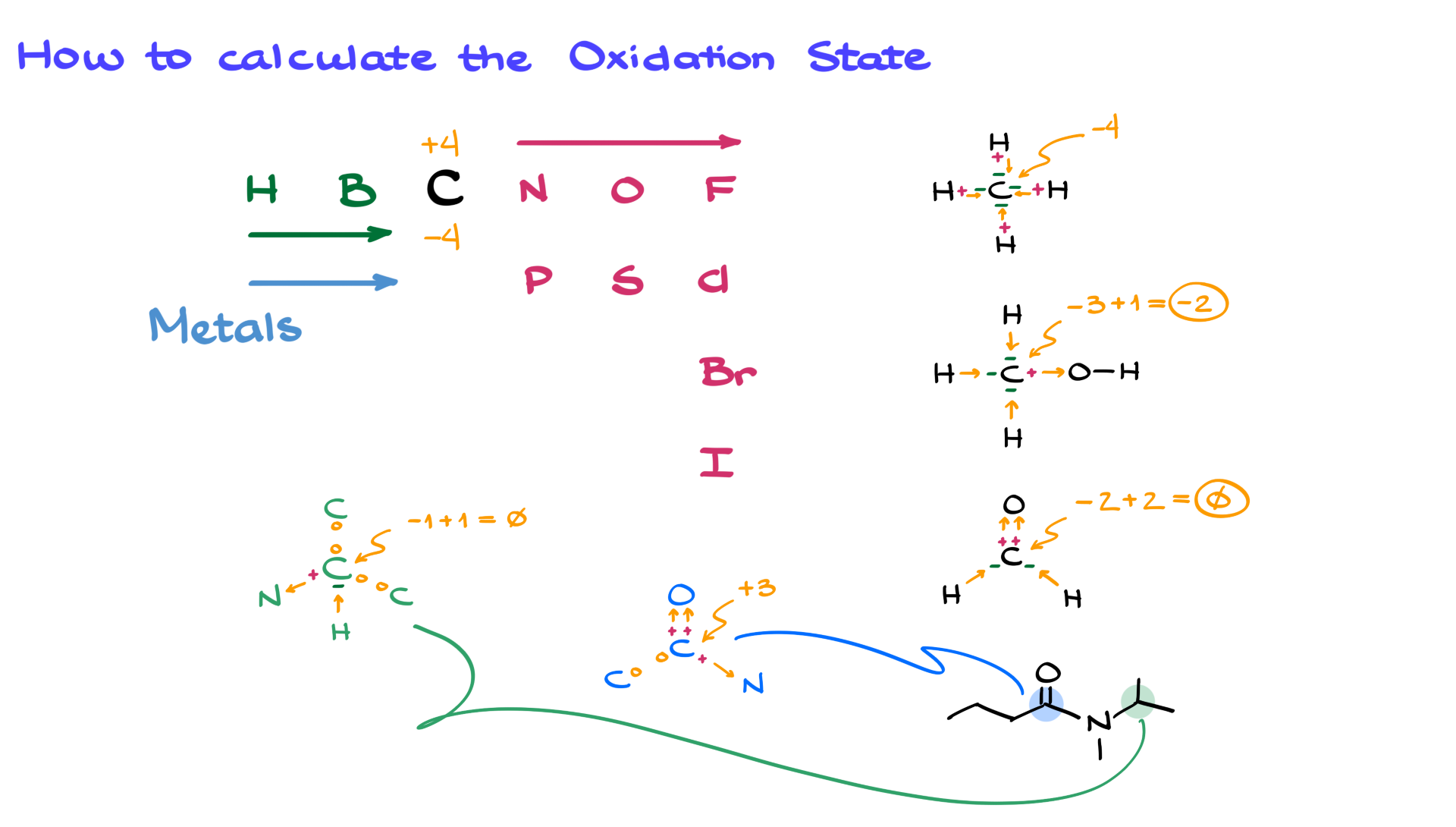
Determine The Oxidation State Of C In Co3 2

Hey there, you lovely chemistry enthusiast! Grab your mug, settle in, because we're about to dive into something super cool, but in a totally chill way. Today, we're gonna tackle a little puzzle: figuring out the oxidation state of carbon in something called Co32-. Sounds fancy, right? Don't worry, it's less complicated than assembling IKEA furniture, I promise!
So, oxidation states. What are they even? Think of them as this imaginary "charge" an atom would have if all its bonds were perfectly ionic. It's like assigning roles in a play, even if the actors are actually just cooperating. It helps us keep track of who's giving electrons away and who's hogging them. Crucial stuff for understanding how reactions actually work, you know?
