An Ideal Gas Originally At 0.85 Atm And 66

So, there I was, wrestling with a particularly stubborn pickle jar. You know the kind, the one that's been sealed tighter than a politician's promise. I tried everything – running it under hot water, whacking it on the counter (don't judge, we've all been there!), even a strategic use of a rubber glove for extra grip. Nothing. It was like the lid had become one with the glass, a monument to preserved cucumbers and my own culinary aspirations.
Then, a little voice in the back of my head – probably the same one that whispers "just one more episode" at 2 AM – said, "What if you heated it up?" Brilliant, right? My initial thought was to just stick the whole jar in the oven. But then I remembered my trusty science teacher, Mrs. Gable, with her perpetually stained lab coat and a fondness for exploding water balloons. She'd probably have a conniption. So, instead, I opted for the hot water bath approach, just like I do for sensitive chocolate.
As the jar warmed, I could almost feel the pressure inside building. It was a subtle thing, not a dramatic pop, but a distinct give. And voilà! The lid twisted open with an ease that was frankly embarrassing. I’d spent ten minutes of my life fighting a jar that just needed a little warmth. It got me thinking, you know, about how much of our everyday lives are governed by invisible forces, by the way things behave when they’re just… existing. And that, my friends, is where our slightly grumpy, initially reluctant, ideal gas comes in.
Must Read
Imagine, if you will, a balloon. Not a fancy, helium-filled party balloon, but a plain, old, rubbery one. Now, let’s say this balloon is filled with air, and that air is our ideal gas. We’re going to put this balloon in a bit of a situation, a situation where it starts off at a pressure of 0.85 atmospheres. Think of an atmosphere as, roughly, the air pressure we feel right now, at sea level. So, it's a bit less than our usual everyday pressure. And it’s also chilling at 66 degrees Celsius. Now, 66 Celsius might sound like it’s boiling, but remember, we’re talking about gas molecules here. For them, it’s more like a slightly warm day, not exactly a sauna. For us, it’s… well, let’s just say it’s warm. Like, "should I have worn shorts?" warm.
Our ideal gas, in this state, is doing its thing. The molecules are zipping around, bumping into each other, colliding with the walls of our balloon. This constant jostling and bouncing is what creates the pressure. It’s like a tiny, energetic dance party happening inside. The temperature, on the other hand, is a measure of how much energy these molecules have, how fast they’re dancing. Higher temperature, faster dance, more energetic collisions.
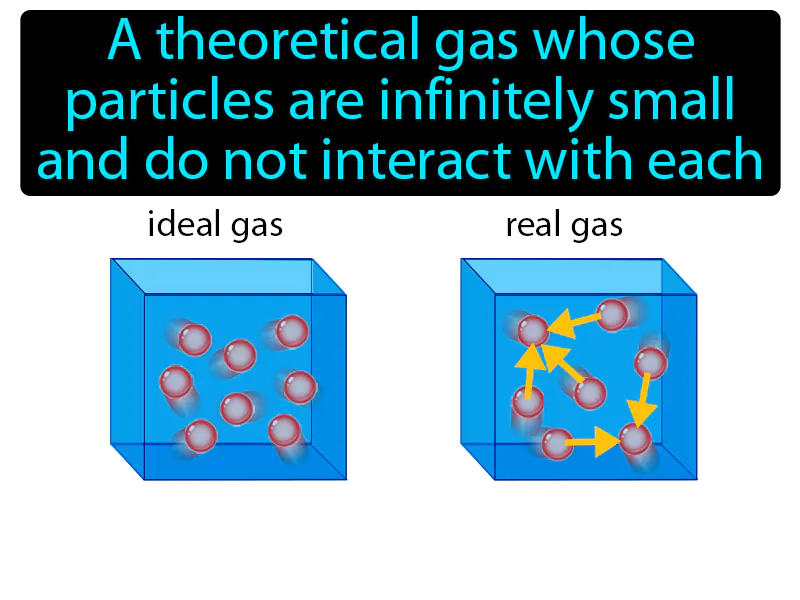
Now, the term "ideal gas" is a bit of a scientist's dream. It’s a theoretical concept, a simplification that helps us understand the real world better. In reality, gas molecules have volume and they do attract or repel each other a little. But for many situations, especially at moderate pressures and temperatures, our ideal gas model is remarkably accurate. It’s like using a blueprint instead of a full 3D scan – you get the essential structure, the key relationships, without getting bogged down in every single tiny detail. So, when we talk about an ideal gas, we're assuming its molecules are point masses (meaning they have no volume themselves) and they don't interact with each other except during perfectly elastic collisions. Think of them as tiny, perfectly bouncy billiard balls.
