Coulomb's Law Practice Problems Answers Key
Hey there, coffee buddy! So, you've been wrestling with Coulomb's Law, huh? Yeah, it can feel a bit like trying to herd cats sometimes. But guess what? You're not alone! And the good news is, we've got your back. Think of this as our little secret weapon for conquering those pesky practice problems. No more staring blankly at your notebook, right?
We're diving headfirst into the glorious world of Coulomb's Law practice problems, and more importantly, the answers key. Because let's be honest, sometimes figuring out how you got an answer is just as important as the answer itself. It's like, you know, finally understanding why your toast always lands butter-side down. A true revelation!
So, grab another sip of your latte, or whatever your caffeinated delight of choice is. Let’s break down some of those classic Coulomb's Law scenarios. You know, the ones with the charged particles zinging around, creating electric fields and forces that would make your hair stand on end (if they were strong enough, that is!).
Must Read
First off, let’s just do a quick refresher. What is Coulomb's Law, anyway? It's basically the granddaddy of all electric force laws. It tells us how much force exists between two charged objects. Pretty neat, huh? It’s like the universe's way of saying, "Hey, these charges are gonna interact!"
The formula itself looks a little something like this: F = k * (|q1 * q2|) / r². Don't let the letters scare you! 'F' is the force – the main event! 'k' is Coulomb's constant, a sort of cosmic speed limit for electric interactions. 'q1' and 'q2' are our little charged buddies, their magnitudes. And 'r' is the distance between them. Simple enough, right? Well, mostly.
The absolute value signs around `q1 * q2` are super important. Why? Because force has direction! And sometimes, our charges might be doing a little dance of repulsion or attraction. We'll get to that. But for the magnitude of the force, we’re just interested in how strong it is, not whether it’s pushing away or pulling in… yet.
Let's Kick Off with Some Straightforward Stuff
Imagine you have two charges, a positive one and a negative one, chilling out a certain distance apart. Problem number one, let's say: Charge q1 is +2 microcoulombs (µC) and charge q2 is -3 µC. They're separated by a distance of 0.5 meters. What's the force between them?
Okay, deep breaths. We’ve got our values! k is approximately 8.99 x 10⁹ N⋅m²/C². Our charges are in microcoulombs, so we need to convert them to Coulombs. Remember, 1 µC = 10⁻⁶ C. So, q1 = 2 x 10⁻⁶ C and q2 = -3 x 10⁻⁶ C. The distance, r, is already in meters, phew! 0.5 m.
Now, plug and chug into our trusty formula: F = (8.99 x 10⁹ N⋅m²/C²) * (|(2 x 10⁻⁶ C) * (-3 x 10⁻⁶ C)|) / (0.5 m)².

Let’s crunch those numbers. The product of the charges is -6 x 10⁻¹² C². Taking the absolute value, we get 6 x 10⁻¹² C². The square of the distance is (0.5 m)² = 0.25 m². So, F = (8.99 x 10⁹) * (6 x 10⁻¹²) / 0.25.
This should give you a force of approximately 0.21576 N. And what kind of force is it? Since we have a positive and a negative charge, they're gonna do the tango! It's an attractive force. So, the answer is 0.216 N (attractive). See? Not so scary when you break it down!
What About When Things Get a Little More… Energetic?
Sometimes, you'll have three charges, or even more, all playing the electric force game. This is where things can get a tad more exciting. You'll need to use the principle of superposition. Basically, it means you calculate the force on one charge due to each of the other charges individually, and then you add up all those forces as vectors. Vectors, my friends! It's like drawing little arrows of force and seeing where they all point in the end.
Let’s try a scenario: Charge q1 is +1 µC, q2 is -2 µC, and q3 is +3 µC. They’re all in a line, with q1 at the origin (0m), q2 at 0.2m, and q3 at 0.4m. What’s the net force on q2?
Alright, time for some vector vibes. We need to find the force q1 exerts on q2 (let's call it F12) and the force q3 exerts on q2 (let's call it F32). Then we’ll add them up.
