Choose The Best Lewis Structure For Icl 5.
Hey there, science enthusiasts and the just plain curious! Ever looked at a molecule and wondered, "What's going on inside that tiny world?" Well, today we're going to peek into the secret life of a molecule called ICl5, or Iodine Pentachloride. Now, I know what you might be thinking: "Why should I care about some fancy chemical formula?" Stick with me, because understanding these little guys can actually be pretty fun, and it's like solving a tiny puzzle that helps us understand the bigger picture of how stuff works around us.
Think of molecules like tiny LEGO structures. We have different colored bricks (atoms) that we can put together in various ways to build something. Sometimes, there are a few different ways to connect those bricks, and we need to figure out which way is the most stable and most likely to exist. That's where Lewis structures come in. They're basically simplified drawings that show us how the electrons, those super energetic little particles, are arranged around the atoms. It's like a blueprint for our molecular LEGOs!
Our star today is ICl5. It's made of one iodine atom (I) and five chlorine atoms (Cl). Now, just like trying to stack too many books on a wobbly table, atoms prefer to arrange themselves in ways that are as stable as possible. They want to feel "comfortable," and in the atomic world, this often means having a certain number of electrons around them. This is where the octet rule comes in, kind of like a golden rule of electron sharing. Most atoms like to have eight electrons in their outer shell, like a perfectly full candy jar!
Must Read
But here's where it gets interesting. Sometimes, atoms can be a bit greedy or, shall we say, generous, and end up with more than eight electrons. This is especially true for larger atoms, like iodine. Iodine is like the big sibling in the family, happy to share or even take on a few extra electrons to make everyone else feel settled. Chlorine, on the other hand, is more of a stickler for the octet rule. It's like the younger sibling who just wants their own neatly portioned snacks.
So, when we look at ICl5, we have one iodine atom and five chlorine atoms all trying to figure out how to share their electrons. This can lead to a few different possible Lewis structures, just like you could build a LEGO tower in a couple of slightly different ways. We have to decide which structure is the best, the one that makes the most sense chemically. It's like choosing the most balanced way to arrange your spice rack – you want everything to be easily accessible and not about to tumble over.
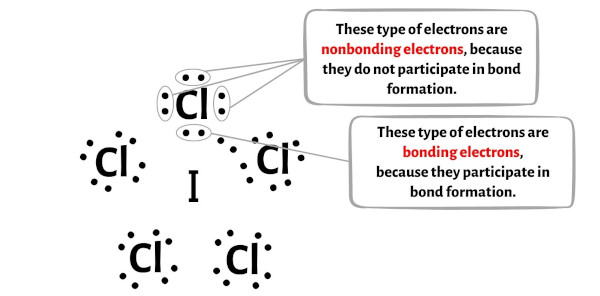
The main players in this electron dance are the valence electrons. These are the electrons on the outermost shell of an atom, and they're the ones that do all the bonding. Iodine has 7 valence electrons, and each chlorine atom also has 7 valence electrons. So, in total, we have 7 (from iodine) + 5 * 7 (from chlorine) = 42 valence electrons to work with. That's a lot of electrons to arrange!
Now, let's think about how these atoms might connect. We know the iodine is likely to be in the center, surrounded by the five chlorine atoms. Imagine iodine as the sun and chlorine atoms as planets orbiting it. But how are these planets arranged? And how are the electrons being shared?
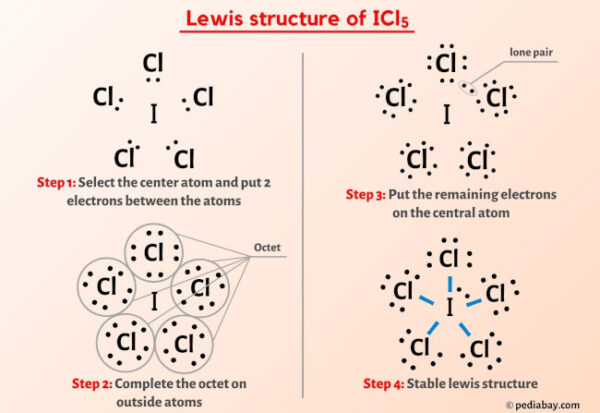
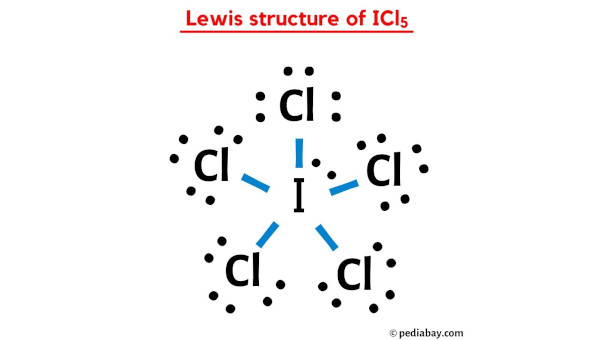
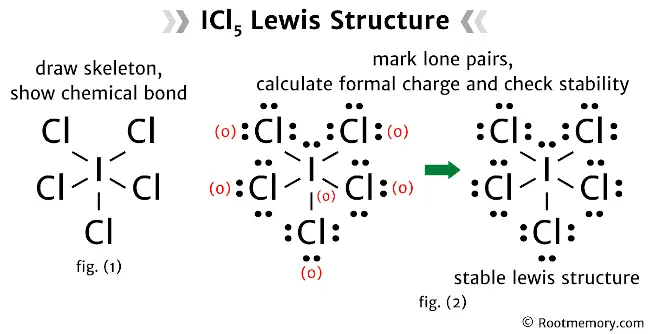
One way to think about it is to give each chlorine atom a single bond to the iodine. This uses up 5 * 2 = 10 electrons. Each chlorine atom would then need 6 more electrons to complete its octet, which is 5 * 6 = 30 electrons. So far, we've used 10 + 30 = 40 electrons. We have 42 total, so we have 2 electrons left over. These two leftover electrons are super important! They form what we call a lone pair. Think of it like a little extra cushion of electrons hanging out near the iodine atom.