The first bit of information we have is the initial pressure, P1, which is 0.85 atm. And the initial temperature, T1, is 66 degrees Celsius. Now, here’s a little trick that scientists (and anyone trying to sound smart at a party) love to do. We often need to convert temperatures to the Kelvin scale for gas law calculations. Why? Because Celsius and Fahrenheit have arbitrary zero points (freezing and boiling of water, respectively). Kelvin, on the other hand, has an absolute zero, where theoretically all molecular motion stops. It’s a much more fundamental scale for talking about energy. To convert from Celsius to Kelvin, you simply add 273.15. So, our 66 degrees Celsius becomes 66 + 273.15 = 339.15 K. See? Not so scary. Just a different way of counting the heat. It’s like switching from counting your money in dollars to euros – it’s still value, just a different unit.
So, our ideal gas is initially at P1 = 0.85 atm and T1 = 339.15 K. Now, what are we going to do with this gas? The real fun of these problems comes when we change one of the conditions and see how the others react. It’s all about relationships. Like a cosmic game of cause and effect, but with molecules instead of dominoes.
Let’s imagine we decide to heat this balloon up. We’re going to crank up the temperature. What do you think will happen to the pressure? If you said it will increase, you’re already thinking like a gas physicist! Remember our dance party analogy? More heat means the molecules dance faster, they bounce around with more gusto. They’re going to hit the walls of the balloon with more force, and more often. This increased impact on the balloon's inner surface is what we perceive as a rise in pressure. It’s a direct consequence of giving those molecules a bigger energy boost.
Or, what if we decide to squeeze the balloon? We’re applying external pressure to make it smaller. This means the gas molecules inside are now confined to a smaller space. They’re still dancing at the same speed (assuming the temperature stays the same), but now they have less room to roam. This leads to more frequent collisions with the walls, and hence, an increase in pressure. The molecules are feeling a bit crowded, a bit like you on a rush-hour subway. They’re bumping into each other and the walls more often!
The beauty of dealing with ideal gases is that their behavior is remarkably predictable thanks to a set of laws that describe these relationships. We have Boyle's Law, Charles's Law, Gay-Lussac's Law, and the grandaddy of them all, the Ideal Gas Law. These aren't just arbitrary rules; they’re derived from the fundamental kinetic theory of gases, which explains the macroscopic properties of gases in terms of the microscopic motion of their molecules. It’s a beautiful piece of scientific logic.
Boyle's Law, for instance, tells us that if you keep the temperature constant, the pressure of a gas is inversely proportional to its volume. So, if you double the pressure, you halve the volume, and vice versa. Imagine squeezing that balloon – as you reduce the volume, the pressure inside goes up. Simple, right?
Charles's Law, on the other hand, deals with the relationship between volume and temperature when the pressure is held constant. It says that the volume of a gas is directly proportional to its absolute temperature. So, if you heat up that balloon and let it expand freely (keeping the pressure the same), it will get bigger. Think of a hot air balloon. You heat the air inside, it expands, and makes the balloon rise. It’s all about those molecules getting more energetic and needing more space.
