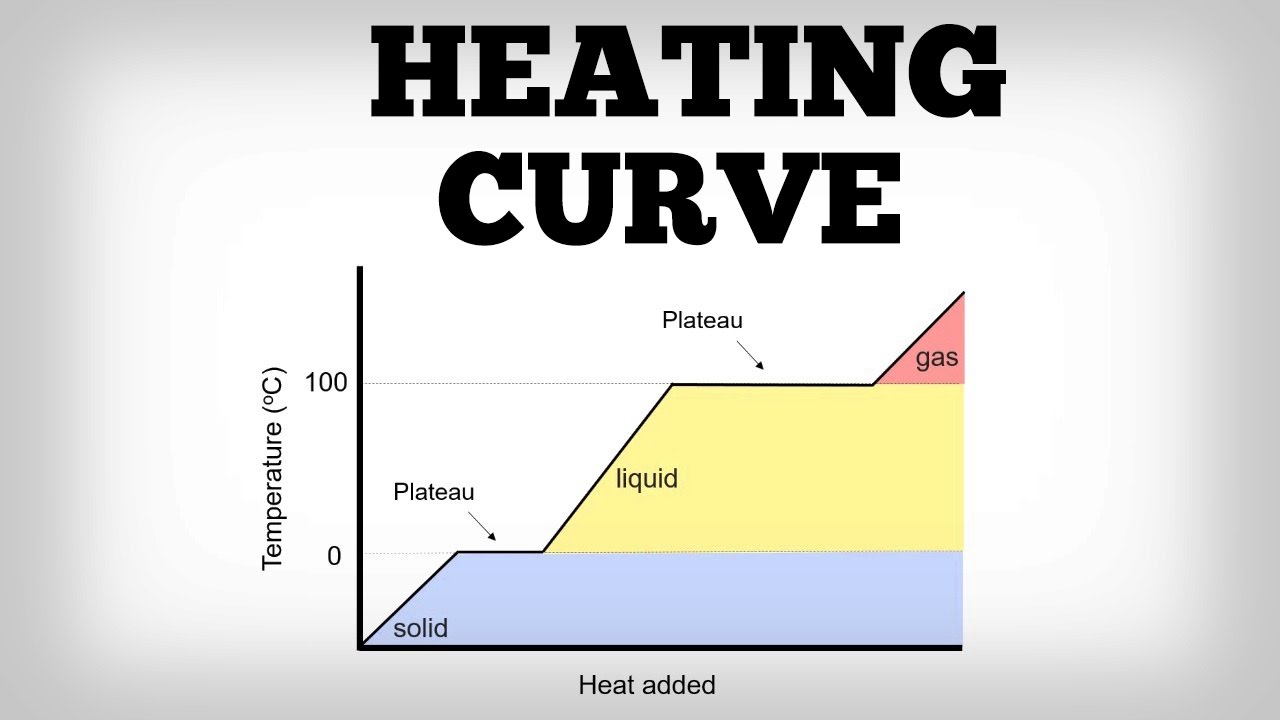
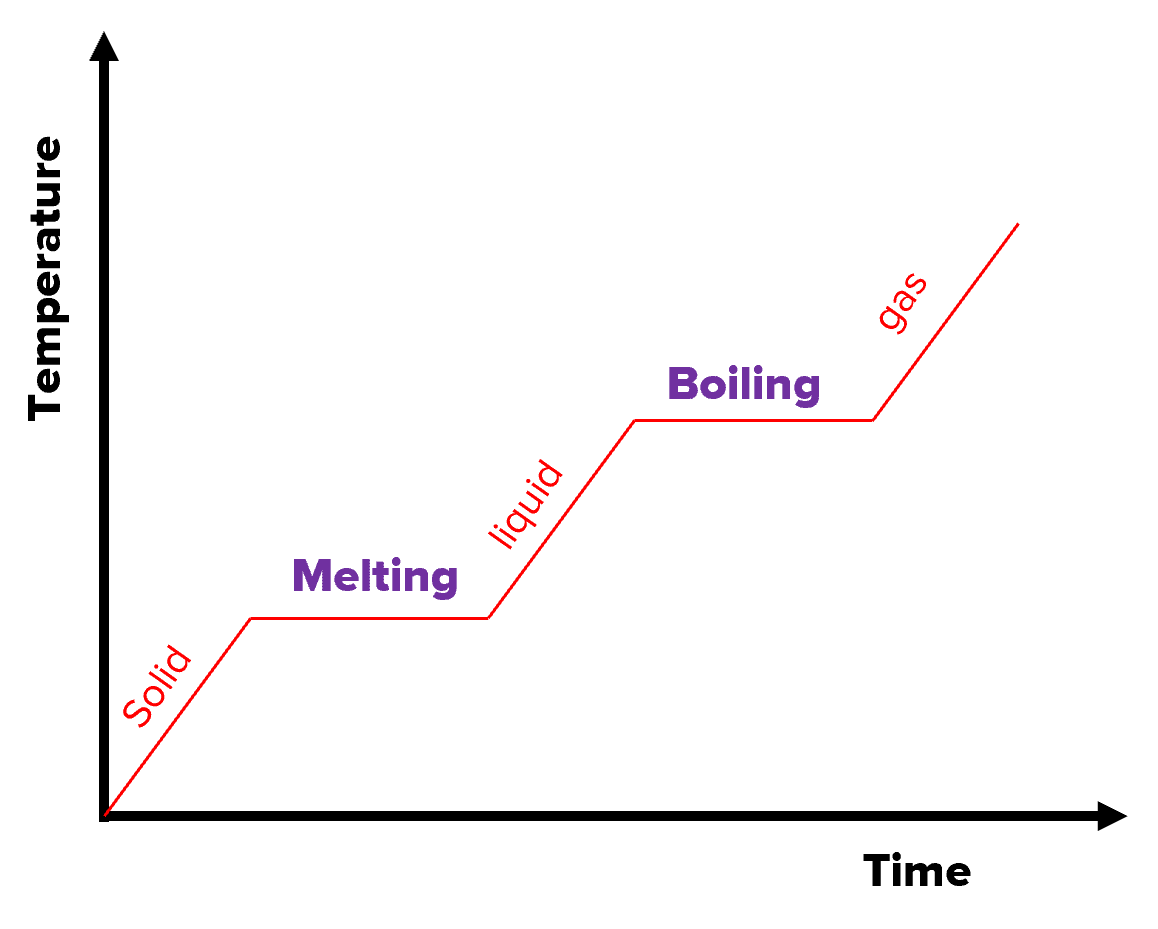
On A Heating Curve A Plateau Corresponds To

Ever watched a kettle get ready for its show-stopping performance? You know, that moment when it starts to rumble, then hiss, then eventually, gloriously, boils? Well, there’s a little magic happening inside, and it’s all about a special kind of pause. Think of it as the kettle’s backstage moment before the big steam-tastic finale.
Imagine you're on a gentle hike, and the path starts to ascend. You’re feeling the burn, your heart’s doing a little jig, and you’re getting warmer, right? That’s like heating something up. The temperature just keeps climbing, like you’re steadily reaching the summit. But then, suddenly, the path levels out. It becomes flat. You’re not going uphill anymore, but you’re not going downhill either. You’re just… there. This flat bit, this plateau on our hiking trail, is where things get really interesting. It’s not about you getting tired and stopping for a snack (though that's tempting!), it’s about something else entirely.
In the world of heating, this plateau is like a secret handshake between energy and matter. You’re still pouring energy into whatever you’re heating – that water in the kettle, maybe a block of chocolate you’re trying to melt for brownies (a noble cause, if ever there was one). But here’s the kicker: the temperature isn’t going up. It’s holding steady. It’s like the energy you’re adding is being used for a completely different, but equally important, job.
Must Read
It’s like the energy is busy changing outfits, not just getting hotter!
