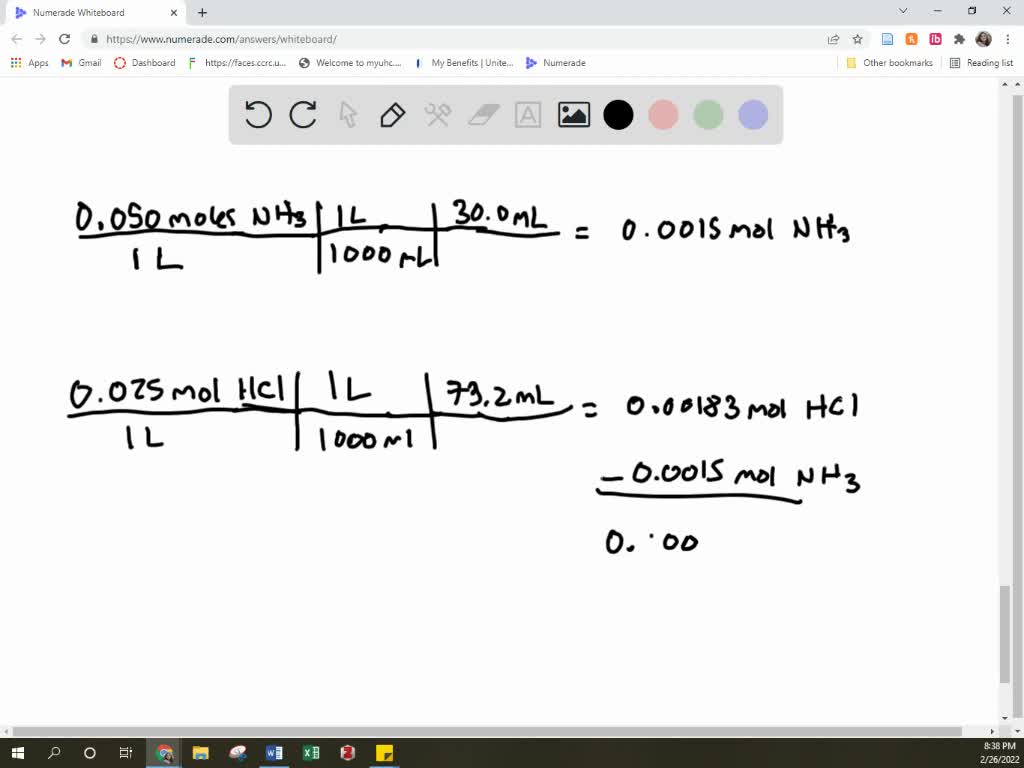
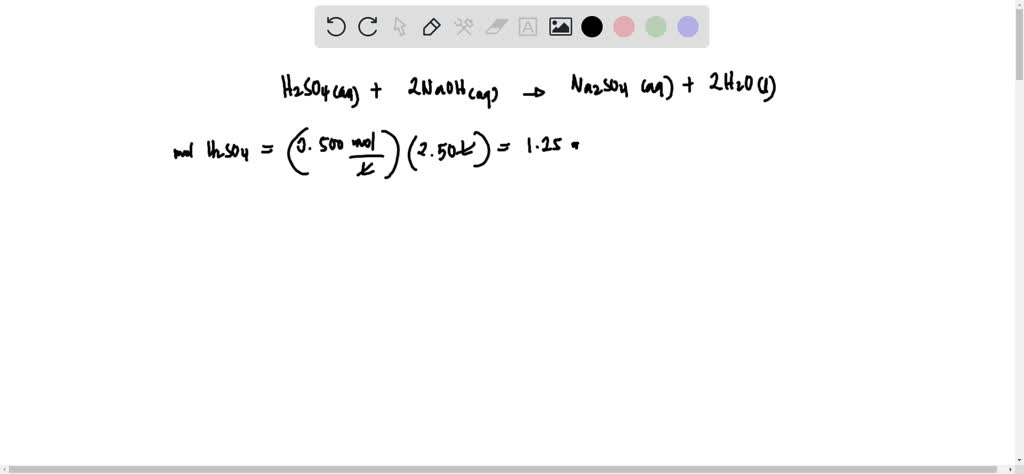
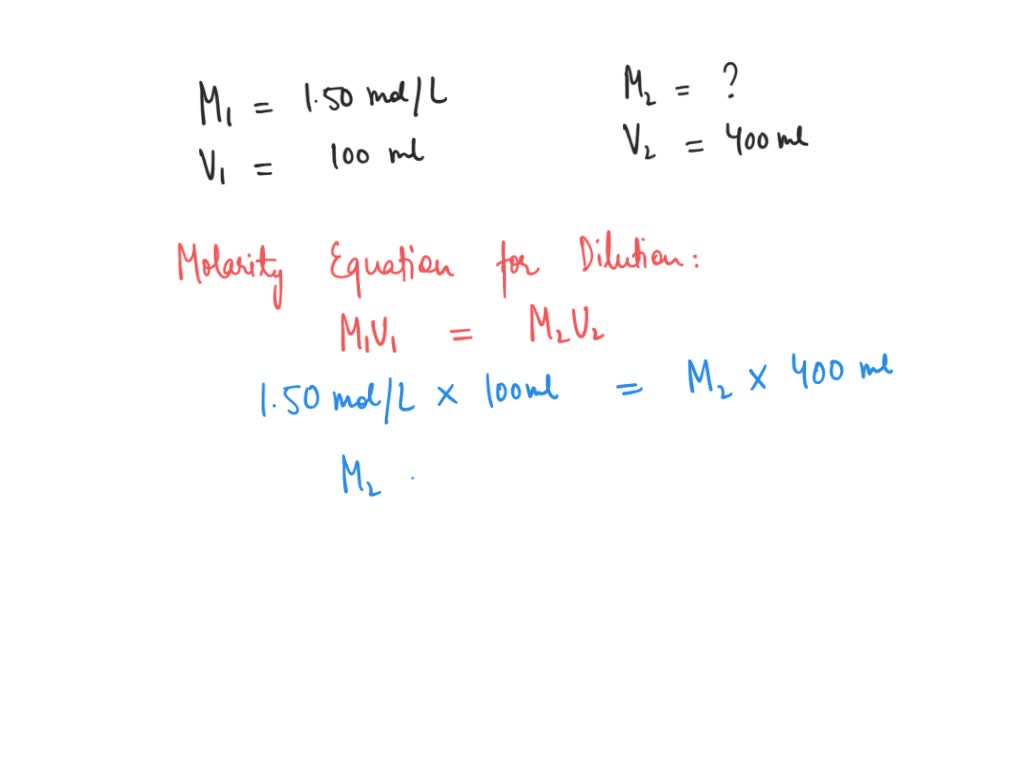Calculate The Volume In Ml Of 0.100 M Cacl2

Hey there, science curious folks! Ever find yourself staring at a bottle of something and wondering, "How much of this stuff is really in here?" It's a question that pops up more often than you might think, especially when you're dealing with those little scientific ingredients that make our lives a whole lot better.
Today, we're going to peek behind the curtain of a common little chemical wonder: calcium chloride, or as the cool kids call it, CaCl₂. Now, calcium chloride might not be as glamorous as a SpaceX rocket launch or as delicious as grandma's apple pie, but oh boy, does it play a starring role in some pretty neat things!
The Sweet, Salty Secrets of CaCl₂
Think about those chilly winter mornings when the roads are like a slippery ice rink. Who's the hero that melts away all that danger? Yep, it's often calcium chloride! It's like a super-powered salt that gets the job done, making our commutes safer and preventing those embarrassing tumbles. Imagine a world without it – we'd all be ice-skating to work, and not in a fun, figure-skating kind of way.
