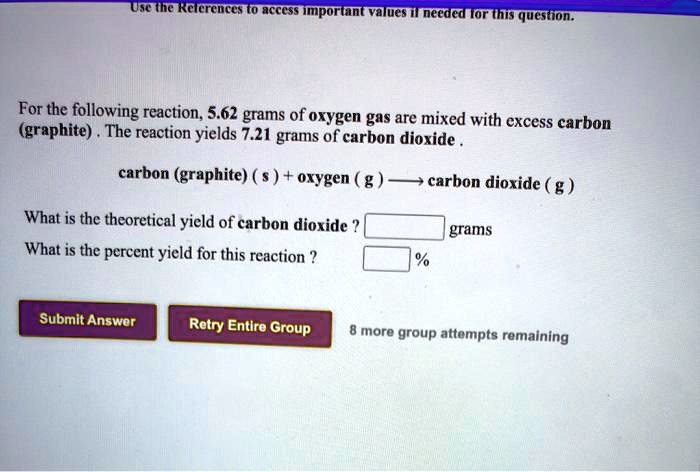
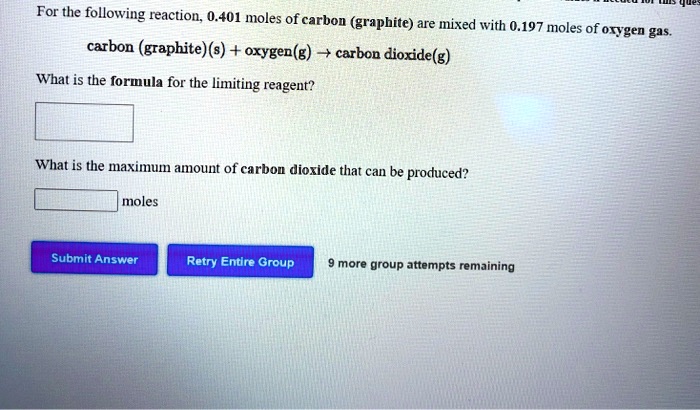
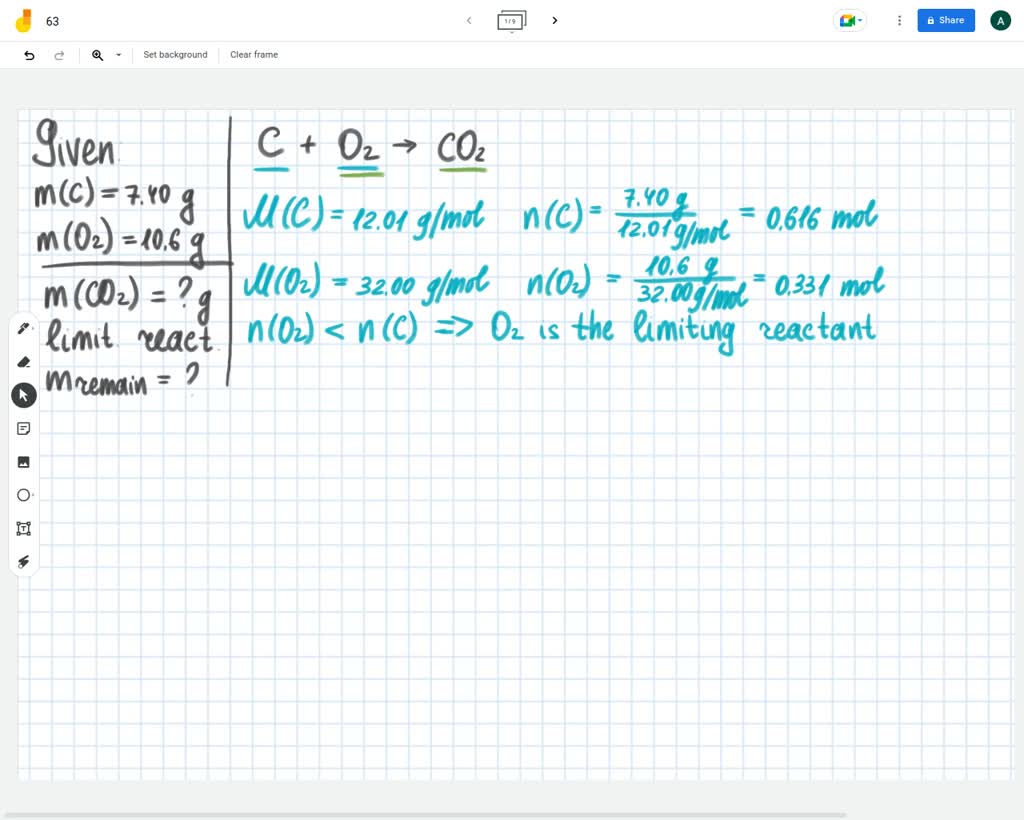
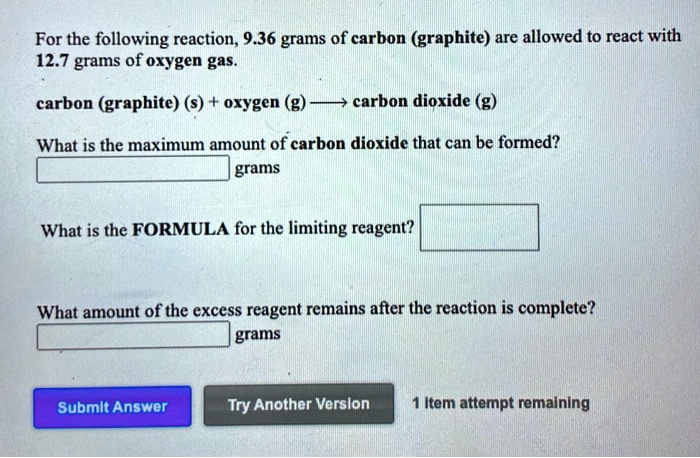
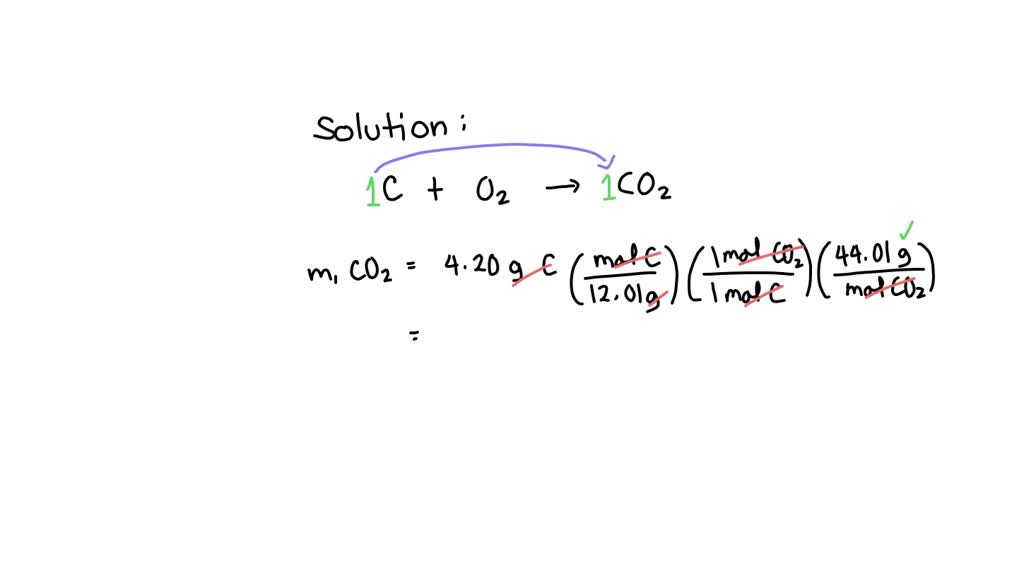
Carbon Graphite S Oxygen G Carbon Dioxide G
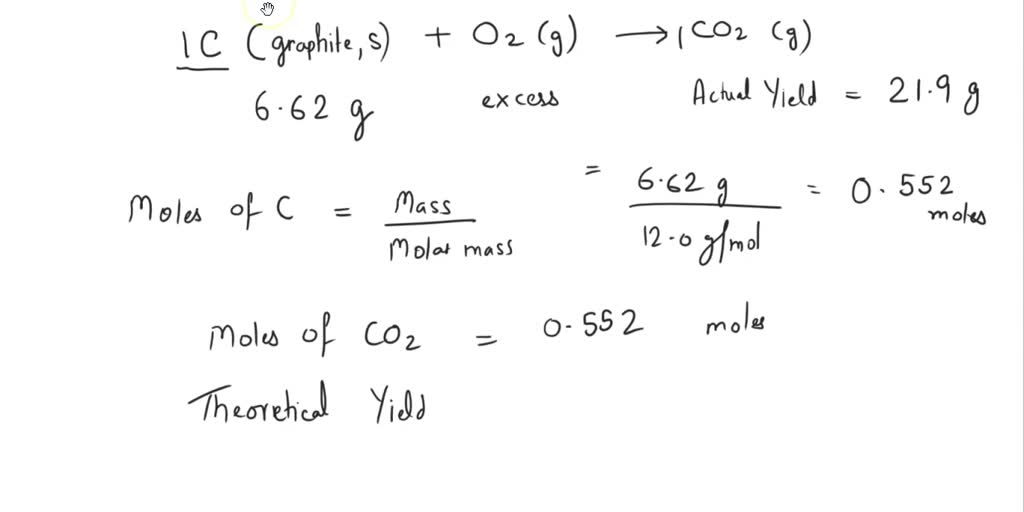
Hey there! Ever get that feeling like you're missing out on something really cool? Well, get ready to have your mind blown, because we're about to dive into a world that's both incredibly simple and surprisingly mind-bending. It’s all about a few familiar elements, but put together in a way that’s, dare I say, entertaining.
Imagine you've got Carbon, that super versatile element that makes up everything from your pencil lead to diamonds. Then you sprinkle in some Oxygen, the stuff we breathe every single second. Sounds pretty normal, right? But when these two get together, things get a whole lot more interesting.