So, in this arrangement, iodine is bonded to five chlorine atoms, and it also has that one lone pair of electrons. This means iodine has a total of 5 * 2 (from bonds) + 2 (from lone pair) = 12 electrons around it. Whoa! That's more than the magic number eight! But remember, iodine is a bigger atom, and it can handle having an expanded octet. This is perfectly fine and actually quite common for elements in the third row and beyond in the periodic table. It’s like having a bigger plate – you can fit more food on it!
Now, let's consider if there are other possibilities. Could we have double bonds? If we tried to form double bonds, we'd be messing with the octet rules for chlorine, which it really likes to follow. Plus, it would create more electron density around the iodine, which, while it can expand its octet, might lead to a less stable situation overall if we can achieve stability with single bonds and lone pairs.

Another way to compare is by looking at formal charges. Formal charge is a way to track electrons and see if any atom is carrying a "debt" or "credit" of electrons. The goal is to have formal charges as close to zero as possible. In our favored structure, with single bonds and the lone pair on iodine, the formal charges are all zero! This is a big win for stability. If we were to try other arrangements, we might end up with some atoms having positive or negative formal charges, which is like having an unpaid bill – generally something you want to avoid.
So, why should we care about this? Well, the Lewis structure tells us a lot about the molecule's shape and how it will interact with other molecules. The presence of that lone pair on iodine is like a little lump or a secret ingredient that influences how ICl5 behaves. It’s the reason why this molecule is not perfectly symmetrical. Imagine trying to balance a perfectly round ball versus a ball with a little dent in it. The dent makes a difference in how it rolls!
Understanding these structures helps scientists predict how molecules will react, what properties they'll have, and even how they can be used in things like pharmaceuticals or materials science. It’s like knowing the ingredients and the cooking method for a recipe – it tells you what delicious (or sometimes not-so-delicious!) dish you're going to end up with.
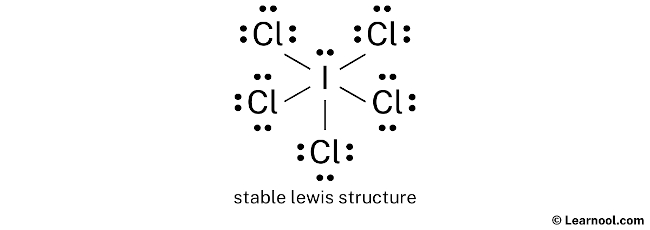
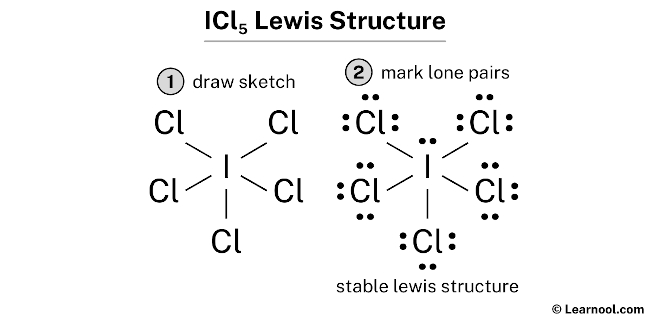
For ICl5, the best Lewis structure shows a central iodine atom single-bonded to five chlorine atoms, with one lone pair of electrons residing on the iodine. This arrangement minimizes formal charges and allows iodine to accommodate its electron needs with an expanded octet, making it the most stable and likely configuration. It's a testament to the fact that even in the microscopic world, there's a constant striving for balance and stability, just like in our own lives when we try to juggle work, family, and a little bit of "me time."
So, the next time you see a chemical formula, remember that it’s not just a jumble of letters and numbers. It's a tiny, intricate puzzle, and understanding its Lewis structure is like unlocking the secret to its personality and its potential. And who knows, maybe by understanding these molecular puzzles, we can even solve some bigger puzzles in the world around us!