A Solution Containing Hcl Would Likely Have

Hey there, my science-curious pals! Ever found yourself staring at a bottle labeled "HCl" and wondering, "Okay, what's the deal with this stuff?" Or maybe you’ve heard of hydrochloric acid in a lab class and thought it sounded a bit intimidating. Well, fear not! Today, we’re going to dive into the wonderful world of hydrochloric acid solutions, and I promise, it’s going to be more like a fun chat over coffee than a dry lecture. We’re talking about what you’d likely find hanging out in a solution of HCl. Think of it as a get-to-know-you party for molecules!
So, what exactly is hydrochloric acid? In its purest form, it's a gas called hydrogen chloride (HCl). But when we talk about an HCl solution, we're usually talking about hydrogen chloride gas that's been dissolved in water. And boy, does it dissolve! This stuff is a champion at mixing with water, like a social butterfly at a party. It’s a bit of a drama queen in the best way possible, completely breaking apart into its constituent ions.
When hydrogen chloride (HCl) meets water (H₂O), something really cool happens. It’s not just a polite handshake; it’s more like a full-on embrace where HCl gives up one of its hydrogen atoms to a water molecule. This is called dissociation, and it’s the key to why HCl solutions are so... well, acidic. Think of it like this: HCl is a bit like a generous friend who always has an extra hydrogen to share. Water molecules are happy to take them!
Must Read
So, in our HCl solution, you're not going to find neat little HCl molecules just floating around. Nope! They’ve gone their separate ways, or rather, they’ve transformed. The HCl molecule itself breaks into a positively charged hydrogen ion, which we often write as H⁺. But, as I just mentioned, this H⁺ is a bit too eager and immediately latches onto a water molecule. It’s like the hydrogen ion is a puppy and the water molecule is a comfy lap.
This union creates a hydronium ion. We usually write this as H₃O⁺. So, instead of plain old H⁺ chilling with water, you have this hydronium ion doing the rounds. It’s the real rockstar of an acidic solution! This H₃O⁺ is what gives the solution its characteristic acidic properties. It's the reason why acids can be stingy with electrons and eager to donate them to other substances, which is super important in tons of chemical reactions.