We're talking about a chemical dance, a fascinating reaction that’s happening all around us, and inside us too! It’s like a tiny, invisible theater playing out constantly. And the star of the show? It’s a gas called Carbon Dioxide. Yep, you know that stuff plants love to munch on? That’s our guy.
Must Read
Now, you might be thinking, "A gas? What's so entertaining about a gas?" Ah, that’s where the magic lies! It’s not just about the gas itself, but the journey it takes. Think of Carbon Graphite, a fancy name for the stuff in your pencil, as a sleepy little guy. It’s pretty stable and happy just chilling.
But then comes Oxygen, our energetic friend. Oxygen is like the life of the party, always ready to mix and mingle. When Carbon Graphite and Oxygen meet, especially with a little nudge from some heat or a spark, they decide to team up. It’s a bit like a chemical handshake that results in something new.
And BAM! Out pops Carbon Dioxide, or CO2 as the cool kids call it. It’s this whole transformation that’s just utterly captivating. It’s like watching a caterpillar turn into a butterfly, but on a microscopic level. You start with one thing, and with a little bit of interaction, you get something entirely different and with its own special properties.
What makes it so special? Well, Carbon Dioxide is everywhere! It’s in the air we exhale after taking a big breath. So, in a way, you’re a part of this whole performance every single day. You’re breathing in oxygen, and when your body uses it, it’s like a mini-chemical factory churning out carbon dioxide.
Then that CO2 is released back into the atmosphere. It's a cycle, a continuous loop that keeps our planet going. Plants then take that CO2 and use it to grow, releasing oxygen back into the air for us. It’s a perfect partnership, a beautiful give-and-take. Isn’t that neat?
Think about it: the very air you’re breathing right now contains a bit of this magical gas. It’s not just some abstract chemical concept; it’s a tangible part of our existence. The next time you exhale, give a little nod to the Carbon Dioxide you’re sending out into the world. It’s got a job to do, after all!