Gay-Lussac's Law is the one that really ties into our pickle jar story! It states that for a fixed amount of gas at constant volume, the pressure is directly proportional to its absolute temperature. So, if you heat up a sealed, rigid container (like our pickle jar, if it were perfectly sealed and didn’t shatter), the pressure inside will increase. This is exactly what happened with my stubborn pickle jar! The heat caused the air molecules inside to move faster, leading to more frequent and forceful collisions with the lid, increasing the pressure until it was enough to overcome the seal.
And then we have the Universal Ideal Gas Law, which brings it all together: PV = nRT. This is the Einstein of gas laws, the all-encompassing equation. P is pressure, V is volume, n is the number of moles of gas (a measure of how much gas there is), R is the ideal gas constant (a universal number that makes the equation work), and T is the absolute temperature. This equation tells us that pressure, volume, and temperature are all intertwined. Change one, and at least one of the others must change, unless you’re incredibly lucky or you’ve stumbled into a very specific, idealized scenario.
Now, back to our balloon, with its initial state of P1 = 0.85 atm and T1 = 339.15 K. Let’s say we decide to heat it up so the temperature reaches 150 degrees Celsius. First step, convert to Kelvin: 150 + 273.15 = 423.15 K. So, T2 = 423.15 K.
The crucial question is: what happens to the pressure? We need to make an assumption here. If we assume the balloon can expand freely and the pressure remains constant at 0.85 atm, then we’re looking at a case of Charles's Law. The volume would increase. But if the balloon is constrained in some way, or we’re interested in the pressure change if the volume were to stay the same (which is less likely for a balloon, but good for illustrating Gay-Lussac's Law), then the pressure would increase. The problem statement, "An Ideal Gas Originally At 0.85 Atm And 66," doesn't specify what happens to the volume or if the amount of gas changes. This is where things get interesting and, frankly, a little open-ended for a simple blog post! It’s like being given ingredients but no recipe.
However, if we consider a scenario where the amount of gas (n) remains constant, and the volume (V) is also kept constant (a hypothetical rigid container for our gas, not a flexible balloon), then we can use Gay-Lussac's Law. In this case, the ratio P1/T1 will be equal to P2/T2. So, we have:
P2 = P1 * (T2 / T1)
Plugging in our values:
P2 = 0.85 atm * (423.15 K / 339.15 K)
P2 ≈ 0.85 atm * 1.2476
P2 ≈ 1.06 atm
So, if our ideal gas was in a rigid container, and we heated it from 66°C to 150°C, the pressure would increase from 0.85 atm to about 1.06 atm. The molecules are still dancing in the same confined space, but now they're dancing with a lot more energy, hitting the walls with more force. It's like a mosh pit getting significantly more energetic!
What if, on the other hand, the scenario implied that the pressure remained constant at 0.85 atm, and we wanted to know the new temperature if the volume changed? Then we’d be looking at Charles’s Law. If the volume doubled, for example, the temperature would also have to double in Kelvin.
The real power of these laws is their universality. They apply to a wide range of gases under many conditions. Whether you’re a chemist designing a new reaction, an engineer building an engine, or just someone trying to understand why your car tires are a bit softer on a cold day, these fundamental principles are at play. They’re the silent architects of so many physical phenomena around us.
The initial state of our ideal gas – 0.85 atm and 66°C (339.15 K) – is just a snapshot in time, a starting point for exploring the fascinating world of gas behavior. It’s a reminder that even seemingly simple things like air are governed by intricate, predictable laws. It’s a bit like that pickle jar. I thought I was just dealing with a stubborn lid, but really, I was witnessing a miniature demonstration of pressure and temperature at play, a tiny physics lesson disguised as a snack emergency. And honestly, isn't that the best kind of lesson?
So, the next time you’re out and about, or just lounging at home, take a moment to appreciate the invisible dance of the gas molecules all around you. They’re maintaining our atmosphere, inflating our tires, and making our world go ‘round, all while obeying some pretty elegant scientific laws. And who knows, maybe understanding them a little better will even help you conquer your next stubborn pickle jar. Happy experimenting (safely, of course)!