Complete An Orbital Diagram For Scandium Sc

Alright, science adventurers, get ready to dive into the wonderful world of atoms! Today, we're going to tackle something super cool: completing an orbital diagram for our friend, Scandium, or Sc. Now, before your eyes glaze over, think of this like decorating your atom's house! We've got these special rooms, called orbitals, where the little electrons, our energetic party guests, like to hang out. And just like in any good party, there are rules for who gets to sit where!
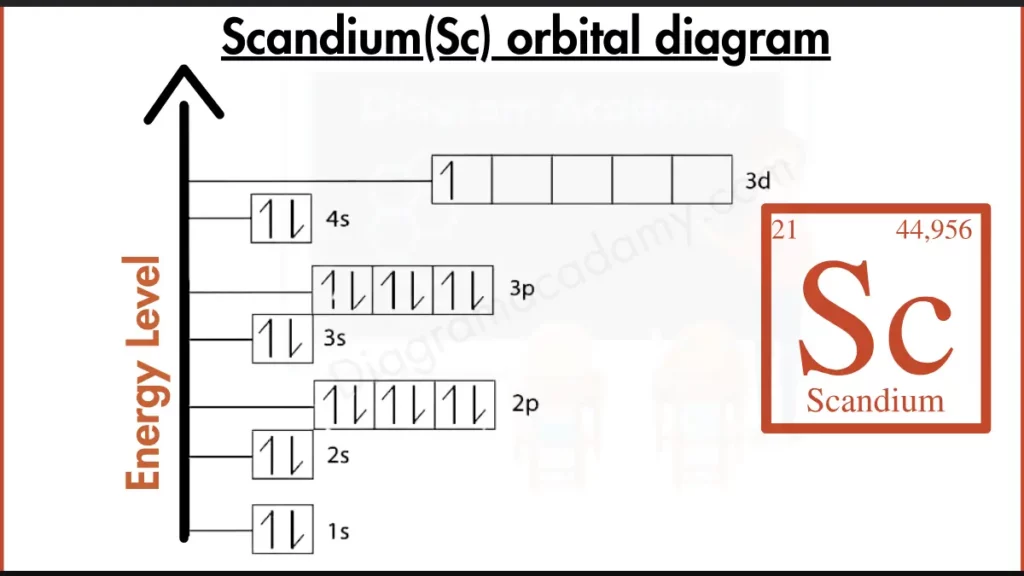
So, our star of the show today is Scandium. Imagine it's a brand-new apartment building, and we need to figure out where all the residents (electrons) are going to live. Scandium, bless its atomic heart, has 21 electrons. That's a lot of tiny energetic beings to find homes for! Our orbital diagram is essentially the blueprint for this atomic mansion, showing us exactly how these electrons are arranged.
First off, we have the 1s orbital. Think of this as the cozy studio apartment right on the ground floor. It's small, but it's the first one available, so it's super popular. This orbital can hold a maximum of two electrons. They're like the two best friends who always want to share a tiny space – they snuggle up together! So, we'll put two electrons in our 1s orbital. Yay, first room is full!
Must Read
Next up, we have the 2s orbital. This is like the next-door studio apartment, still on the ground floor, a little bit bigger maybe, but still very desirable. It also has enough room for two electrons. So, those two are going to hop in there and make themselves at home. We're filling up the ground floor nicely!
Now, things get a little more interesting. After the 2s, we move to the 2p orbitals. These are like a set of three connected, slightly nicer apartments on the second floor. The cool thing about 2p orbitals is that there are three of them, and each one can hold two electrons. That means the 2p level can accommodate a total of six electrons! It's like a little suite with three bedrooms, perfect for a slightly larger group of friends who like their personal space. So, we'll fill up all three 2p orbitals with our electrons. We're on the second floor now, feeling fancy!

We're still counting our electrons, remember? We've used 2 (in 1s) + 2 (in 2s) + 6 (in 2p) = 10 electrons so far. We've got 11 more to go! Scandium isn't done decorating yet!
Moving on, we hit the 3s orbital. You guessed it! This is like another ground-floor-style apartment, maybe a bit further down the hall, but still a prime spot. It can hold another two electrons. So, in they go! Now we're at 10 + 2 = 12 electrons placed.

Then comes the 3p orbitals. Just like the 2p orbitals, there are three of these, and each can hold two electrons, for a grand total of six electrons. So, we'll fill up all six spots in the 3p orbitals. We're really starting to pack this atomic mansion! Now we've placed 12 + 6 = 18 electrons.
We're almost there! We have 21 electrons total, and we've only placed 18. That means we have three more electrons to find homes for. Where do they go? They're heading to the 4s orbital. Now, this is a bit of a surprise in the atomic world! Even though the 3d orbitals are "next" in line in terms of energy level, the 4s orbitals are filled first. It’s like there’s a special express elevator to the 4s that gets used before the slightly slower stairs to the 3d. So, our 4s orbital, like the 1s and 2s, can hold two electrons. We'll pop two of our remaining three electrons in there. That makes 18 + 2 = 20 electrons placed. Just one more!

And where does that very last electron go? It’s heading to the next available energy level, which happens to be the very beginning of the 3d orbitals. The 3d orbitals are a set of five different rooms, and each can hold two electrons. But since we only have one electron left, it gets its own little private room for now. It's like the last guest arriving at the party and getting the best single room with a view! So, that final electron settles into one of the 3d orbitals.
And there you have it! Our complete orbital diagram for Scandium. We’ve successfully housed all 21 of its electrons. You've navigated the energy levels, the different orbital shapes (s, p, and d), and the rules of electron occupancy. You're basically an atomic interior designer now! Isn't that amazing? You've just visualized the very heart of an element, and it’s all thanks to these handy orbital diagrams. High fives all around for tackling Scandium!