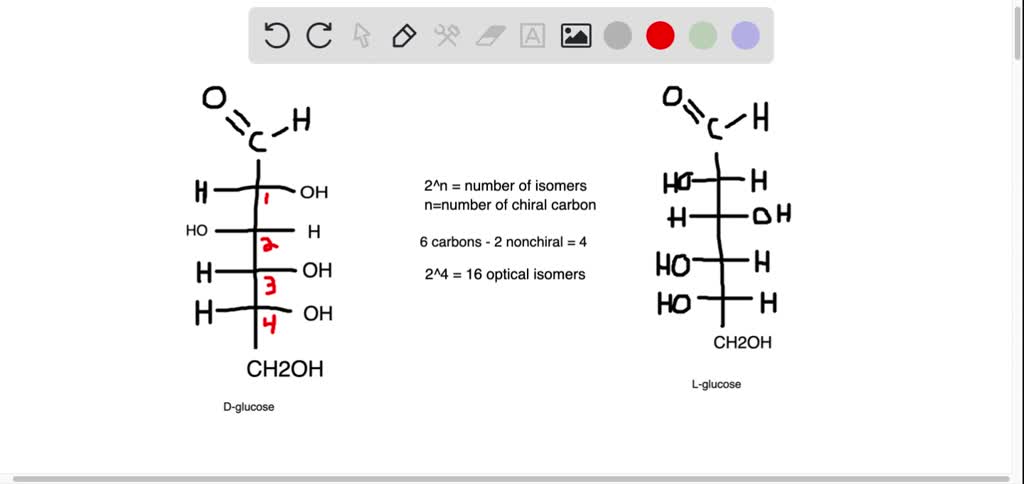
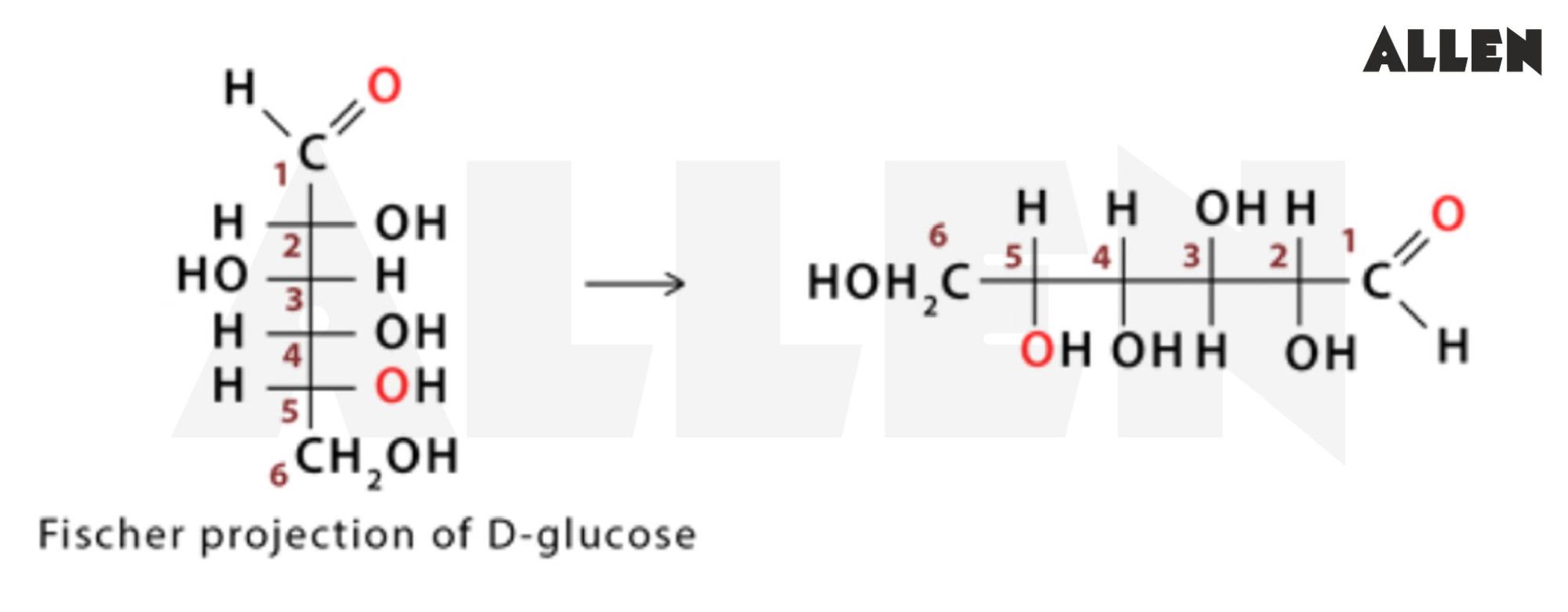
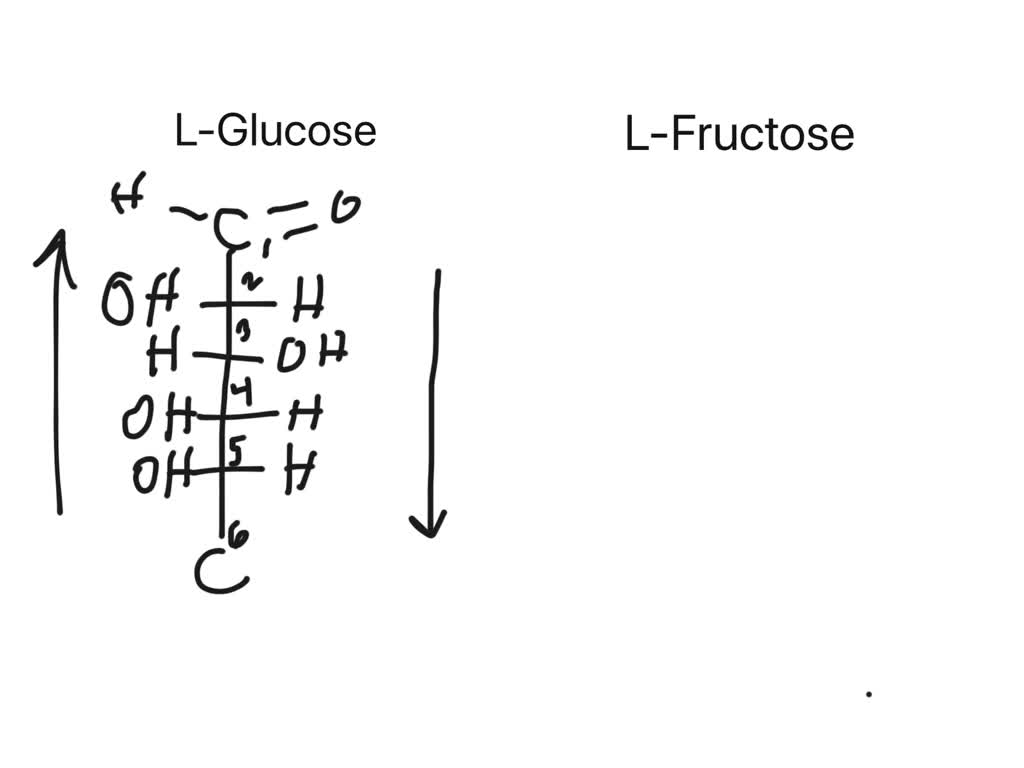
Draw The Fischer Projection For L-glucose

Hey there, sugar fiends and science curious folks! Ever wonder about those fancy molecular drawings that look like little stick figures having a party? Today, we're diving into the sweet world of sugars, and specifically, a star player: L-glucose. No, it's not just another candy bar. This is where things get deliciously complex, and dare I say, kinda fun!
So, what’s the big deal about L-glucose? Well, imagine all sugars are like twins. Most of them are right-handed, you know, like your dominant hand. We call them D-sugars. But then there are their left-handed twins, the L-sugars. L-glucose is one of these cool, less common twins. It’s like the quirky cousin at the family reunion.
Now, how do we draw this L-glucose character? That’s where the Fischer Projection comes in. Think of it as a special handshake for molecules. It’s a way chemists represent a 3D sugar molecule on a flat piece of paper. Pretty neat, right? Like drawing a superhero’s cape in 2D when it’s actually flowing all around them.
