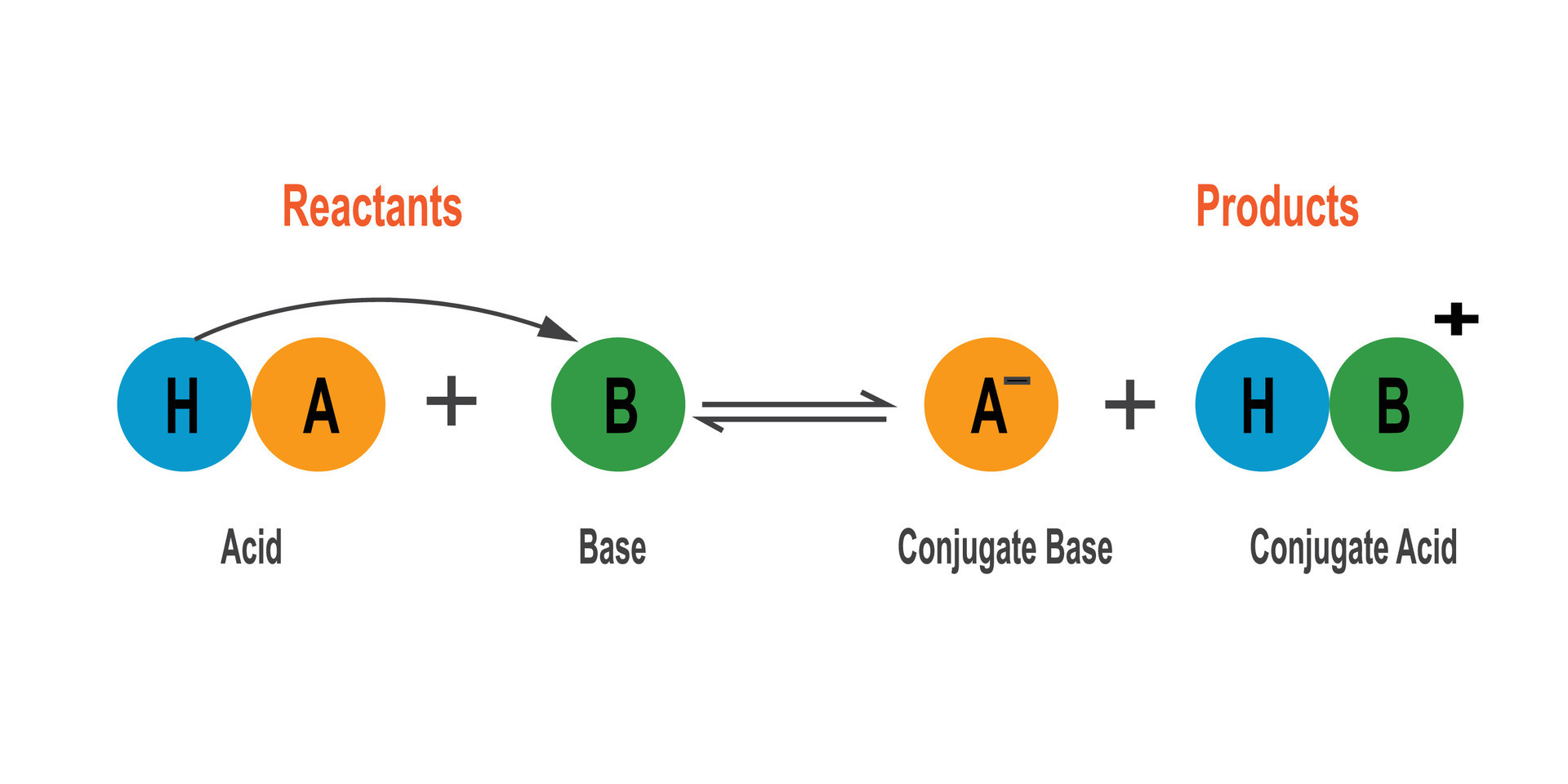
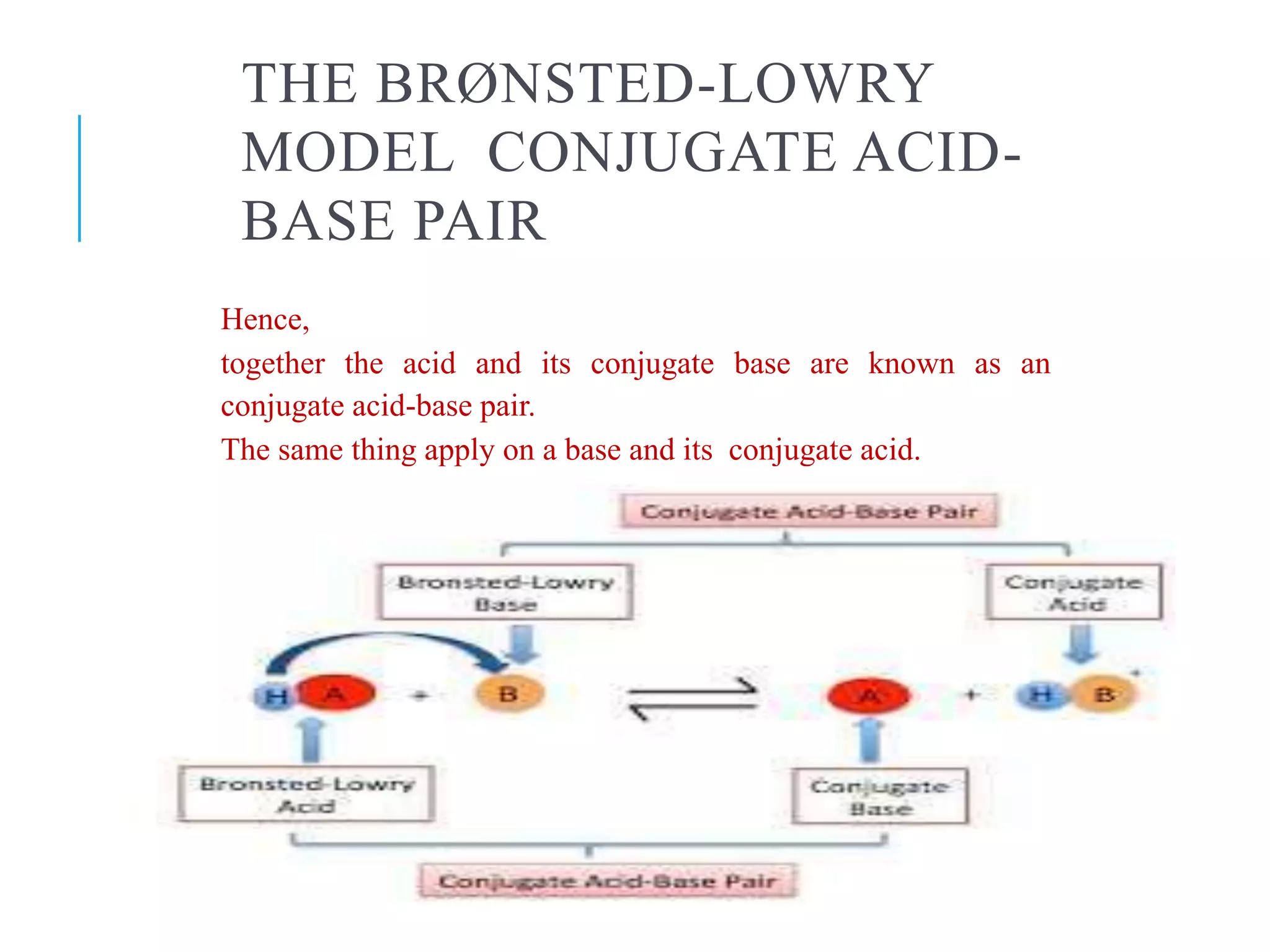
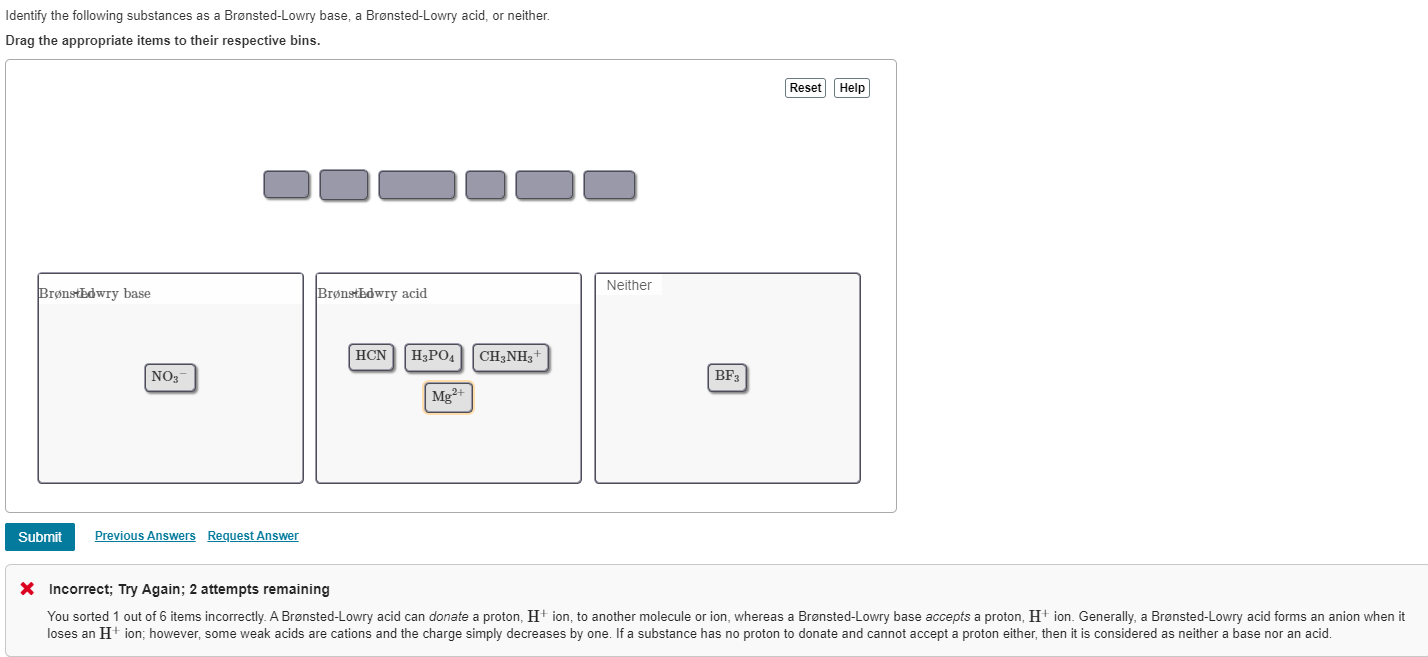
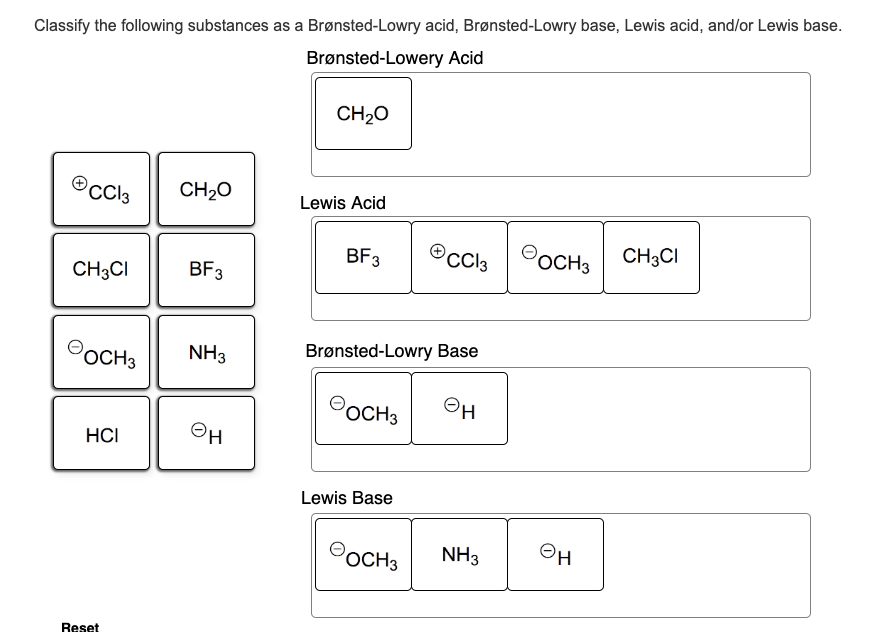
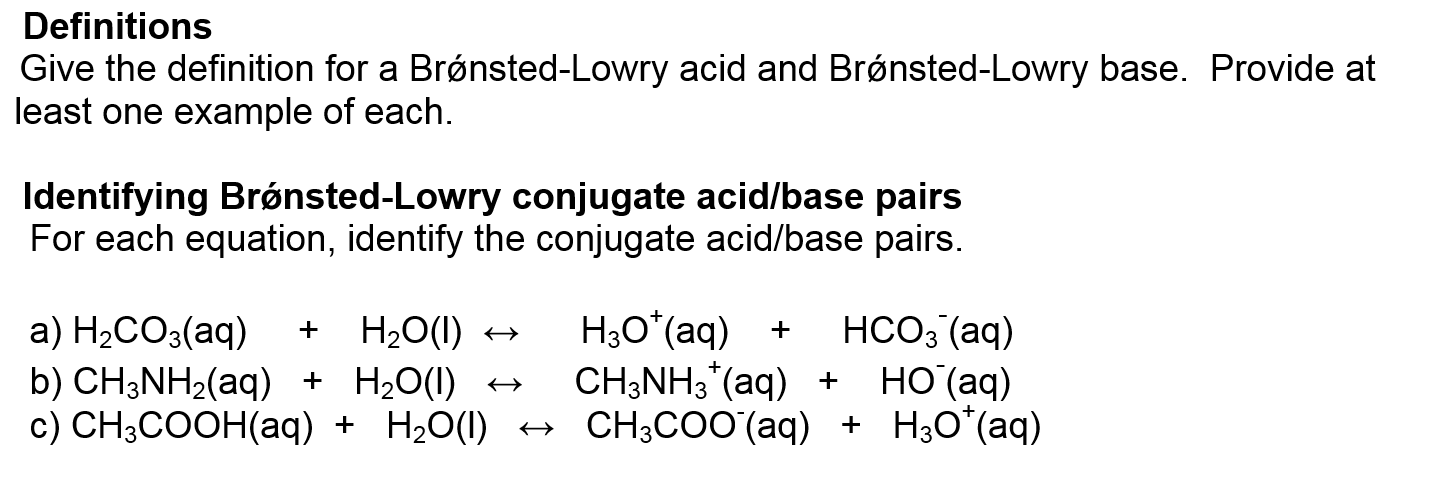A Brønsted Lowry Base Is A Substance Which
Ever wondered what makes some things feel "slippery" like soap, or why baking soda can help calm a tummy ache? It all boils down to some pretty neat chemistry, and one of the key players is something called a Brønsted-Lowry base. Don't let the fancy name fool you; understanding this concept can be surprisingly fun and incredibly useful in our everyday lives. It's like unlocking a secret code that helps us understand the world around us a little better, from the kitchen to the garden!