And this particular ion, Co32-, looks a bit like a secret code. We've got the "Co" there. Now, my brain immediately goes, "Cobalt!" Right? That's a metal. And then there's the "3". And that little "2-"? That's a negative two charge for the whole thing. Easy peasy so far, right?
Must Read
But wait a minute! Hold the phone! If "Co" is cobalt, and cobalt is a metal, and we're talking about the oxidation state of carbon in this thing, that means there must be carbon in there somewhere! So, Co32-... is that really Cobalt and Oxygen? Or is it something else entirely? This is where the fun begins, folks!
Sometimes, chemical formulas can be a little bit sneaky. They don't always spell everything out in big, friendly letters. It’s like finding a hidden message in a crossword puzzle. The "Co" could also stand for something else. And in this case, my friends, it does! It stands for... wait for it... carbon!
Yes, you heard me! The "Co" in this context, especially when we're being asked about the oxidation state of carbon, is actually just representing carbon itself. So, Co32- is not some exotic cobalt compound. It's actually C32-! Mind. Blown. Right? It’s like realizing your favorite song artist is actually three people working together. A collective!
Now, why would they write it like that? Sometimes, older nomenclature or specific contexts might use "Co" for carbon. Or it could be a typo. Who knows! But for the sake of our sanity and solving this puzzle, we're going to assume that "Co" here means carbon.
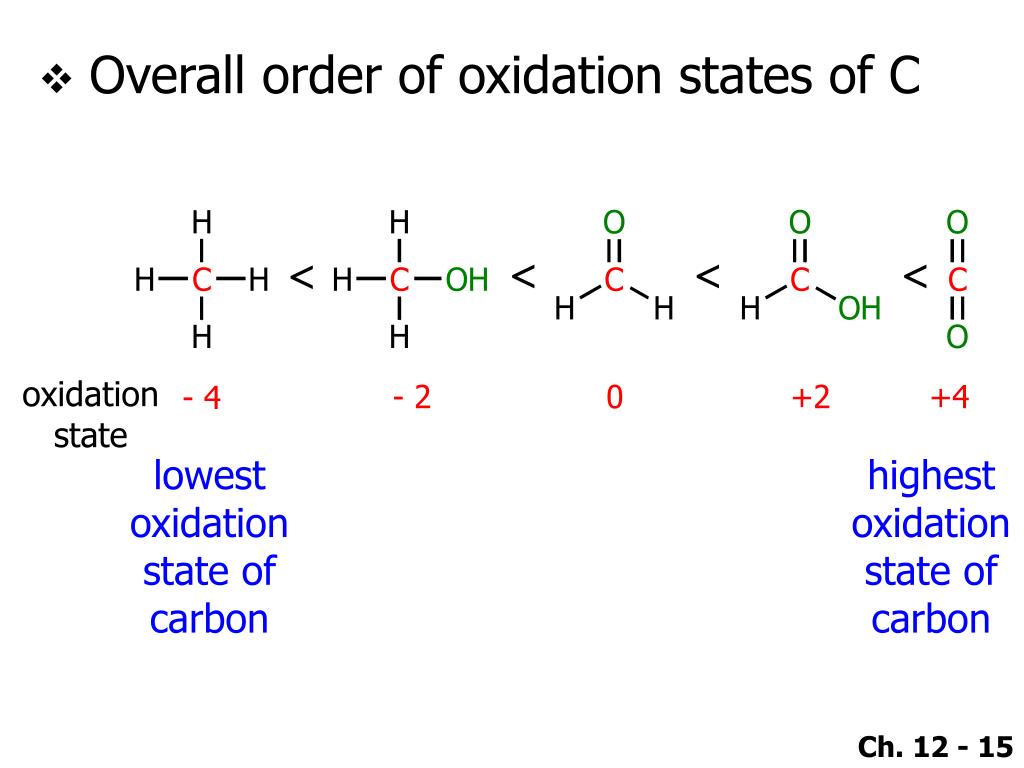
So, our mission, should we choose to accept it, is to find the oxidation state of carbon in the ion C32-. This ion is actually called the tricarbide ion. Sounds pretty hardcore, doesn't it? Like something from a sci-fi movie. Imagine tiny carbon atoms forming a little club and rocking a negative charge.
Okay, back to the rules of the game. We need some basic principles to guide us. First off, the sum of the oxidation states in a neutral molecule is always zero. Pretty straightforward. But we have an ion here, with a charge of -2. So, the total oxidation state of all the carbons in C32- must add up to -2. This is our golden ticket, our guiding star.
Another crucial rule: oxygen usually has an oxidation state of -2. Unless it's in a peroxide (like H2O2, where it's -1) or bonded to fluorine (which is super electronegative, making oxygen positive). But in most common compounds, oxygen is our dependable -2. And in C32-, there's no oxygen! So that simplifies things immensely. We only have carbon to worry about. Phew!
