Balanced Equation For Combustion Of Hexane

Imagine you're at a cozy campfire, the flames dancing merrily, warming your face. Or perhaps you're enjoying a delicious barbecue, the smell of grilled food filling the air. These moments, filled with warmth and deliciousness, all rely on a fascinating chemical dance happening right before our eyes, even if we don't see the tiny steps. Today, we're going to peek behind the curtain of one of these everyday miracles: the combustion of hexane.
Now, hexane might sound like a complicated science word, and in a way, it is. But think of it as a special kind of fuel, like the gasoline in your car or the propane in your grill. It's made of tiny building blocks, mostly carbon and hydrogen atoms, all holding hands in a specific arrangement. This arrangement is what gives it its energy-holding power.
When we talk about combustion, we're essentially talking about a very enthusiastic party where hexane gets to meet its best friend: oxygen. You know, the stuff we breathe in all the time? This meeting is not just a polite handshake; it's a full-on celebration!
Must Read
This party needs a little spark to get started, like striking a match. Once that spark happens, the hexane and oxygen get super excited and start rearranging themselves. It's like they're playing a chemical game of musical chairs!
The main goal of this energetic party is to release all the stored-up energy that was holding the hexane molecule together. This energy is what gives us heat and light, making our campfires glow and our grills sizzle. It's a beautiful transformation, turning something that was just sitting there into something dynamic and useful.
So, what happens to the little atoms after all that excitement? Well, they tidy up and form new molecules. The carbon atoms from the hexane decide they really like hanging out with oxygen, and they form carbon dioxide. Yep, that's the same stuff we exhale, which plants love to use for their own food-making adventures.
And the hydrogen atoms? They also find a new buddy in oxygen. Together, they form water. Isn't that neat? The same stuff that quenches our thirst and makes rain is also a byproduct of burning fuel!

Now, let's talk about the "balanced equation" part, which sounds very official but is actually quite logical. Think of it like baking a cake. You need precise amounts of flour, sugar, and eggs to get the perfect cake. If you have too much or too little of something, the cake might not turn out quite right.
In the world of chemistry, the balanced equation ensures that all the atoms that go into the reaction are accounted for in the products. It's like making sure no atoms get lost in the shuffle during the fiery party. Everything that starts out must end up somewhere!
For hexane, its chemical formula is C6H14. This tells us it has 6 carbon atoms and 14 hydrogen atoms. Oxygen, in its natural form, is O2, meaning it comes in pairs of oxygen atoms.
When hexane burns, it reacts with oxygen to produce carbon dioxide (CO2) and water (H2O). The equation looks like this: C6H14 + O2 → CO2 + H2O.
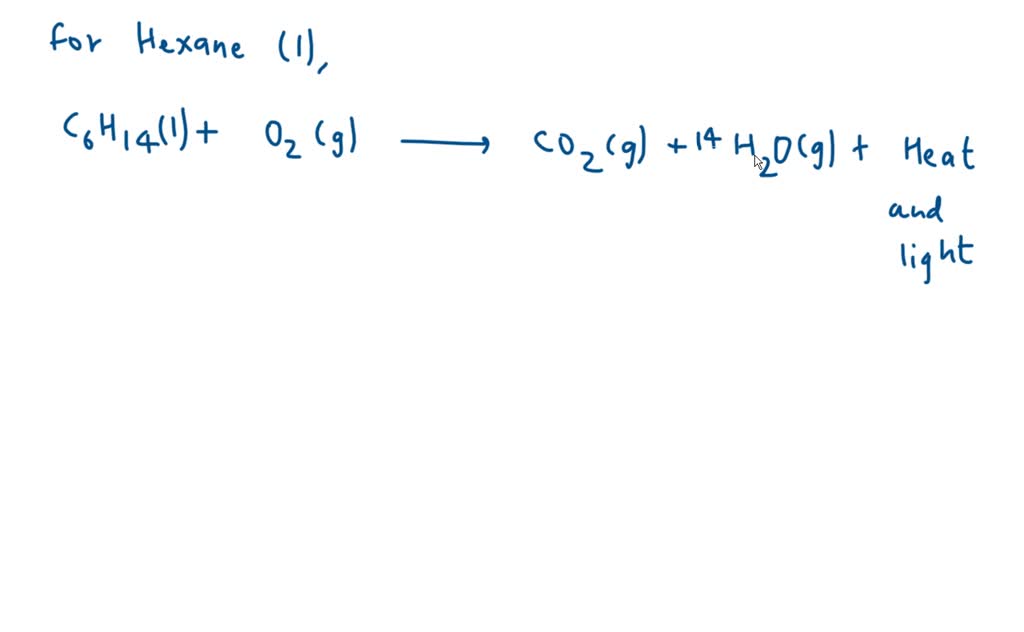
But this equation is like a recipe with ingredients scattered everywhere. We need to arrange it so that the number of atoms of each element on the "before" side (the reactants) is exactly the same as on the "after" side (the products). This is where the balancing act comes in.
Let's start with the carbon. We have 6 carbons in hexane. So, we need 6 molecules of carbon dioxide on the other side to match them. So, our equation starts looking like: C6H14 + O2 → 6CO2 + H2O.
Now, let's look at the hydrogen. We have 14 hydrogens in hexane. Since each water molecule has 2 hydrogens, we need 7 molecules of water to have 14 hydrogens. Our equation evolves to: C6H14 + O2 → 6CO2 + 7H2O.
The tricky part often comes with oxygen. On the product side (the "after" side), we have 6 * 2 = 12 oxygen atoms from the carbon dioxide, and 7 * 1 = 7 oxygen atoms from the water. That's a total of 12 + 7 = 19 oxygen atoms!

