with hydrogen atoms filling in the remaining bonds. The simplest alkanes are like single building blocks, and as they get more complex, they branch out, creating intricate structures. The IUPAC naming system provides a clear set of rules to describe these structures precisely. It’s like a step-by-step guide to describing a molecule’s architecture.
Must Read
The core principle is to find the longest continuous chain of carbon atoms. This longest chain determines the "parent" name of the alkane. Think of it as the main trunk of a tree. For example, if the longest chain has four carbon atoms, the parent name will be based on butane. The prefixes are pretty straightforward:
- meth- for 1 carbon
- eth- for 2 carbons
- prop- for 3 carbons
- but- for 4 carbons
- pent- for 5 carbons
- hex- for 6 carbons
- hept- for 7 carbons
- oct- for 8 carbons
- non- for 9 carbons
- dec- for 10 carbons
And it continues alphabetically from there! All alkanes end with the suffix "-ane", which tells us it's an alkane. So, a four-carbon chain becomes butane.

But what happens when the carbon chain isn't a straight line? When there are branches, or "side chains," attached to the main chain? This is where it gets really interesting! These branches are called alkyl groups, and they are named by taking the alkane name and replacing the "-ane" with "-yl". So, a one-carbon branch (methane) becomes a methyl group. A two-carbon branch (ethane) becomes an ethyl group, and so on.
The next crucial step is to number the carbon atoms in the longest chain. You want to start numbering from the end that gives the substituents (the branches) the lowest possible numbers. This is like finding the shortest route to a destination. If you have a methyl group attached to your main chain, and you can number the chain from either end, you pick the numbering that puts the methyl group on the smallest number carbon.
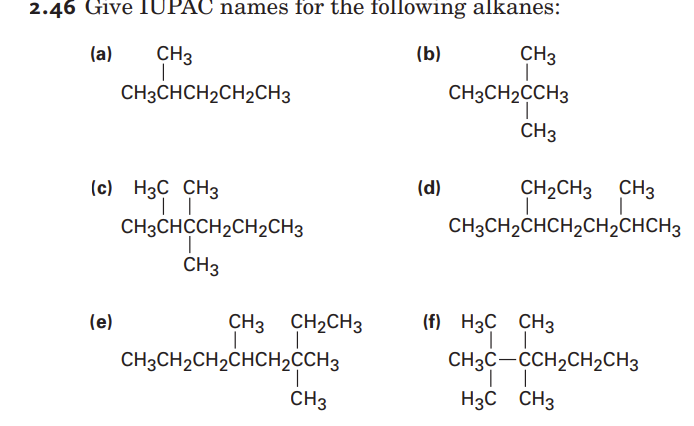
To give the full IUPAC name, you combine the number indicating the position of the substituent, a hyphen, the name of the substituent, and finally, the name of the parent alkane. For example, if you have a two-carbon chain (ethane) with a methyl group attached to the first carbon, it would be named 1-methylethane. However, if the methyl group is attached to the second carbon, it would still be 1-methylethane because you number from the end that gives the lower number. This is why it’s important to find the longest chain first.
What if there's more than one branch? You list the substituents in alphabetical order before the parent name. For example, if you have both a methyl and an ethyl group on your main chain, the ethyl group comes first in the name. You also use prefixes like di- (for two), tri- (for three), and tetra- (for four) if there are multiple identical substituents. So, if you have two methyl groups on the same parent chain, you'd call them dimethyl. For instance, if you have a three-carbon chain (propane) with two methyl groups attached to the second carbon, you would number it from either end, and the name would be 2,2-dimethylpropane.

Let's try a hypothetical challenge: Imagine an alkane structure where the longest continuous chain of carbon atoms has six carbons. Attached to the third carbon of this chain is a branch consisting of two carbon atoms. To name this, we first identify the parent chain. A six-carbon chain gives us the prefix hex-, so the parent name is hexane. Next, we identify the branch. A two-carbon branch is an ethyl group. Finally, we specify its position. Since it's attached to the third carbon, we add the number 3- before the substituent name. Putting it all together, the IUPAC name for this alkane is 3-ethylhexane. It’s a systematic process that ensures every alkane has a unique and unambiguous name. Pretty neat, right?
This system, while it might seem a bit daunting at first, is incredibly powerful. It allows us to describe even the most complex molecular structures with precision and clarity. So, the next time you encounter a long string of letters and numbers trying to represent a molecule, remember that there's a logical, fun, and incredibly useful system at play – the IUPAC naming convention for alkanes!