Must Read
But the magic of CaCl₂ doesn't stop when the snow melts. Have you ever seen those signs at swimming pools that warn about 'hard water'? Well, calcium chloride is actually a key player in what makes water 'hard' – and sometimes, that's a good thing! It's all about the minerals, you see. It’s like the tiny, invisible architects that help build strong bones and keep our bodies humming along. So, next time you’re enjoying a refreshing dip, give a little nod to CaCl₂ for its quiet contribution.
And get this, it’s also a secret ingredient in some of your favorite snacks! It helps firm up canned tomatoes, giving them that perfect bite. It’s also used in making cheese and even in some brewing processes. So, while you’re munching on your chips or savoring a delicious meal, remember the humble calcium chloride, working its wonders behind the scenes. It’s like the invisible elf in the kitchen, making sure everything tastes just right.
Now, sometimes in the lab, scientists need to be super precise. They're not just throwing ingredients around willy-nilly. They're measuring, mixing, and calculating with the kind of focus that would make a brain surgeon proud. And when they're working with something like calcium chloride, they often talk about its "concentration."
The Mystical M: More Than Just a Letter
You might have heard the term "M" thrown around. In the science world, that little "M" stands for something called molarity. Don't let the big word scare you! Think of it like this: if you're making a big batch of lemonade, molarity is just a fancy way of saying how much lemon juice you've squeezed into a certain amount of water. It tells you how concentrated your lemonade is. The stronger it is, the more 'zing' you get, right?
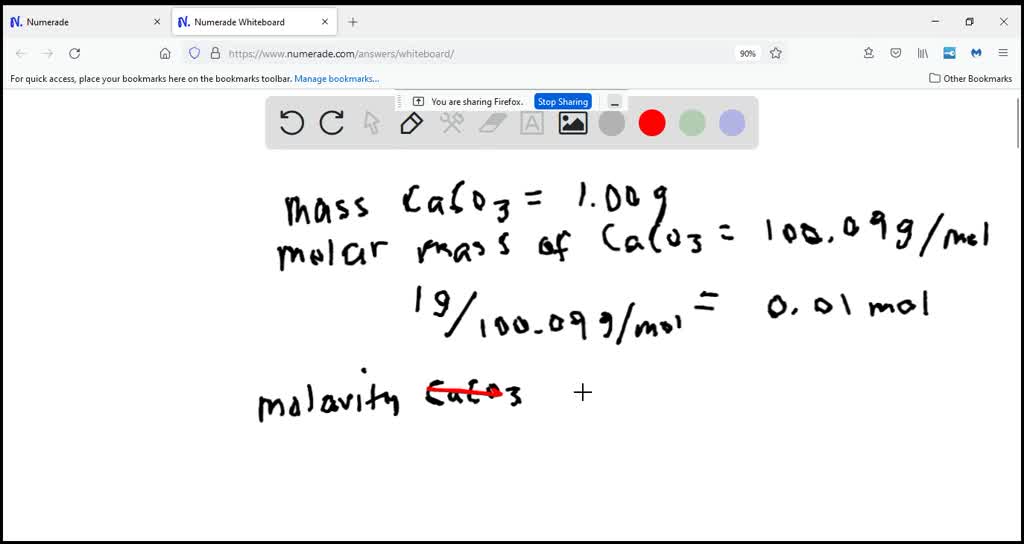
So, when you see "0.100 M CaCl₂," it's basically telling us that for every liter of water, there are 0.100 "units" of calcium chloride dissolved in it. It's a precise recipe, ensuring that the chemical reaction or the effect they're going for is just right. It's like following a carefully crafted baking recipe – too much or too little of an ingredient can change the whole outcome!
Now, here’s where it gets a little bit like a treasure hunt. We’re asked to find the volume in milliliters (mL) of this 0.100 M CaCl₂. It sounds like a riddle, doesn't it? But in the world of science, it's more like a puzzle where all the pieces fit together beautifully.