Besides the trusty hydronium ions, you'll also find chloride ions chilling out in the solution. Remember that HCl molecule? It broke into H⁺ and Cl⁻. The H⁺ became part of the H₃O⁺, but the Cl⁻ ion is now a free agent, just a negative charge hanging out with all the positive charges and the neutral water molecules. So, in an HCl solution, the main players you'll find are hydronium ions (H₃O⁺) and chloride ions (Cl⁻), all swimming around in a sea of water molecules (H₂O).
The concentration of these ions is what really matters. If you have a lot of HCl dissolved in a little water, you've got a concentrated solution. This means there are tons of hydronium ions zipping around, and things are going to get pretty intense. Think of it as a really energetic party – lots of noise, lots of activity! These concentrated solutions can be quite corrosive and you definitely need to handle them with care. Safety first, always!
On the flip side, if you have a tiny bit of HCl in a whole ocean of water, you've got a dilute solution. The hydronium ions are still there, but they're spread out and have a lot more personal space. It's like a chill gathering with just a few friends. Dilute solutions are much less reactive and generally safer to work with. It’s the difference between a playful pup and a majestic, but somewhat aloof, lion.
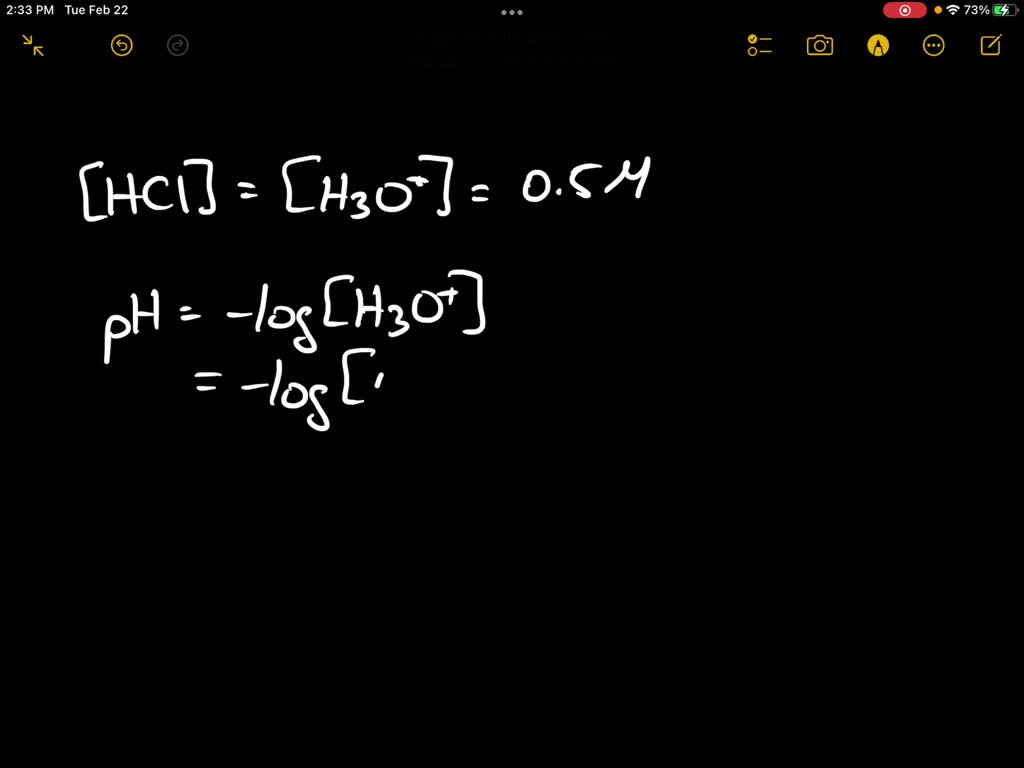
So, the key takeaway is that an HCl solution isn’t just water with some HCl molecules bobbing around. It’s a dynamic mix where HCl has completely transformed. The presence of those hydronium ions (H₃O⁺) is the defining characteristic. They’re the secret sauce that makes it acidic. Without them, it would just be salty water, and nobody wants that when they’re expecting an acid!
Why is this important, you ask? Well, hydrochloric acid is a workhorse in the chemical world. It’s used in everything from cleaning metal (pickling steel, fancy term for it!) to producing dyes and even in your own stomach to help digest food! Yes, your tummy has its own little HCl factory working overtime to break down that burger you had for lunch. Pretty neat, huh? It’s a natural marvel!
Understanding what’s actually in the solution helps us predict how it will behave. Knowing about those hydronium ions tells us it will react with bases (the opposite of acids), it will change the color of indicators (like litmus paper – blue turns red, a classic!), and it will generally be a bit of a chemical powerhouse. It’s like knowing your friend is a master baker; you expect delicious cookies! You expect acidity!

Let's recap our little molecular party. In a solution of HCl, you're primarily dealing with:
- Hydronium Ions (H₃O⁺): These are the main event, the reason it's acidic.
- Chloride Ions (Cl⁻): They're the quiet observers, the byproducts of HCl's dissociation.
- Water Molecules (H₂O): The ever-present host, making sure everyone has enough space and a comfortable environment.
It's fascinating how a seemingly simple substance like hydrogen chloride can create such a powerful and versatile solution when combined with water. It’s a testament to the amazing world of chemistry, where things are rarely what they seem at first glance. It's like a magic trick, but with real scientific explanations!
The beauty of chemistry is in these transformations. It’s not just about elements and compounds; it’s about how they interact, change, and create new properties. An HCl solution is a perfect example of this – a substance that’s more than the sum of its initial parts, thanks to the incredible power of dissociation and solvation.

So, the next time you hear about hydrochloric acid, you can think of it as a lively mix of hydronium ions, chloride ions, and water molecules, all working together to make the world a little more interesting (and a lot cleaner!). It’s a powerful reminder that even the most common substances have complex and exciting stories to tell.
And hey, even if you're not planning on becoming a chemist, a little understanding of these basic principles can make the world around you feel a bit more magical. It’s like having a secret decoder ring for everyday science! So, keep those curious minds buzzing, explore the wonders of chemistry, and remember that even the most potent solutions are just a collection of molecules having a grand old time.
Isn't science just the coolest? It’s everywhere, in everything we do, and understanding it just opens up a whole new universe of appreciation. So, go forth, be curious, and remember that every solution, no matter how simple or complex, has a story waiting to be discovered. Happy exploring!