So, we have three carbon atoms. Let's call the oxidation state of each carbon atom 'x'. Because, let's be honest, these carbon atoms are probably all buddies in this little ion, so they're likely to have the same oxidation state. We're not expecting one carbon to be a superhero and another to be a villain, are we? Usually, in simple polyatomic ions like this, the atoms of the same element have the same oxidation state.
So, we have three carbons, each with an oxidation state of 'x'. And the total charge of the ion is -2. Plugging this into our trusty equation: 3 * (oxidation state of carbon) = total charge of the ion.

That translates to: 3x = -2.
Now, we just need to solve for 'x'. This is where our basic algebra skills come in handy. We divide both sides by 3.
x = -2 / 3.
And there you have it! The oxidation state of carbon in the C32- ion is -2/3.
Now, I know what you might be thinking. "Wait a minute! A fraction? Oxidation states are usually whole numbers, right?" Well, not always! That's the beauty of chemistry. It loves to keep us on our toes. Fractions can happen, especially when you have multiple atoms of the same element in an ion or molecule, and they're all sharing electrons in a way that results in an "average" oxidation state.
Think of it like splitting a pizza. If you have three friends (our carbon atoms) and you need to account for a charge of -2 (the overall "pizza deficit"), each friend is effectively responsible for -2/3 of that deficit. They're all contributing to the overall negative vibe, but in equal measure.
This fractional oxidation state is a strong indicator that the bonding in the tricarbide ion isn't purely ionic. It suggests there's a fair bit of covalent character going on. The electrons aren't completely transferred; they're being shared. And when you have delocalized electrons, or resonance structures (which is a whole other exciting topic!), you can end up with these averaged, fractional oxidation states.
The tricarbide ion, C32-, is actually quite interesting. It's not something you encounter every day in a standard introductory chemistry class. It's often found in certain intermetallic compounds or in complex organic structures. It's a testament to carbon's incredible versatility. It can do so many different things, form so many different bonds!
So, let's recap our little adventure. We started with a question mark about "Co32-". We figured out, with a little detective work, that "Co" in this context most likely means carbon. Our ion is actually C32-. We used the rule that the sum of oxidation states in an ion equals its charge. And we applied a bit of algebra to find that the oxidation state of each carbon atom in C32- is -2/3.
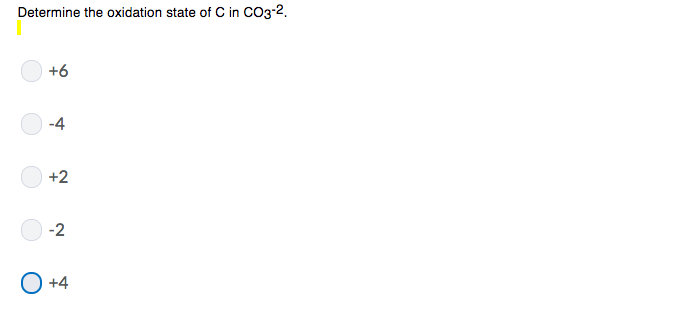
Isn't that neat? It's a little peek into the more complex world of bonding and charge distribution. It shows that not everything in chemistry fits neatly into whole numbers. Sometimes, the most interesting answers are found in the fractions, in the averages, in the shared electrons.
So, next time you see a weird chemical formula, don't panic! Take a deep breath, channel your inner Sherlock Holmes, and remember the fundamental rules. And if you're ever asked about "Co32-" and the oxidation state of carbon, you can confidently say, "-2/3"! You'll be the star of the chemistry party. Or at least the most knowledgeable person at the coffee shop.
It's this kind of unraveling that makes chemistry so fascinating. It’s like solving a puzzle, but the pieces are atoms and the picture is the way the universe works. And sometimes, the most unassuming symbols hide the most surprising truths. The "Co" for carbon? Totally unexpected, but totally makes sense once you know the context. It’s the hidden plot twist!
And remember, if you ever get stuck, just think about those three carbon atoms sharing the burden of that -2 charge. It's a team effort, a cooperative venture in the world of ions. They're not going it alone; they're in it together. And that’s what chemistry is all about, really. Atoms working together, sharing, transferring, and creating the amazing world around us. Even if it means one of them has to have a fractional oxidation state. It’s all part of the grand, complex, and utterly captivating dance of molecules.
So, there you have it. A little journey into the land of oxidation states, all thanks to a slightly ambiguous chemical formula. I hope you found it as fun and illuminating as I did. Now, go forth and impress your friends with your newfound knowledge of the tricarbide ion and its fractional carbon oxidation state. You’ve earned it! And maybe grab another coffee, because learning about chemistry is thirsty work, wouldn't you agree? Cheers to understanding the universe, one atom at a time!