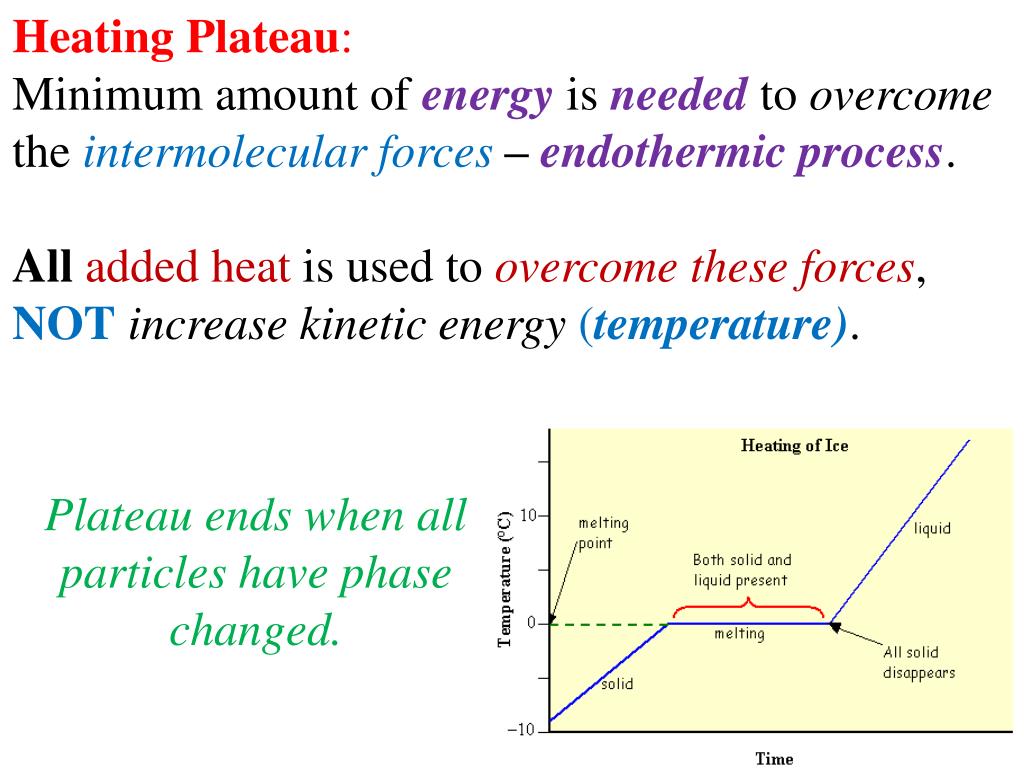
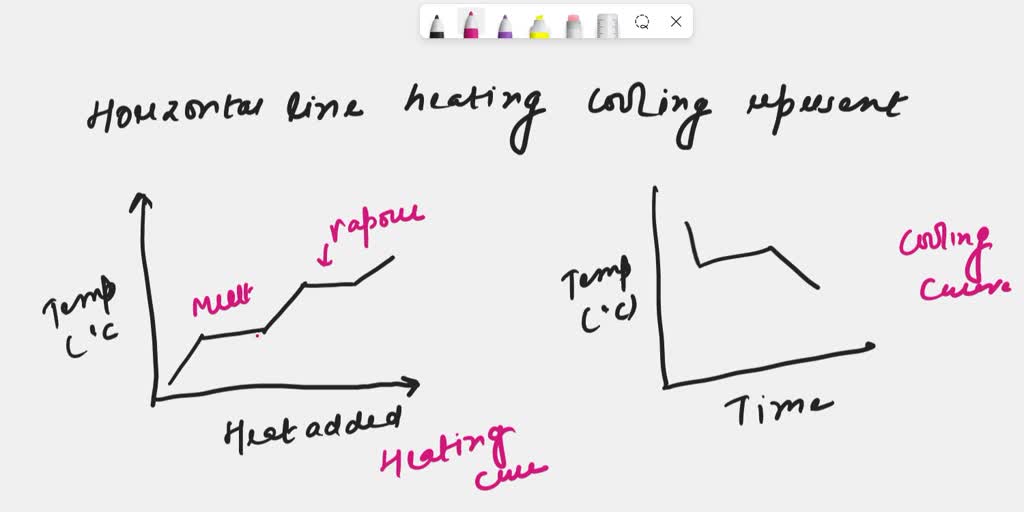
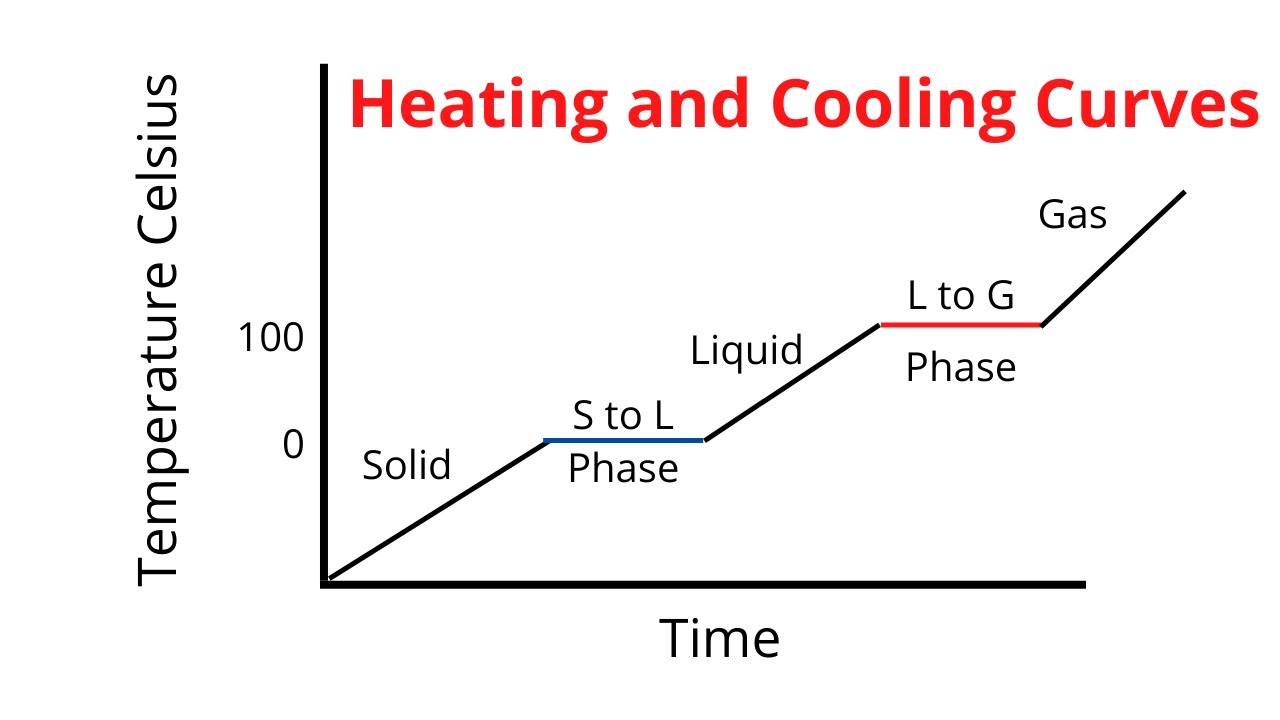
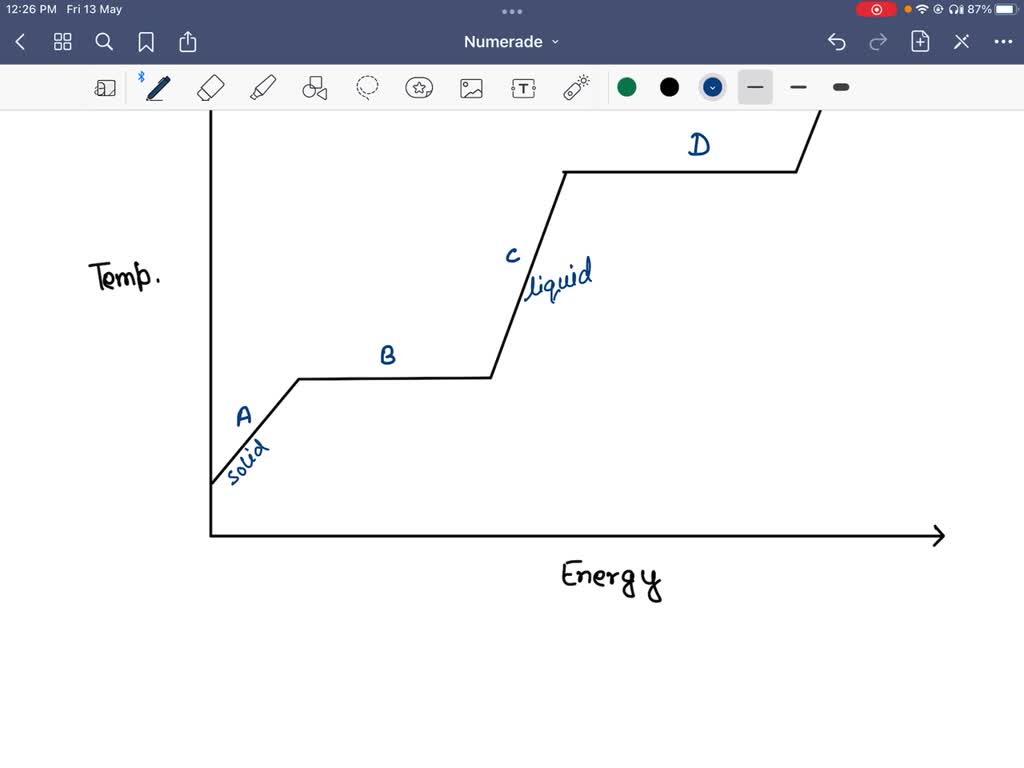
So, what’s this magical outfit change? It’s a phase transition. Think of your water in the kettle. It starts as lovely, liquid water. But as you heat it, it reaches a point where it decides, "You know what? I'm ready for a change. I want to be a fancy gas!" This transition from liquid to gas (or steam, in our kettle’s case) doesn’t happen instantly, like flicking a light switch. It takes time, and it takes energy. And during that time, that energy isn't busy making the water hotter. Oh no, it’s busy breaking the tiny, invisible bonds that hold the water molecules together in their liquid form, giving them the freedom to float around as a gas.

This is where the heartwarming comes in. Imagine all those tiny water molecules, huddling together as a liquid, all cozy and connected. The energy comes along, not to shove them around and make them jumpier (hotter), but to gently encourage them to