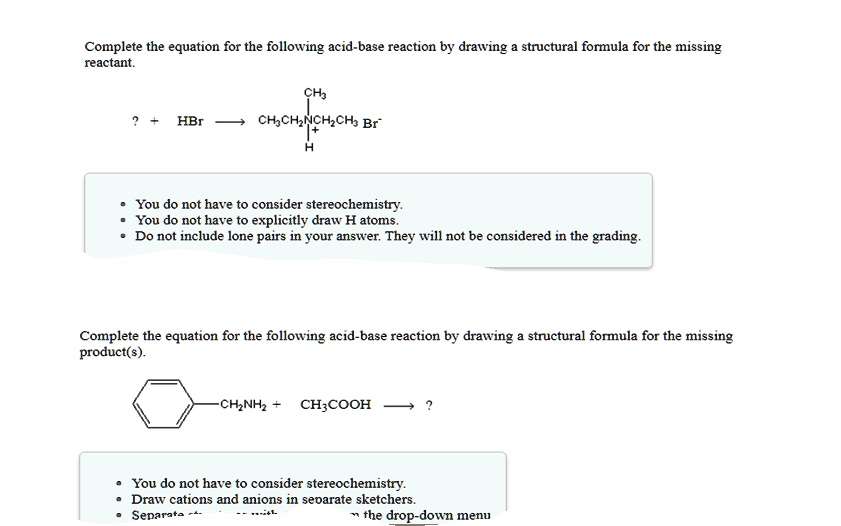
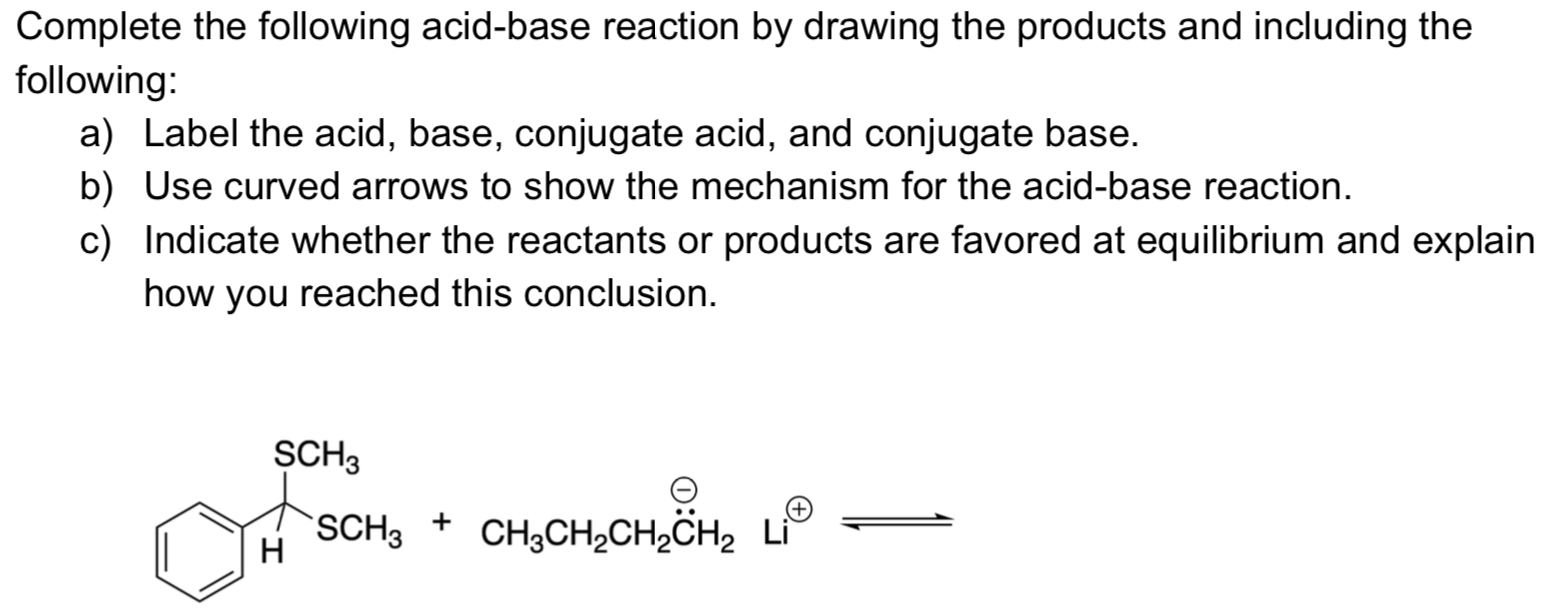
Complete The Following Acid Base Reaction
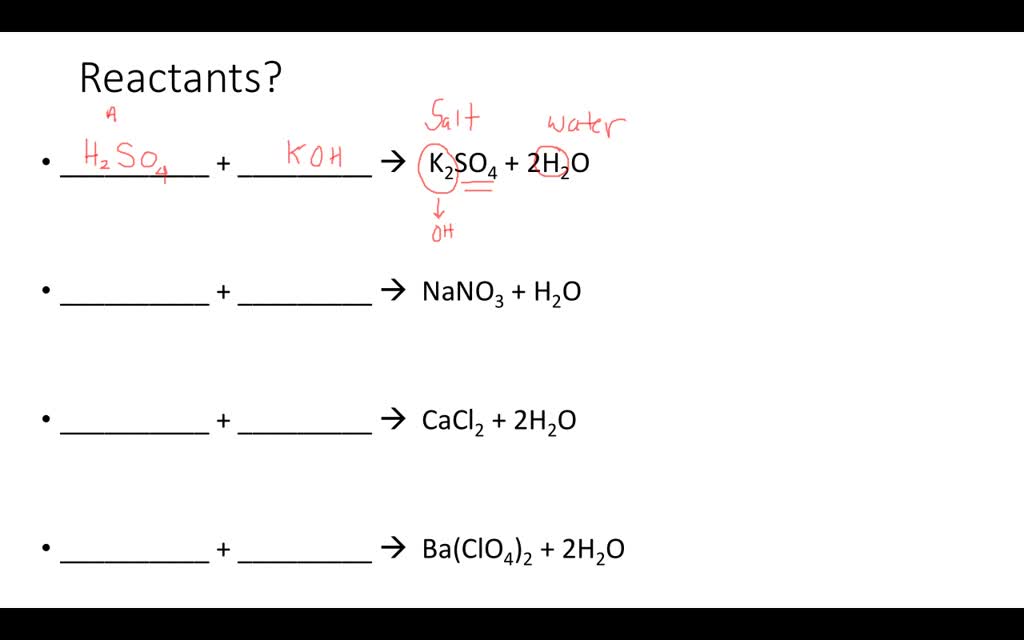
Hey there, science curious cats! Ever feel like you're staring at a jumble of letters and symbols and wondering, "What in the fizzing beaker is going on here?" That's totally me sometimes. Especially when we dive into the wild world of
Think of it like this: acids are like the
So, let's get our hands (metaphorically, of course!) a little dirty. We’re going to tackle a classic. Imagine you’ve got a bit of
Must Read
The Usual Suspects: Acids and Bases
Acids are basically molecules that have a proton (H+) they're itching to get rid of. They make solutions taste sour (don't try this at home, seriously!). Think lemon juice, vinegar. They can also be a bit
Bases, on the other hand, are the opposite. They’re typically the ones that
When an acid and a base get together, they do this awesome thing called
Let's Play "Complete the Reaction!"
Our mission, should we choose to accept it, is to complete this reaction:
HCl + NaOH → ?
So, we’ve got our acid,
Remember how acids like to
And our base, NaOH? It's like a welcoming committee. It’s got a
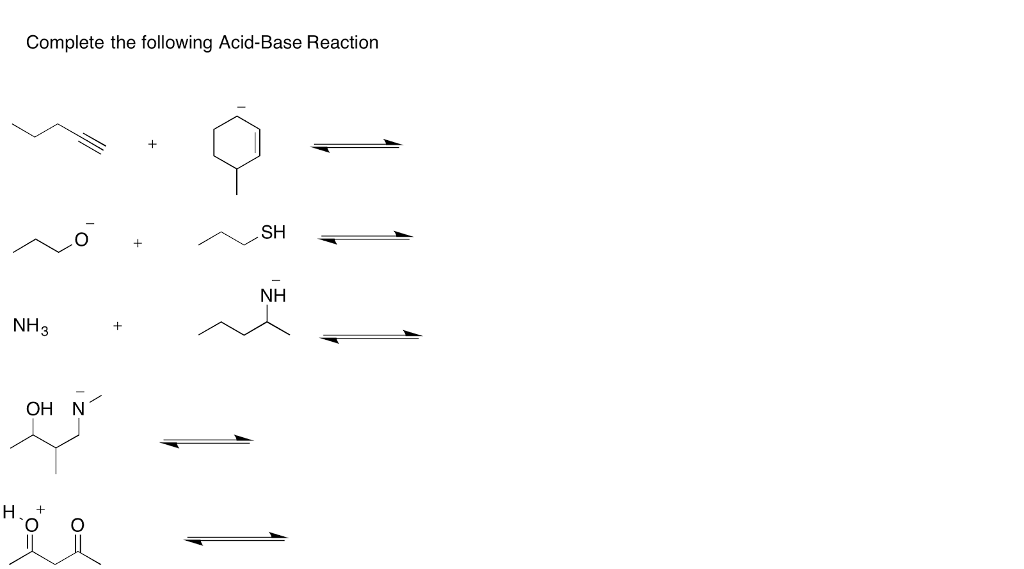
The Big Moment: The Proton Swap!
So, what happens when HCl and NaOH meet? It’s a beautiful dance. The H+ from HCl sees the OH- from NaOH and thinks, "Hey, you look like you need a friend!" And the OH- is all like, "Finally! I’ve been waiting for you!"