So, what exactly is a Brønsted-Lowry base? In simple terms, it's a substance that can accept a proton. Now, a proton is just a tiny part of an atom. Think of it like a little charged LEGO brick that can be passed around. A Brønsted-Lowry base is the one that's happy to pick up that brick from another molecule. This might sound a bit abstract, but it has real-world implications. For beginners, it's a fantastic entry point into the world of chemistry without getting bogged down in complex theories. For families, it can turn everyday activities like baking or cleaning into mini science experiments. And for hobbyists, whether you're into brewing, gardening, or even making your own skincare, understanding bases can help you achieve better results.
Let's look at some familiar examples. You might know ammonia, the pungent stuff in some cleaning products. It's a classic Brønsted-Lowry base because it readily accepts protons. Another common one is baking soda (sodium bicarbonate). That's why it's great for neutralizing stomach acid – it accepts those extra protons making your stomach feel better. Even things like milk and egg whites have slightly basic properties. The "slippery" feel of soaps and detergents is also a characteristic of many bases. They often have hydroxide ions (OH-), which are excellent proton acceptors.
Must Read
Getting started with exploring Brønsted-Lowry bases is easier than you think. You don't need a lab coat! A great starting point is to observe common household items. Try adding a little baking soda to vinegar – that fizzing reaction? That's an acid (which gives away protons) reacting with a base (which accepts them). You can also explore the pH scale, which measures how acidic or basic something is. Products with a pH above 7 are considered basic. Just remember to always be careful, and perhaps supervise children during any experiments!
In essence, a Brønsted-Lowry base is a substance that acts as a proton acceptor. It's a fundamental concept in chemistry that helps explain a wide range of phenomena we encounter daily. From making our food taste right to keeping our homes clean, understanding bases adds a layer of fascination and practicality to our lives. So next time you're baking or cleaning, take a moment to appreciate the hidden chemistry at play – it's more enjoyable and useful than you might imagine!