Ethane Reacts With Oxygen Balanced Equation
Ever wonder what happens when two common things get together and put on a show? Today, we're talking about a chemical reaction that's like a tiny, explosive dance! It's all about Ethane reacting with Oxygen.
Think of Ethane as a simple building block. It's a gas you can find in things like natural gas. And Oxygen? That's the very air we breathe, the stuff that keeps fires burning!
When these two meet, especially with a little spark of energy, things get exciting. It's not just a gentle handshake; it's more like a full-on party! The balanced equation tells us exactly how they party.
Must Read
This isn't some complicated science experiment you need a lab coat for. It's a fundamental concept that shows how nature likes to rearrange things. It’s the fundamental blueprint for a fiery performance.
So, what is this balanced equation? It’s like a recipe. It tells us precisely how much of each ingredient we need to get a specific outcome. No guesswork here, just pure chemical precision.
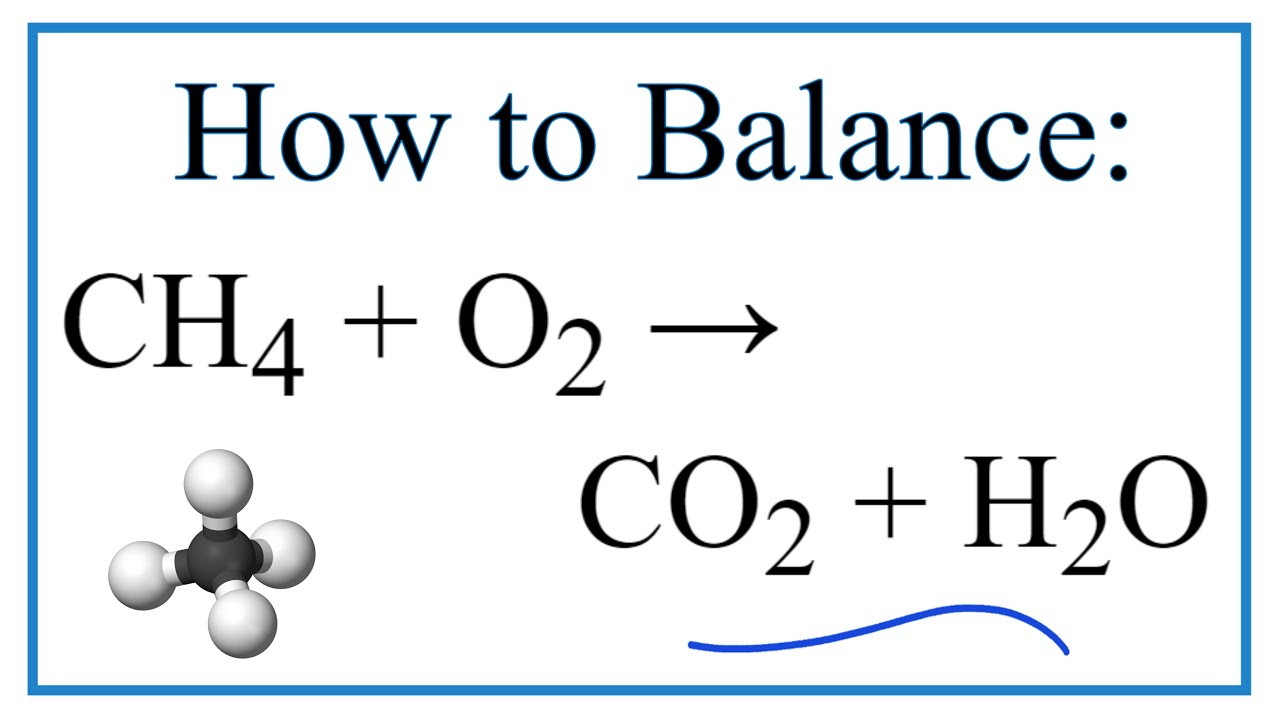
The equation we're talking about is: 2 C₂H₆ + 7 O₂ → 4 CO₂ + 6 H₂O. Don't let the numbers and letters scare you! Let's break down this superstar reaction.
First, we have Ethane. Its chemical formula is C₂H₆. This means each molecule of Ethane has two carbon atoms and six hydrogen atoms. It's a small, but mighty molecule.
Then, we have Oxygen, which we know as O₂. It's a pair of oxygen atoms joined together. And we need quite a bit of it for this show to really go!
The numbers in front of the formulas are super important. They are called coefficients. They tell us the ratio of molecules involved. It’s like saying for every two Ethane molecules, we need seven Oxygen molecules.
Now, let’s look at what happens after the reaction. This is where the magic truly unfolds. The "products" are what we get at the end.
We get Carbon Dioxide, or CO₂. This is what we breathe out, and it's also a greenhouse gas. Two carbon atoms and one oxygen atom make up this molecule.

