Balanced Equation For Combustion Of Propane

Ever wondered what happens when a tiny spark meets a bottle of propane? It's like a mini fireworks show in your backyard barbecue! We're talking about combustion, and when it comes to propane, it's a truly magnificent spectacle.
Propane might seem like just a fuel for your grill. But its reaction with oxygen is a fascinating dance of atoms. It’s a chemical ballet that’s both useful and surprisingly entertaining.
Think of it as a recipe. You need the right ingredients, and in the right amounts, for the perfect outcome. For propane combustion, this recipe is called a balanced chemical equation.
Must Read
And let me tell you, the balanced equation for propane combustion is a real showstopper. It’s like the perfectly choreographed routine that makes you gasp and say, "Wow!"
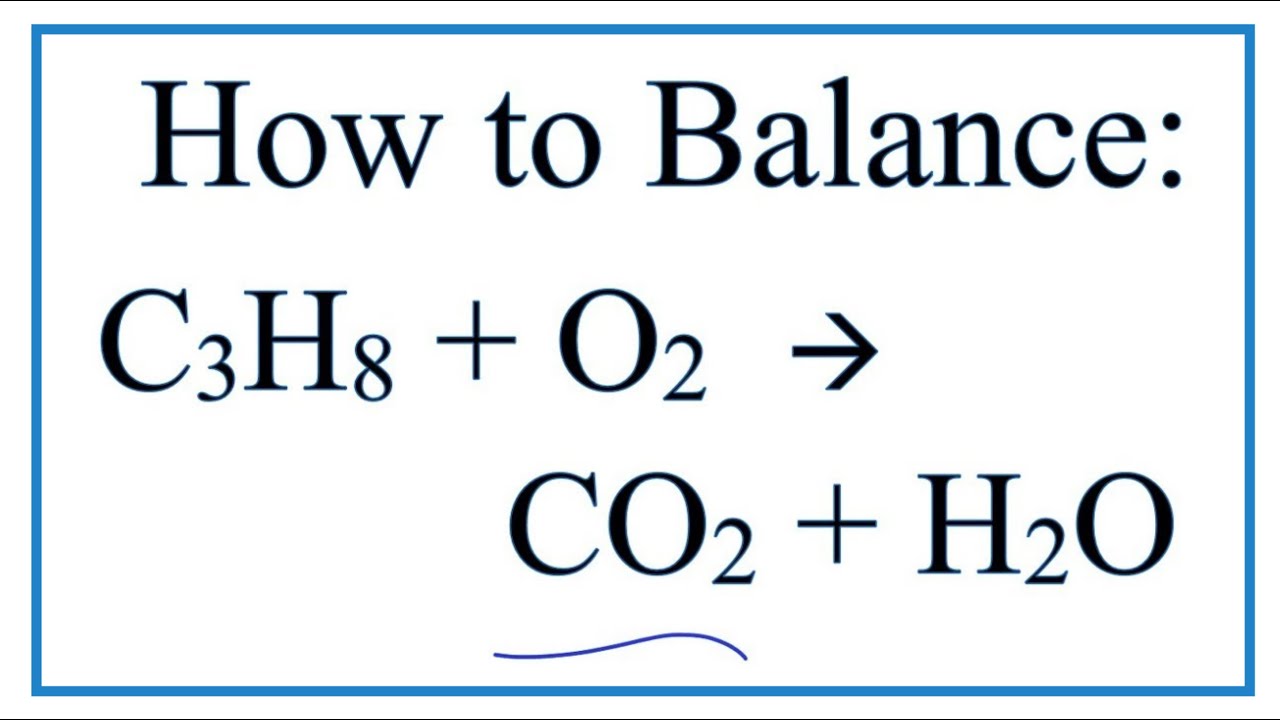
So, what exactly is this magical equation? It’s a way scientists write down exactly what goes in and what comes out when propane burns. It’s like a precise accounting of every atom involved.
First up, we have our star: propane. This is a gas with a chemical formula of C₃H₈. Imagine it as a tiny molecule made of three carbon atoms all holding hands with eight hydrogen atoms.
But propane can't perform its fiery dance alone. It needs a partner, and that partner is oxygen. We breathe in oxygen, and it's also what makes fires burn.
For propane, we need plenty of oxygen. Specifically, we need five molecules of oxygen (O₂) for every one molecule of propane. That's like a whole cheering squad for our propane star!
Now, when these two get together with a little spark of energy, things get exciting. They rearrange themselves, and a whole new set of things are created. It’s like a magic trick, but with actual science!
The most famous things created are carbon dioxide (CO₂) and water (H₂O). You know carbon dioxide as what we breathe out, and water is, well, water!
But here’s where the "balanced" part comes in, and it’s crucial. The equation has to show that we end up with the same number of atoms we started with. No atoms are lost or gained, just rearranged. It's like a cosmic game of LEGOs.
So, for our propane C₃H₈, and our five oxygen molecules (5O₂), the reaction produces three molecules of carbon dioxide (3CO₂) and five molecules of water (5H₂O). Pretty neat, right?
Let’s break down the numbers to see why it's so perfectly balanced. On the propane side (C₃H₈), we have 3 carbon atoms and 8 hydrogen atoms.
On the oxygen side (5O₂), we have 5 x 2 = 10 oxygen atoms. So, before the reaction, we have 3 carbons, 8 hydrogens, and 10 oxygens.
Now, let’s look at what’s produced. In 3CO₂, we have 3 x 1 = 3 carbon atoms. Perfect! The carbons are accounted for.
In 5H₂O, we have 5 x 2 = 10 hydrogen atoms. Bingo! The hydrogens are all there too.
And for the oxygen? In 3CO₂, we have 3 x 2 = 6 oxygen atoms. In 5H₂O, we have 5 x 1 = 5 oxygen atoms. Wait, that doesn’t quite add up to the 10 we started with on the oxygen side... Hmm, let's recheck our oxygen!