The H+ and the OH- get together and form… wait for it…
But what's left behind? The HCl lost its H+. All that’s left is the

The Leftovers: Not So Boring After All!
So now we have Na+ and Cl-. What do they do? They’re both
The positive Na+ and the negative Cl- come together and form…
Isn't that wild? From a zesty acid and a calming base, we get the two most fundamental things: water and the seasoning for our popcorn. It’s like nature’s ultimate
The Complete Picture: A Chemical Symphony
So, to recap our epic reaction:
HCl + NaOH → H₂O + NaCl
Hydrochloric acid plus sodium hydroxide gives us water and sodium chloride. It’s a perfect example of a
Why is this so fun to talk about? Because it shows how seemingly complex chemical reactions are actually just elegant interactions. It's all about
Think about it: the pH of our bodies, the way food digests, even the bubbles in your soda – they all involve acid-base chemistry. It’s everywhere! It’s the
So next time you see an acid and a base hanging out, remember they're not just chemicals. They're potential ingredients for something new, something essential. They're the building blocks of our world, having little chemical parties and making magic happen. And that, my friends, is something worth celebrating with a little bit of scientific curiosity!



![[Solved]: Complete the equation for the following acid-bas](https://media.cheggcdn.com/study/1de/1de71575-ef3e-42c3-b348-b536f09e3e14/Screenshot2024-07-10at2.58.38AM.png)