Calculating F12:

q1 = 1 x 10⁻⁶ C, q2 = -2 x 10⁻⁶ C. Distance between them (r12) = 0.2 m. F12 = (8.99 x 10⁹) * (|(1 x 10⁻⁶) * (-2 x 10⁻⁶)|) / (0.2)²
F12 = (8.99 x 10⁹) * (2 x 10⁻¹²) / 0.04 F12 ≈ 0.4495 N
Now, the direction. q1 is positive, q2 is negative. They attract. Since q1 is to the left of q2, the attractive force will pull q2 to the left (towards q1). So, F12 is approximately 0.45 N to the left. Or, if we think of "right" as positive, it's -0.45 N.
Calculating F32:
q3 = 3 x 10⁻⁶ C, q2 = -2 x 10⁻⁶ C. Distance between them (r32) = 0.4m - 0.2m = 0.2 m. F32 = (8.99 x 10⁹) * (|(3 x 10⁻⁶) * (-2 x 10⁻⁶)|) / (0.2)²
F32 = (8.99 x 10⁹) * (6 x 10⁻¹²) / 0.04 F32 ≈ 1.3485 N
Direction time! q3 is positive, q2 is negative. They attract. q3 is to the right of q2, so the attractive force will pull q2 to the right (towards q3). So, F32 is approximately 1.35 N to the right. Or, in our positive-is-right convention, it's +1.35 N.

Net Force on q2:
Now we add our vector forces: Net Force = F12 + F32 = -0.45 N + 1.35 N = 0.90 N.
So, the net force on q2 is approximately 0.90 N to the right. Ta-da! See? You’re basically a force-wielding superhero now.
Sometimes Things Get a Little… Angular
What if your charges aren't in a neat little line? What if they form a triangle, or a square? That's when things get really interesting, and you might need to break down your forces into x and y components. It’s like solving a tiny physics puzzle for each force!
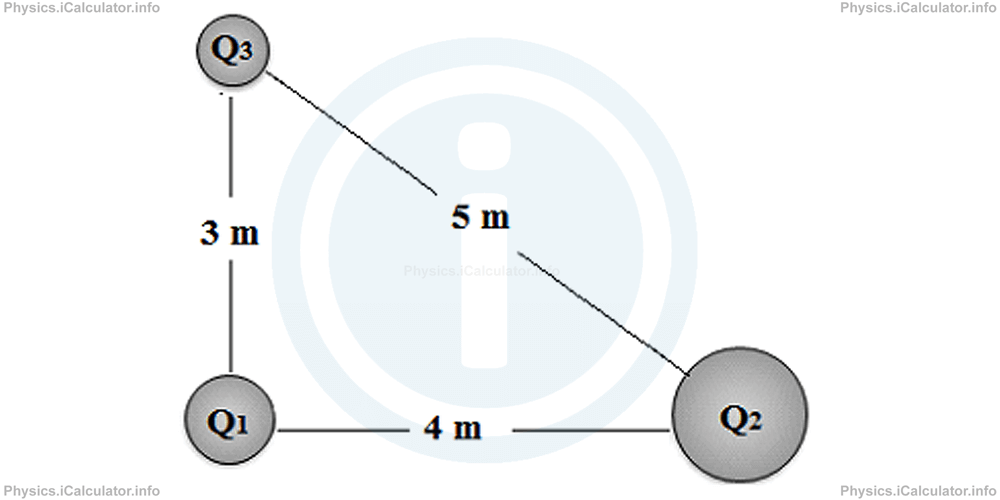
Let's say you have three charges forming an equilateral triangle. Charge q1 (+5 µC) at the origin, q2 (-3 µC) along the positive x-axis at a distance 'd', and q3 (+2 µC) forming the third vertex of the triangle. We want the net force on q1.
This requires a bit more geometry and trigonometry. You'd calculate the force between q1 and q2 (F12), and the force between q1 and q3 (F13). F12 would be purely along the x-axis (or y, depending on orientation, but let's assume q2 is on the x-axis). F13, however, would have both an x and a y component because of the angle.