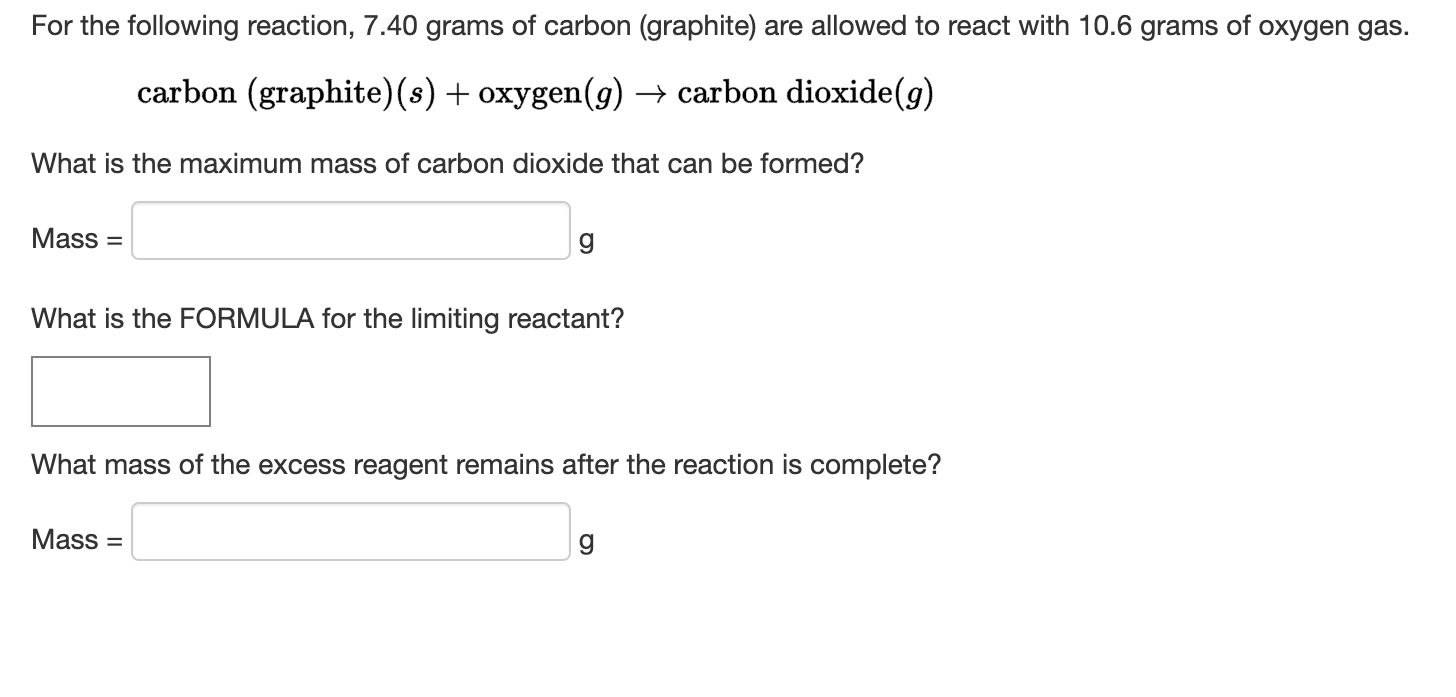
And the way Carbon Graphite becomes Carbon Dioxide is a prime example of chemical reactions. These reactions are the fundamental building blocks of the universe. They’re what make everything happen, from the formation of stars to the growth of a tiny seed.
The sheer variety of ways Carbon can combine with Oxygen is also a part of the fun. While Carbon Dioxide is the most common outcome we’re chatting about, there are other possibilities too. It’s like a recipe with different outcomes depending on the ingredients and how you mix them.
For instance, sometimes, when there’s plenty of oxygen, you can get Carbon Monoxide. This one’s a bit more dramatic, and not as friendly as its cousin. It’s a reminder that even with the same basic elements, the resulting compounds can have vastly different personalities and effects.
But let’s get back to the star of our show, Carbon Dioxide. Its role in the grand scheme of things is truly awe-inspiring. It’s a greenhouse gas, which means it helps trap heat from the sun, keeping our planet warm enough to live on. Without it, Earth would be a frozen wasteland. Who knew that simple gas could be so important?
