Have you ever wondered why some things are more sour or stingier than others? It all comes down to something called acidity, and surprisingly, the little things we can't even see – electrons – play a starring role. Today, we’re going to dive into a curious question that might sound a bit technical but is actually quite fascinating: Do electron-donating groups increase acidity? It's a question that unlocks a deeper understanding of the world around us, from the tang of a lemon to the way some materials behave.
So, what's the big deal about acidity and electrons? Acidity, in simple terms, is the ability of a substance to give up a proton (a positively charged part of an atom). The easier it is for a molecule to let go of that proton, the more acidic it is. Now, think of electron-donating groups as little helpers that can either make it easier or harder for a molecule to part with its proton. These groups have extra electrons they're happy to share. The purpose of understanding this relationship is to predict and explain the chemical behavior of different substances. This knowledge is incredibly useful in fields like chemistry, biology, and even medicine.
You might be surprised by the practical applications. In education, this concept is fundamental to teaching organic chemistry. When students learn about the structure and reactivity of molecules, understanding the influence of electron-donating and electron-withdrawing groups is crucial. In daily life, this principle helps explain why some foods are more acidic than others. For instance, the presence of certain functional groups on organic molecules contributes to the sourness of citric acid in oranges or lactic acid in yogurt. It also plays a role in the development of pharmaceuticals, where tweaking molecular structures can affect how drugs are absorbed and interact with our bodies.
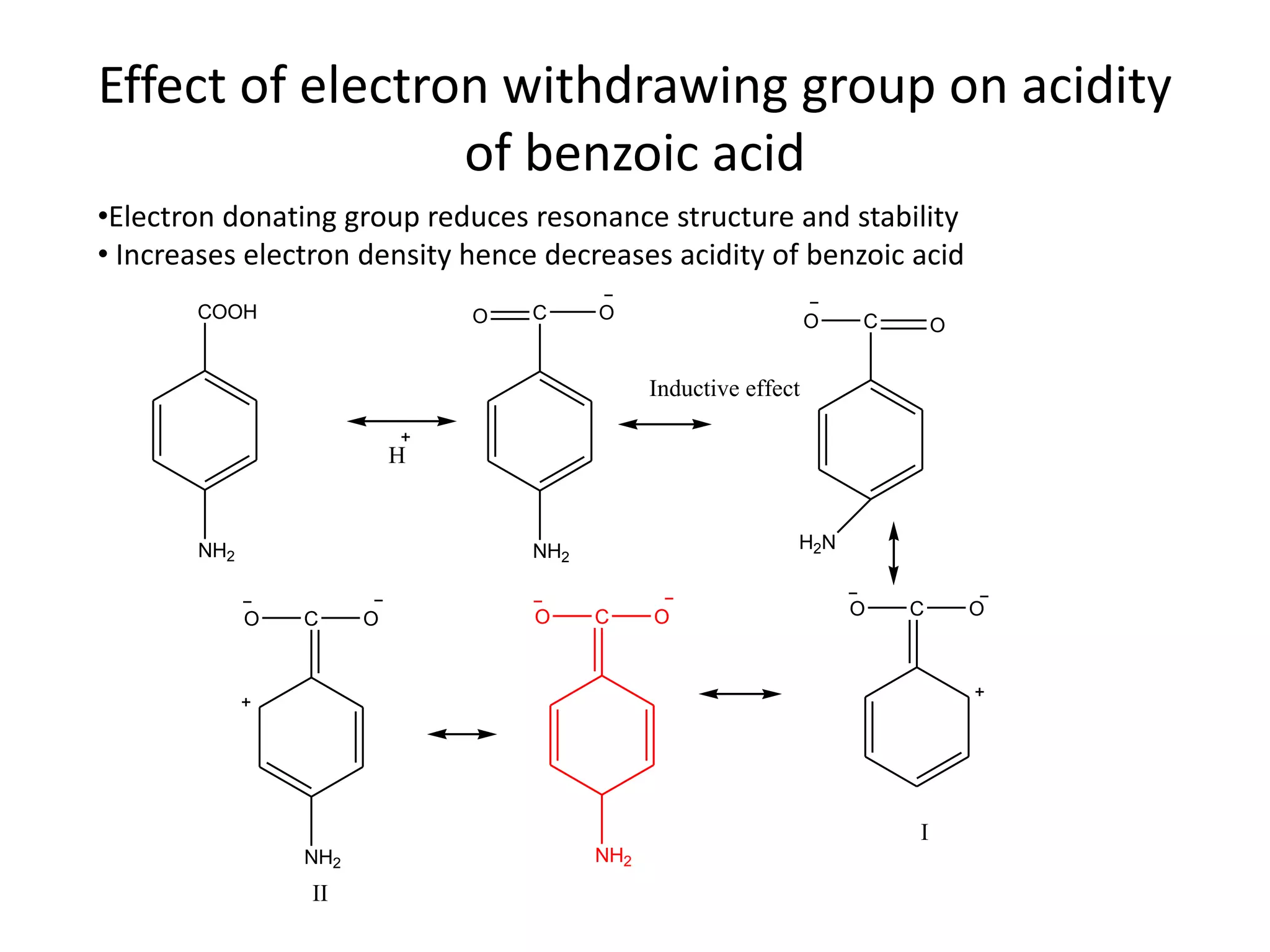
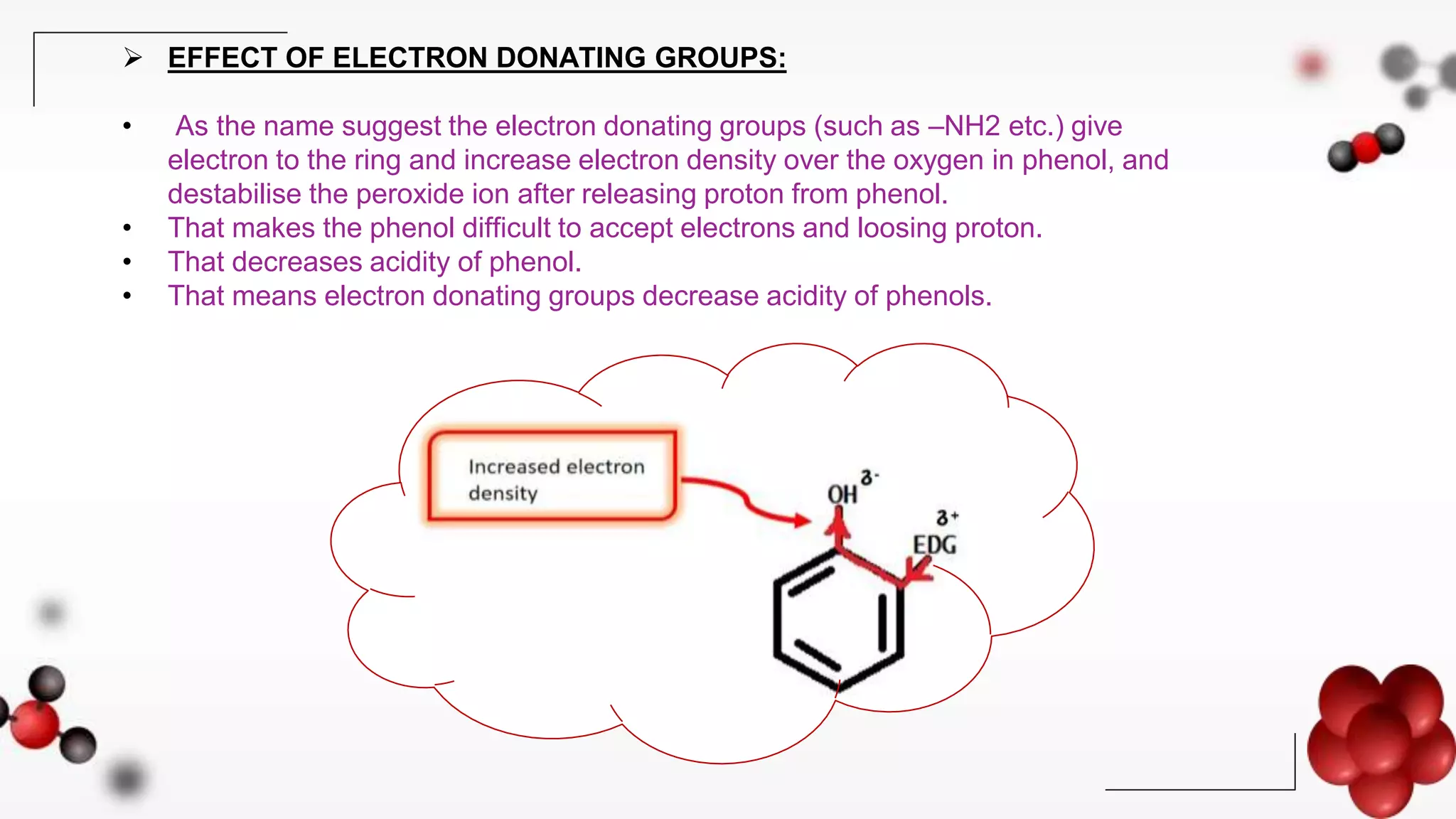
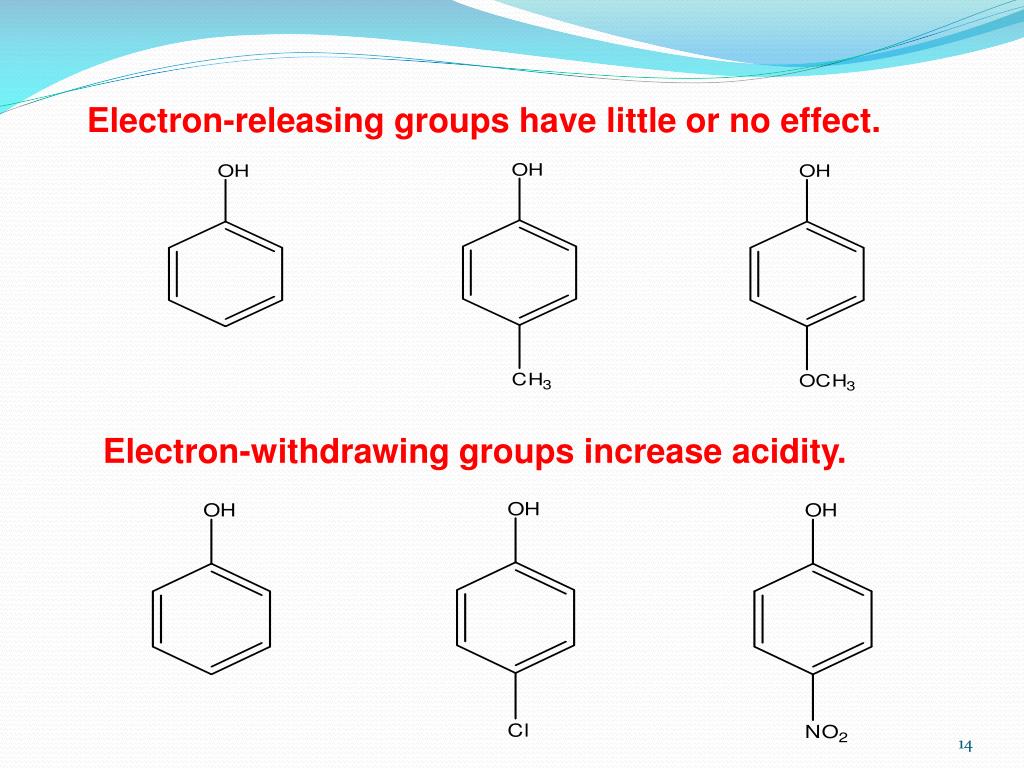
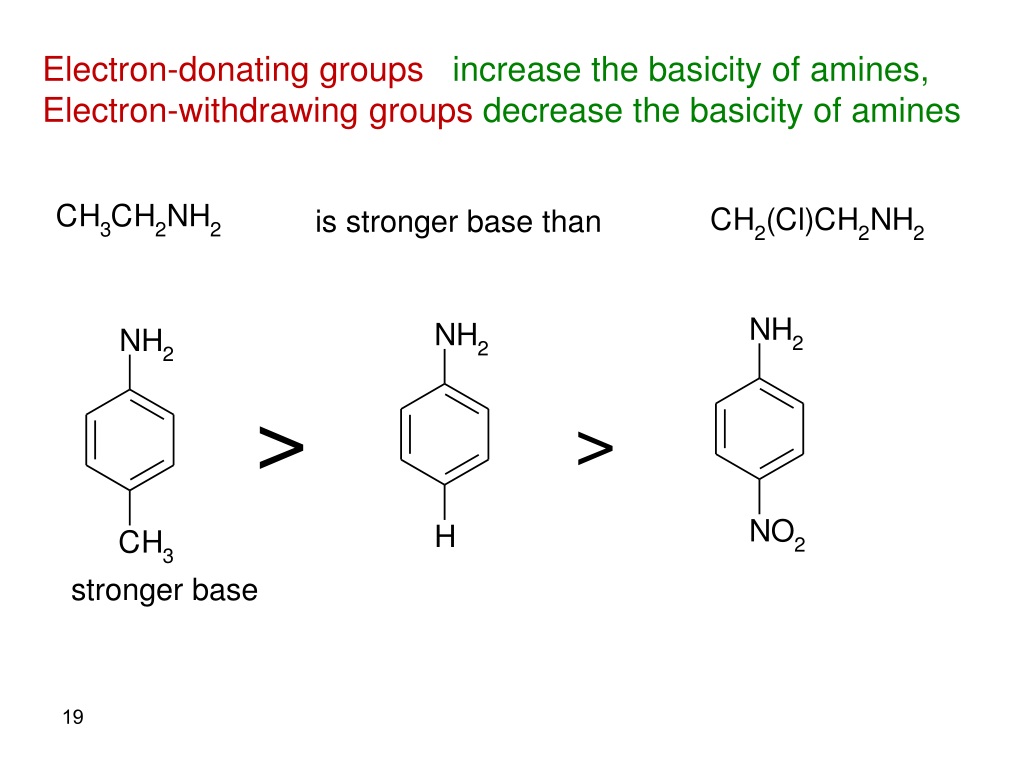
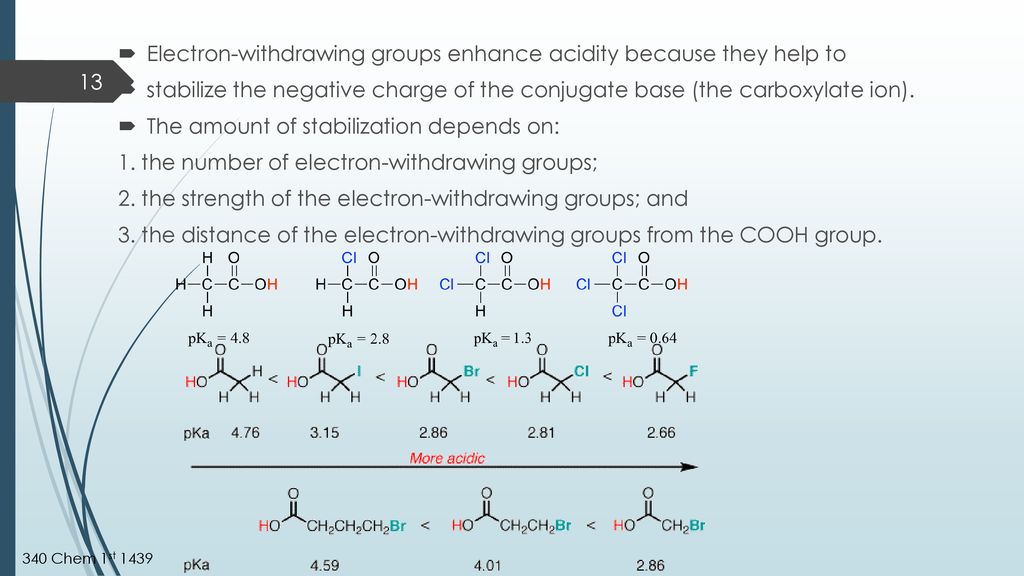
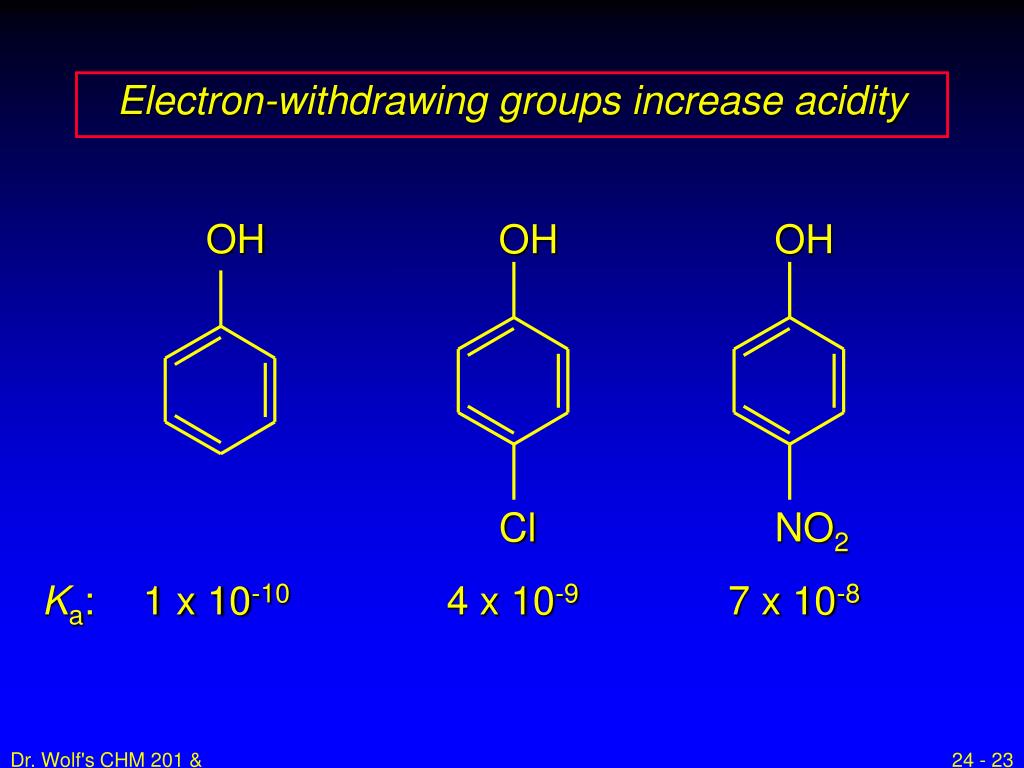
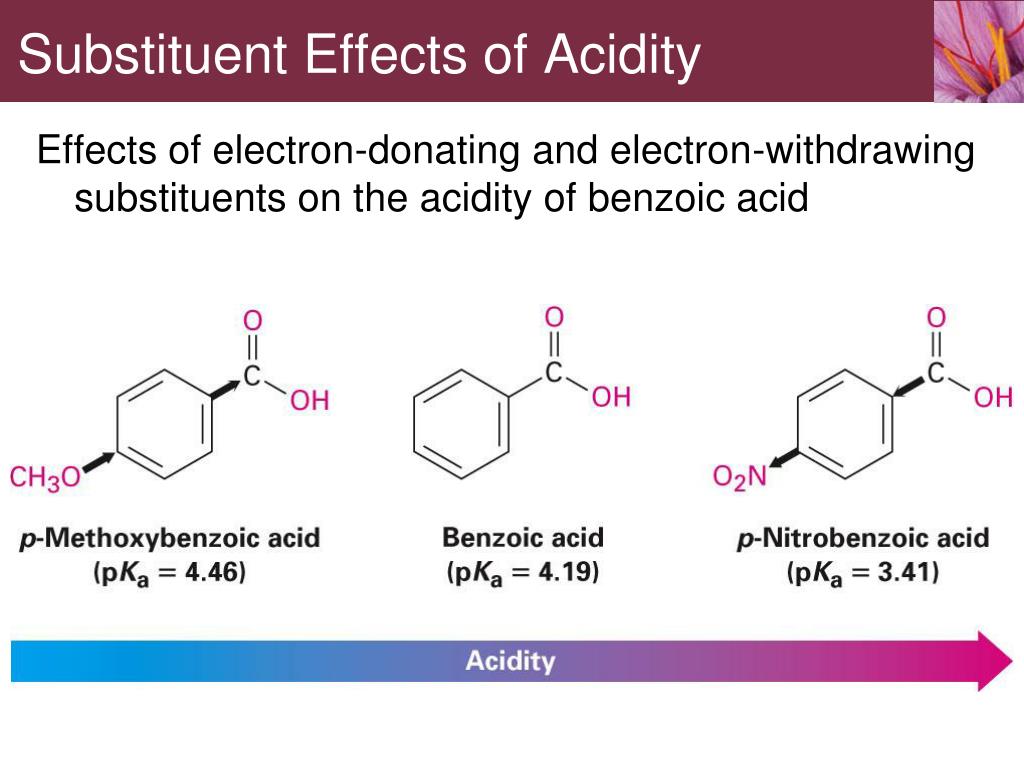
Now, let's tackle the core question. Do electron-donating groups increase acidity? The short answer, and the more common scenario, is actually no, they tend to decrease acidity. This might seem counterintuitive! Think of it this way: when a molecule loses a proton, it often becomes negatively charged. An electron-donating group, by generously pushing its electrons towards the molecule, can stabilize that negative charge. This stabilization makes it easier for the molecule to form that negative charge in the first place, meaning it's more willing to give up its proton. So, if a group donates electrons, it makes the resulting negative ion more stable, thereby making the original molecule more acidic.
However, there’s a twist! The effect can be more nuanced and depends on the specific molecule and the position of the electron-donating group. Sometimes, electron-donating groups can influence the molecule in ways that indirectly affect acidity. For a hands-on exploration, you could try comparing the acidity of different vinegars. While they are all acidic, you might notice subtle differences in their "bite." Or, if you're into baking, the reaction of baking soda (a base) with acidic ingredients like lemon juice or vinegar is a great example of acid-base chemistry in action, indirectly demonstrating the role of acidity. You can also look up simple diagrams of common organic acids and their substituents to see how electron-donating groups are represented!