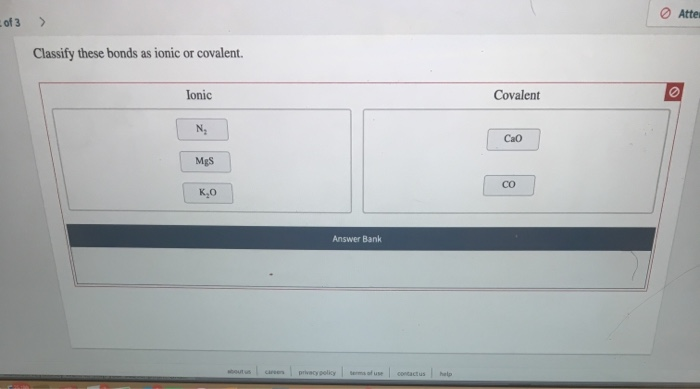
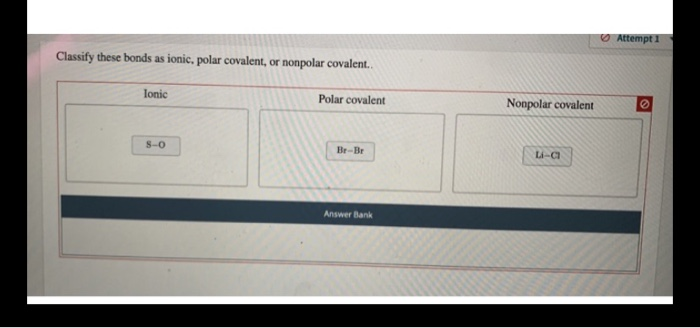
Classify These Bonds As Ionic Or Covalent

Hey there, awesome science explorers! Ever looked at the world around you and wondered what makes it tick? Like, why is water wet? (Okay, that's a bit complex for this chat, but you get the idea!) Today, we're diving into something super cool: bonds! Not the kind you break, but the kind that hold atoms together. And guess what? We're going to figure out if they're ionic or covalent. It sounds fancy, but trust me, it's like a fun puzzle.
Think of atoms as tiny LEGO bricks. They love to connect with other LEGO bricks to build everything we see. These connections? That's what we call bonds. And there are two main ways they like to stick together. It's like different personalities in the atom world!
Ionic Bonds: The "Give and Take" Crew
First up, we have the ionic bond. Imagine two atoms who are, well, a little different in how much they like their electrons. Electrons are like tiny, energetic kids running around the atom. Some atoms are super keen to hold onto their electrons. Others? They're practically begging to give them away.
Must Read
In an ionic bond, you have a "giver" atom and a "taker" atom. The giver is happy to say, "Here, you can have this electron!" And the taker is like, "Yes! Thank you! I've been wanting one of those!"
This electron transfer creates a really strong attraction. It's like magnets! One atom becomes positively charged (because it lost a negative electron) and the other becomes negatively charged (because it gained a negative electron). Opposite charges attract, right? Boom! Ionic bond formed.
Think of table salt. That's sodium chloride (NaCl). Sodium (Na) is a happy giver, and chlorine (Cl) is a very eager taker. Sodium gives an electron to chlorine. Now you have Na+ and Cl-. They hug super tight and form those crunchy salt crystals we love on our fries!
Quirky Fact Alert! Ionic compounds often form beautiful crystals. It’s like they’re showing off their perfectly organized structure. It’s a testament to their strong, electrostatic love affair.
Another example? Calcium fluoride (CaF2). Calcium is a giver, and fluorine is a taker. Calcium gives electrons to fluorine atoms. Hello, fluoride for your teeth! Those white, sparkly pearls are thanks to ionic bonds.

The key takeaway for ionic bonds: one atom completely gives an electron to another. It's a bit dramatic, but hey, it works!
Covalent Bonds: The "Sharing is Caring" Squad
Now, let's meet the covalent bond crew. These guys are more into cooperation. Instead of one atom snatching an electron, they decide to share. They say, "You know what? Let's just hold hands and keep our electrons together."
This happens when atoms have a similar desire for electrons. Neither wants to give one up entirely, and neither wants to take one entirely. So, they compromise!
Imagine two friends who both want to play with the same cool toy. Instead of fighting, they decide to share it. That's a covalent bond. The shared electrons orbit around both atoms, holding them together.
Water! You know, that stuff that makes up most of you? That's H2O. It's made of one oxygen atom and two hydrogen atoms. Oxygen and hydrogen are covalent buddies. They share electrons. This sharing is what makes water molecules so stable and essential for life.

Think about the air you breathe. Oxygen gas (O2) is two oxygen atoms covalently bonded. They share their electrons. They're happy together. Fun fact: Without these covalent bonds, we wouldn't have air to breathe!
Another awesome example is methane (CH4), the main component of natural gas. Carbon and hydrogen atoms share electrons. It's a beautiful dance of shared electrons that powers our stoves and heats our homes.
Covalent bonds can be single, double, or even triple! That just means they're sharing one pair of electrons (single), two pairs (double), or three pairs (triple). It's like they're holding hands, holding arms, or even doing a full group hug!
The big difference here: atoms share electrons. It's a much more collaborative vibe.
So, How Do We Classify Them?
Alright, the moment of truth! How do you know if you're looking at an ionic or covalent bond? It all comes down to who is involved.
Generally, if you have a metal and a nonmetal, you're probably looking at an ionic bond. Metals are the givers, and nonmetals are the takers. Think sodium (metal) and chlorine (nonmetal) for salt.

If you have two nonmetals, it's usually a covalent bond. They're both "taker-ish" so they share. Think oxygen and hydrogen in water, or carbon and hydrogen in methane.
A quick peek at the periodic table is your best friend here! Metals are usually on the left side, and nonmetals are on the right (with hydrogen being a sneaky nonmetal even though it's on the left).
Let's try a few! What about potassium iodide (KI)? Potassium is a metal, iodine is a nonmetal. That's right, ionic! Potassium gives an electron to iodine.
How about carbon dioxide (CO2)? Carbon is a nonmetal, oxygen is a nonmetal. You guessed it, covalent! They share electrons.
What about magnesium oxide (MgO)? Magnesium is a metal, oxygen is a nonmetal. Bingo! Ionic. Magnesium gives electrons to oxygen.

It's like a fun game of "Is it a giver or a sharer?"
Why Is This Fun?
Because understanding these bonds unlocks the secrets of the universe! Seriously! From the salt on your food to the water you drink, from the air you breathe to the materials that build our cities, it's all about how atoms connect.
It's a fundamental concept that explains so much. It's like learning the alphabet before you can read a book. And the world of chemistry is a super fascinating book!
Plus, it's a great way to impress your friends at your next dinner party. "Did you know that your delicious steak is held together by ionic and covalent bonds?" They'll be like, "Whoa!"
So next time you see something, anything, take a moment to think about the tiny atoms inside and how they're holding hands (or giving electrons) to make it all happen. It's a little bit of magic, powered by science!
Keep exploring, keep questioning, and keep having fun with the amazing world of chemistry. You're already halfway there!