But remember, oxygen comes in pairs (O2). So, to get 19 oxygen atoms, we need 19/2, or 9.5, molecules of oxygen. So the equation would be: C6H14 + 9.5O2 → 6CO2 + 7H2O.
Now, chemists don't usually like using half molecules in their balanced equations. It's like having half a Lego brick – it can be a bit awkward. So, to make everything whole, we multiply the entire equation by 2.
This means we double everything: 2 molecules of hexane, 19 molecules of oxygen, 12 molecules of carbon dioxide, and 14 molecules of water. The final, beautifully balanced equation is: 2C6H14 + 19O2 → 12CO2 + 14H2O.
This equation is like a perfect choreography for the atoms. It tells us that for every 2 molecules of hexane that decide to have their energetic party, they need 19 molecules of oxygen to join in. And when the dust settles, they'll have produced 12 molecules of carbon dioxide and 14 molecules of water.

It's a subtle reminder that even in the simplest of joys, like a crackling fire or a perfectly grilled steak, there's a complex and elegant dance of atoms happening. And the balanced equation is simply the conductor’s score, ensuring every performer plays their part perfectly. It's a little bit of magic, neatly written down.
So, the next time you're enjoying the warmth of a flame or the aroma of a barbecue, you can smile knowing about the incredible, precisely balanced party that hexane and oxygen are having. It’s a testament to the order and beauty that exists, even in the fiery heart of everyday life.
It's a story of transformation, of energy released, and of new beginnings for the atoms involved. From the humble fuel to the gases that fill our air and the water that sustains life, it's a complete cycle of sorts. And all thanks to a little bit of hexane, a lot of oxygen, and a well-choreographed chemical equation.
The beauty of it lies in its universality. This same principle of balancing atoms applies whether it's a tiny laboratory experiment or a massive industrial process. It’s a fundamental law of nature, ensuring that nothing is truly lost, just rearranged into something new and wonderful.
So, let's raise a metaphorical glass to hexane and its fiery, balanced dance! It’s a performance that fuels our world in more ways than we often realize, bringing light, warmth, and deliciousness into our lives, one balanced equation at a time. It's a heartwarming thought, isn't it?