let go of each other. It’s like a fond farewell, a permission slip to go explore the vastness of the air. The energy is the kind friend saying, "Go on, have an adventure!" And the temperature stays the same because everyone’s busy with this big, important life change. It’s a communal metamorphosis, and the plateau is the sign that the whole group is undergoing this transformation together.
Think about ice melting into water. That solid ice cube is chilling in your drink, keeping it cool. But as it absorbs heat, it doesn’t immediately become scalding water. No, it reaches a plateau. During this plateau, all the energy is being used to persuade those rigid ice molecules to loosen up, to break free from their frozen embrace and become the free-flowing liquid water we know and love. The temperature of the ice-water mixture stays exactly at 0 degrees Celsius (or 32 degrees Fahrenheit) until every last bit of ice has had its say and melted.

This is the secret life of everyday things! That chocolate bar you’re about to melt? When it hits its melting point, it’s not going to suddenly jump in temperature. It’s going to pause. That pause, that plateau, is the chocolate going through its transformation from solid to liquid. The energy is busy rearranging all those chocolatey molecules, giving them a new, more fluid arrangement. It’s a moment of delicious change, all happening at a constant temperature.
So, next time you see something boiling, or melting, or even freezing (because a plateau also happens when things change from gas to liquid, or liquid to solid – it’s a two-way street of transformation!), remember the hidden story. Remember the plateau. It’s not just a flat line on a graph; it’s a testament to the energy invested in change, the quiet, determined work happening at the molecular level. It’s the moment when matter decides to try on a new state of being, and the energy is there to help them do it, without rushing the process. It’s a beautiful, sometimes surprising, and definitely enjoyable part of the world around us, all happening on a level we can’t always see, but can certainly appreciate.