Of course, like anything, too much of a good thing can be… well, a bit much. The balance of CO2 in our atmosphere is something scientists are constantly studying. It’s a delicate dance, and when it gets out of sync, it can lead to big changes.
But let's not get bogged down in the complexities just yet. The beauty of Carbon Graphite turning into Carbon Dioxide is in its fundamental nature. It’s a constant reminder of the dynamic world we live in, a world of constant change and transformation.
Imagine a campfire. That flickering flame? That's a prime example of Carbon from the wood reacting with Oxygen in the air to produce Carbon Dioxide, heat, and light. You're literally watching chemistry happen right before your eyes!
The smoke you see is a visual representation of this process. It’s the release of these gases, the evidence of the chemical reaction at play. It’s a beautiful, albeit sometimes smoky, spectacle.
And what about those fizzy drinks you love? That delightful tingle on your tongue? That’s dissolved Carbon Dioxide! When you open the bottle or can, the pressure is released, and the CO2 escapes as bubbles, creating that satisfying fizz. It’s chemistry you can taste!
So, the next time you’re enjoying a soda, remember the journey those bubbles have taken. From Carbon and Oxygen to the bubbly Carbon Dioxide, it’s a little bit of everyday magic.
It’s also fascinating to think about how we harness these elements. We use Carbon Graphite in pencils and batteries. Oxygen is essential for life and used in industries. And Carbon Dioxide, despite its potential downsides in excess, is crucial for plant life and has industrial uses too.
The interaction between Carbon and Oxygen is a cornerstone of so many natural processes. It’s a fundamental relationship that underpins much of what we observe and experience.
Think about rusting. While that’s iron and oxygen, the principle of elements reacting with oxygen to form new compounds is similar. The way Carbon forms Carbon Dioxide is a more common and fundamental example of this basic chemical behavior.
It’s this constant interplay, this unending cycle of reactions, that makes the world so alive. And at the heart of it, you have simple elements like Carbon and Oxygen, doing their thing and creating wonders like Carbon Dioxide.
The sheer elegance of it all is what’s so captivating. You don’t need a fancy lab coat or a degree in advanced physics to appreciate this. It’s happening in your kitchen, in the air you breathe, and even in the way your car engine works (though that's a bit more complex!).
The concept of Carbon Graphite, a solid form of carbon, transforming into a gaseous form like Carbon Dioxide is a testament to the power of chemical change. It’s a transformation that’s both fundamental and profoundly impactful.
It’s the basis of combustion, respiration, and photosynthesis. These are not just scientific terms; they are the engines of life and activity on our planet.
So, what makes it so entertaining? It’s the universality, the accessibility, and the sheer wonder of it all. It’s the fact that something so fundamental can be so fascinating. It’s a reminder that the world around us is a constant, ongoing chemical ballet.
The next time you see a flame, or take a sip of a fizzy drink, or even just take a breath, think about the incredible journey of Carbon and Oxygen. Think about the birth of Carbon Dioxide. It's a story that’s been playing out for billions of years, and it’s still going strong.
It’s a story that’s easy to get hooked on, a story that makes you look at the world with a little more curiosity and a lot more awe. So, go ahead, be curious! You might be surprised at what you discover.
The world is a chemical reaction, and we are all part of the show!
The dance between Carbon and Oxygen is a never-ending performance. It’s a constant reminder of how interconnected everything is. From the smallest atom to the largest ecosystem, these simple elements are at play.
And the star of our little drama, Carbon Dioxide, is a crucial character in this grand play. Its presence, its interactions, and its cycles are what keep our planet vibrant and alive.
So, keep your eyes open, your mind curious, and your curiosity piqued. There’s a whole universe of fascinating chemical interactions waiting to be explored, starting with the simple, yet extraordinary, story of Carbon Graphite, Oxygen, and Carbon Dioxide.
It’s a story that’s both educational and utterly delightful. You might even find yourself wanting to learn more. And that, my friends, is the best kind of entertainment!