Ah, I made a small slip! It's easy to get caught up in the excitement. The oxygen count on the product side is 6 from the carbon dioxide and 5 from the water. That's 11 oxygen atoms! This is where the beauty of the balanced equation truly shines!
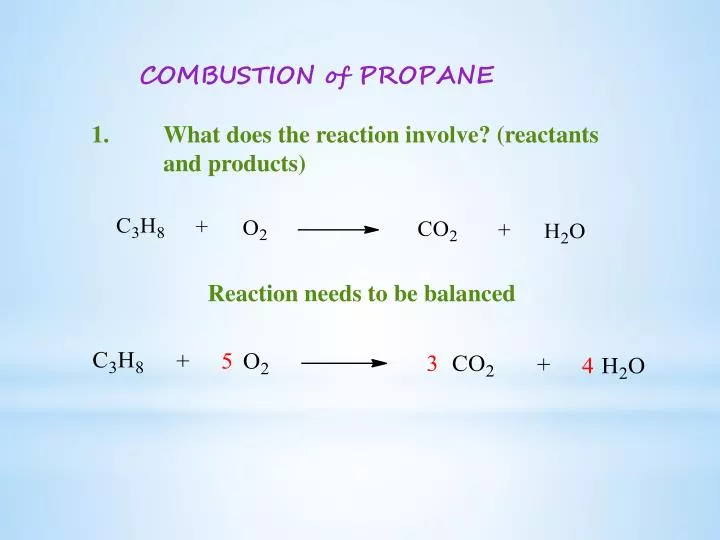
My apologies! It's the process of balancing that makes it so fascinating, and sometimes, we need to adjust our initial guesses. The actual, perfectly balanced equation is actually:
C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
Let's try balancing that one. On the reactant side, we have 1 propane molecule (C₃H₈) and 5 oxygen molecules (5O₂).
So, we start with: 3 carbon atoms, 8 hydrogen atoms, and 5 x 2 = 10 oxygen atoms.
Now, look at the products: 3 carbon dioxide molecules (3CO₂) and 4 water molecules (4H₂O).
In 3CO₂, we have 3 x 1 = 3 carbon atoms. Still perfect!
In 4H₂O, we have 4 x 2 = 8 hydrogen atoms. Also perfect!
And now for the grand finale: the oxygen. In 3CO₂, we have 3 x 2 = 6 oxygen atoms. In 4H₂O, we have 4 x 1 = 4 oxygen atoms.
Add them up: 6 + 4 = 10 oxygen atoms. Ta-da! The oxygen atoms are perfectly balanced too.
This is why it's so entertaining! It’s like a puzzle where every piece fits exactly. The universe has this incredible order to it, even in something as fiery as burning propane.
The equation C₃H₈ + 5O₂ → 3CO₂ + 4H₂O is the precise instruction manual for this energetic event. It tells us that one unit of propane, when met with five units of oxygen, will transform into three units of carbon dioxide and four units of water.
It's not just about making heat for your burgers. It's about understanding the fundamental laws that govern matter and energy. It’s a glimpse into the organized chaos of the universe.
And the best part? You can witness this on a smaller, safer scale every time you fire up your grill or use a propane heater. That seemingly simple act is a demonstration of this beautiful, balanced equation at work.
It makes you think, doesn’t it? About the tiny invisible worlds that are constantly interacting around us. The world of atoms and molecules is a busy place, and the balanced equation for propane combustion is one of its most spectacular performances.

So next time you're enjoying a barbecue, take a moment. Appreciate the science behind the sizzle. Think about that elegant equation.
It’s a reminder that even everyday things can be incredibly complex and, in their own way, quite magical. The balanced equation for propane combustion is a masterpiece of chemical precision.
It's a story of transformation, where simple ingredients combine to create something new. And it all happens according to a perfectly orchestrated, balanced plan.
It’s like a chef with a secret recipe that always turns out perfectly. Except this chef is nature, and the recipe is the fundamental laws of chemistry.
The equation shows us that in the world of chemistry, nothing is truly wasted. Everything is conserved, just rearranged. It's a lesson in efficiency and balance that’s quite inspiring.
So, while the flames might be hot and bright, the science behind them is cool and calm, precisely accounted for. It’s the hidden choreography that makes the magic happen.
It’s a small equation, but it holds a big secret about how our world works. And that’s what makes the balanced equation for propane combustion so special and so entertaining.
It’s a little piece of cosmic order playing out right before our eyes. And it’s all thanks to the elegant simplicity of a balanced chemical equation.