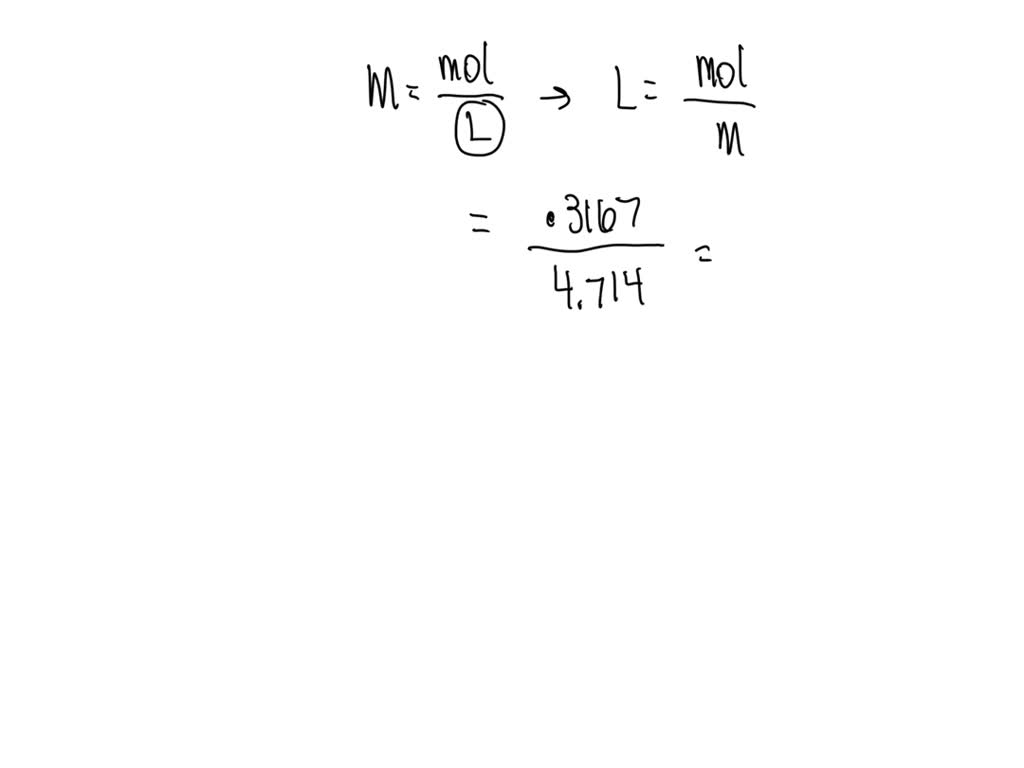
Imagine you have a recipe for, say, a magical potion. This potion needs a specific amount of a special ingredient, and the recipe tells you how concentrated that ingredient needs to be. But it doesn't tell you how much potion you're making in total. That's where our little calculation comes in!
Think about it this way: if you have a really strong cup of coffee (high molarity!), you might only need a small sip to get your energy boost. But if you have a weaker cup (lower molarity!), you might need a whole mug. The concentration changes how much liquid you need to get the same "kick."

Our 0.100 M CaCl₂ is like that. It's a specific concentration. The question isn't asking us to make more of it, or to magically conjure it out of thin air. It's asking, if we have a certain amount of this 0.100 M CaCl₂ solution, what volume does it occupy? It’s like asking, "If I have a gallon of 10% salt water, how much space does that gallon take up?" The answer is, well, a gallon!
The beauty of it is that the question itself is a bit of a playful trick. When you're given a concentration, like 0.100 M, it's usually in relation to a volume of solution. So, if you're working with this specific solution, you're probably already holding a certain volume of it. The calculation is less about discovering a hidden quantity and more about understanding the relationship between concentration and the amount of stuff dissolved in a liquid. It’s a reminder that in science, sometimes the simplest answers are hidden in plain sight, just waiting for us to appreciate them!
So, the next time you encounter a concentration like 0.100 M CaCl₂, don't be intimidated. Think of it as the recipe for a useful ingredient, and remember all the fun and important jobs it does in our everyday lives. It’s a little bit of chemistry magic, making the world around us a little bit safer, a little bit tastier, and a whole lot more interesting!