And we also get Water, or H₂O. Yes, the same water you drink! Two hydrogen atoms and one oxygen atom make up this essential molecule.
But here's the exciting part. This reaction isn't just about making new stuff. It’s about energy! When Ethane burns with Oxygen, it releases a lot of energy.
This energy is usually released as heat and light. Think of a campfire or a gas stove. That's the energy from burning fuels like Ethane and Oxygen at play. It’s a spectacular display of power.
The balanced equation ensures that all the atoms that went in, come out. It's like a cosmic accounting system. No atoms are lost or created, just rearranged into new forms.
For every two molecules of Ethane that get together with seven molecules of Oxygen, we get four molecules of Carbon Dioxide and six molecules of Water. See? The atoms are accounted for.
It's this precise balance that makes the equation so elegant. It’s a statement of nature's order. Even in something as energetic as burning, there’s a deep, underlying structure.
Why is this so entertaining? Because it’s the fundamental process behind so much of our energy. It’s the reason we can heat our homes, cook our food, and power our cars.
Imagine a tiny, contained explosion happening over and over. That's essentially what combustion is. And the balanced equation is the script for that explosive performance.
It’s special because it’s a perfect example of a chemical transformation. Things change, but the fundamental pieces remain. It's a lesson in conservation and creation.
The visual of Ethane and Oxygen coming together, igniting, and producing heat and light is pretty awesome. It's a reminder of the powerful forces at work all around us. We are constantly surrounded by chemical reactions.
This particular reaction is a type of reaction called combustion. It’s a rapid reaction between a substance with an oxidant, usually oxygen, to produce heat and light. And Ethane is a great fuel for this process.
Think about the simplest fuels. Ethane is one of the simpler hydrocarbons. It's a good starting point for understanding how more complex fuels behave.
The beautiful thing about a balanced equation is its universality. It doesn't matter if you're in a lab or looking at a distant star; the principles are the same. This reaction respects the laws of physics.
It's also a gateway to understanding more complex chemistry. Once you grasp this basic combustion, you can start to explore other reactions. It’s a stepping stone into a vast world.
What makes it engaging is the tangible outcome. We see the results of combustion every day. It’s not some abstract theory; it’s a real-world phenomenon.
So, next time you see a flame, remember the humble molecule of Ethane. Remember Oxygen from the air. And remember the elegant dance they perform, guided by the balanced equation: 2 C₂H₆ + 7 O₂ → 4 CO₂ + 6 H₂O.
It’s a simple equation, but it unlocks a world of energy and transformation. It’s a testament to the power of chemistry. It’s a performance worth watching.

The coefficients are the stars of this show, really. They ensure that everything is perfectly matched for the grand finale. Without them, the equation wouldn't tell the whole story.
This reaction is also incredibly important in industry. It’s a key process for generating energy. It powers so much of our modern world.
The energy released is the reason we care about this reaction. It’s not just about making new molecules; it’s about harnessing power. It’s about turning potential into reality.
The balanced equation is the key to understanding how much fuel we need, and how much product we'll get. It's essential for efficient energy production. It's a fundamental tool for engineers.
Think of it as a chemical blueprint. It guides us on how to use fuels effectively and safely. It’s a fundamental principle of chemical engineering.
The simple act of burning Ethane with Oxygen is a powerful demonstration of chemical principles. It shows how atoms bond, rearrange, and release energy. It's a miniature universe of transformation.
It’s a reaction that’s both fundamental and awe-inspiring. It’s happening all around us, powering our lives. It’s a beautiful example of chemistry in action.
So, go ahead, learn about Ethane reacting with Oxygen. It’s a reaction that’s as fundamental as it is fascinating. It’s a showstopper in the world of chemistry.
You might find yourself looking at a flame a little differently after this. You'll see the dance of molecules, guided by the precise steps of a balanced equation. It’s a captivating spectacle.

It's a story of transformation, energy, and the elegant order of the universe. It's a story told by chemistry. And this particular chapter is a fiery one.
The balanced equation for the reaction of Ethane with Oxygen is a powerful statement. It’s a concise and elegant representation of a fundamental chemical process. It’s a cornerstone of understanding combustion.
It’s amazing how much information can be packed into just a few symbols and numbers. The equation 2 C₂H₆ + 7 O₂ → 4 CO₂ + 6 H₂O is a perfect example. It tells a complete story.
This reaction is a vibrant reminder of the dynamic nature of matter. It’s a demonstration that even simple substances can combine to create powerful and useful outcomes. It's a testament to the potential within everyday elements.
So, explore further! This is just the beginning of understanding the exciting world of chemical reactions. The universe is full of them, and this one is a great place to start.
"The balanced equation is the choreographer of this fiery ballet."
It ensures every atom plays its part perfectly. No missed steps, no dropped props. Just pure, efficient transformation.
It's a reaction that's both visually dramatic and scientifically profound. It’s a perfect blend of the observable and the conceptual. It sparks curiosity.
Understanding this equation helps us appreciate the energy that fuels our lives. It’s a peek behind the curtain of the everyday. It’s a fundamental understanding of how the world works.
So, next time you see a flame, think of the dance of Ethane and Oxygen. Think of the balanced equation, orchestrating this energetic display. It's a performance that never fails to impress.