Must Read
The Fischer Projection is all about understanding how things are arranged in space. For glucose, we're talking about a six-carbon sugar. Six carbons! That’s like a little ladder of carbon atoms. And each carbon has stuff hanging off it. We’ve got hydroxyl groups (-OH), hydrogen atoms (-H), and at the top, an aldehyde group (-CHO). Ooh la la, fancy chemistry terms! But don’t let them scare you. They’re just pieces of the puzzle.
Let’s break down the L-glucose Fischer Projection, shall we? It’s not rocket science, it’s more like… sugar science! The projection is drawn with the carbon chain going vertically. The most oxidized carbon, the aldehyde group in our case, is at the very top. So, picture that at the peak of your little molecular mountain.

Then, we have carbons 2 through 5. These are the middle children of our glucose molecule. And on these carbons, we have those hydroxyl groups and hydrogen atoms hanging out. They’re like the arms and legs of our carbon ladder. For L-glucose, the magic happens with the hydroxyl group on the second-to-last carbon. This is where the "L" part really makes its mark!
In D-glucose, the most common type, that hydroxyl group on carbon 5 (the second-to-last one, remember?) is on the right. It's like it's waving hello with its right hand. But in L-glucose, it’s a rebel! That hydroxyl group is on the left. It’s giving a little left-handed wave. That’s the key difference, folks. Super subtle, but totally changes everything.
So, when you draw it, the carbon chain is vertical. Aldehyde at the top. Then, carbons 2, 3, 4, and 5. The trickiest part? The arrangement of the -OH and -H groups on carbons 2, 3, and 4. For L-glucose, it’s a specific pattern. Carbon 2: -OH on the left. Carbon 3: -OH on the right. Carbon 4: -OH on the left. See the pattern? Left, right, left. It’s like a little molecular dance step!

And the very bottom carbon, carbon 6? That’s the little guy at the base of the ladder. It’s usually a -CH2OH group. It’s just chilling there, holding things down. No major drama there.
Why is this even a thing, you ask? Well, our bodies are super picky about sugars. They’re designed to interact with D-sugars. Think of it like keys and locks. Our enzymes have locks that are shaped for D-glucose. L-glucose, with its left-handed twist, just doesn’t fit. It’s like trying to use a car key in your house door. It’s the wrong shape!
This is why L-glucose isn’t your everyday energy source. Your body mostly ignores it. It’s like serving your guests coffee when they asked for tea. They might appreciate the gesture, but it’s not what they’re used to. Some bacteria, though? They’re a bit more adventurous and can use L-sugars. Nature is full of surprises, isn't it?
Drawing Fischer Projections might seem a bit tedious at first. Like learning to tie your shoelaces. But once you get the hang of it, it’s like unlocking a secret code. You can see the 3D structure hinted at on a 2D page. It’s a powerful tool for chemists and biochemists. They can tell apart these tiny molecular differences that have huge implications.
Think about it: the exact same atoms, just arranged a little differently in space, and one is the fuel for your body, while the other sails right through. That’s the power of chirality, the fancy word for this "handedness." L-glucose is a prime example of this fascinating phenomenon.

And the name! L-glucose. It just sounds… chic, doesn’t it? Like a designer label for molecules. While D-glucose is the supermodel of the sugar world, L-glucose is the cool, indie artist. It has a certain je ne sais quoi.
So, next time you see a Fischer Projection, don't just see a bunch of lines and crosses. See the molecule’s personality! See the subtle differences that make one sugar the life of your cellular party and the other a wallflower. See the elegance of how a slight twist can change the whole story.
Drawing L-glucose’s Fischer Projection is more than just an academic exercise. It’s a glimpse into the intricate dance of molecules that make up life. It’s a reminder that even the smallest changes can have big consequences. And honestly, it's pretty cool to be able to draw a molecule that's just a little bit different, a little bit left-handed. It’s a conversation starter, a science snack, and a whole lot of fun. Go ahead, give it a try! You might just find yourself hooked on the sweet, complex world of sugars.