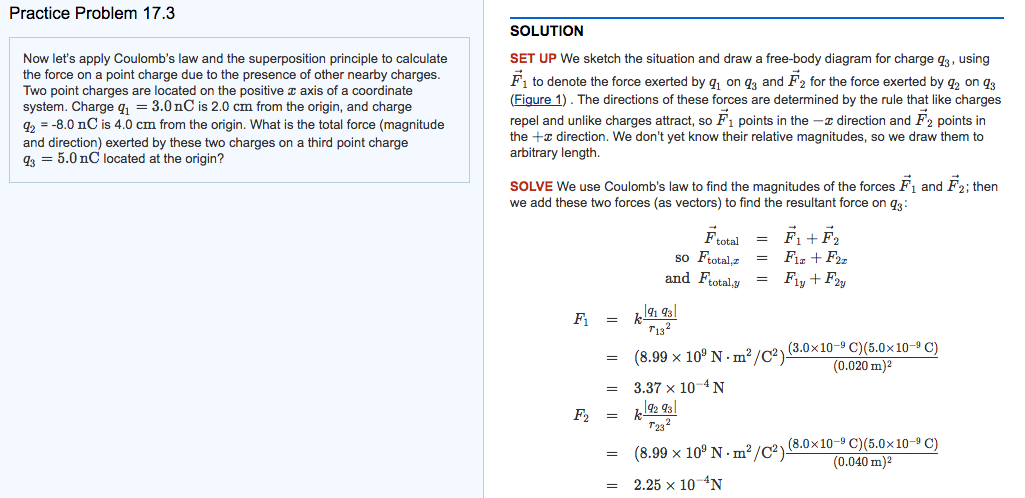
For an equilateral triangle, each angle is 60 degrees. So, the force F13 would be at a 60-degree angle to the x-axis. You'd use `F13x = F13 * cos(60)` and `F13y = F13 * sin(60)`.
Then, you'd add the x-components of F12 and F13 to get the total x-force, and the y-component of F13 to get the total y-force. Finally, you'd use the Pythagorean theorem to find the magnitude of the net force: F_net = sqrt(Fx_total² + Fy_total²). And of course, you'd find the direction using arctangent!
Don't worry if this sounds a bit daunting. Practice is your best friend here. The more problems you work through, the more natural the vector decomposition becomes. It’s like learning to ride a bike; a few wobbles at first, but then you’re cruising!
Key Takeaways from Our Little Answer Session
So, what have we learned from our coffee-fueled romp through Coulomb's Law? A few golden nuggets to keep in your physics toolkit:
- Always check your units! Microcoulombs? Nanocoulombs? Meters? Kilometers? Make sure everything is in the standard units (Coulombs and meters) before you plug anything in. It's the number one way to avoid those silly mistakes.
- Pay attention to the signs of the charges. Positive and negative mean attraction. Two positives or two negatives mean repulsion. This determines the direction of the force. Don't forget this!
- Vector addition is your friend (and sometimes your enemy). For multiple charges, you must consider forces as vectors. Break them down into components if they aren’t in a straight line.
- The inverse square law is key. That `r²` in the denominator means doubling the distance reduces the force by a factor of four! It’s a pretty significant effect.
- Coulomb’s constant, k, is your trusty sidekick. Keep its value handy: 8.99 x 10⁹ N⋅m²/C².
And the most important takeaway of all? Don't be afraid of the practice problems! They are designed to help you learn. Every single one you tackle, even if you get it wrong, is a step in the right direction. The answers key is not to cheat; it's to verify and understand.
If you get an answer that doesn't match the key, don't just shrug it off. Go back through your steps. Did you mess up a calculation? Did you forget to convert units? Did you mix up attraction and repulsion? Troubleshooting is a vital skill in physics, and in life, really!
Think of each practice problem as a mini-adventure. You're exploring the invisible forces that hold the universe together. How cool is that? So next time you’re faced with a Coulomb's Law problem, remember this chat. Take a deep breath, channel your inner physics guru, and remember that the answers key is just a guide, not the destination. The real destination is your own understanding. Now go forth and conquer those charges!