Why Would A Cell Ferment Rather Than Respire

Imagine your tiny cell, living its best life, working away inside you. It needs energy to do all its cool jobs, like building new bits or sending messages. Now, think of energy production as a party. Cells have two main ways to get this energy party started: respiration and fermentation. Respiration is like the big, fancy bash, lots of people, lots of food, very organized. Fermentation? Well, fermentation is more like a spontaneous, slightly chaotic but super fun dance-off in the kitchen!
So, why would a cell ditch the big, organized party (respiration) for the impromptu kitchen dance-off (fermentation)? It’s not that fermentation is better in the long run. Respiration is way more efficient at cranking out energy, like getting way more bang for your buck. But sometimes, the cell just can't get the ingredients for the big party. Or, maybe it’s in a rush!
Let's dive into the kitchen dance-off, also known as fermentation. Think about when you eat that delicious bread, or enjoy a tangy yogurt. You're actually enjoying the byproducts of fermentation! Yeast, those little guys who make bread rise, and bacteria, those tiny organisms in yogurt, are masters of this. They’re not doing it for us, of course. They’re doing it for themselves, to get that quick burst of energy.
Must Read
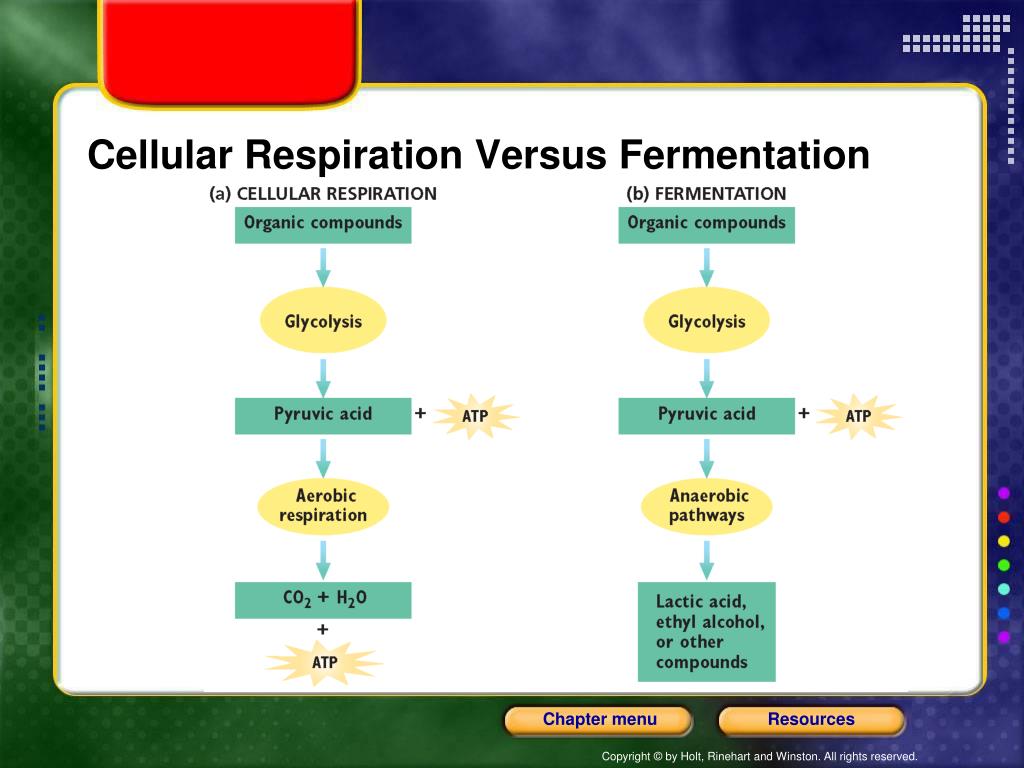
The main ingredient for both respiration and fermentation is glucose. Glucose is like the cell's energy snack bar. When oxygen is around in good amounts, the cell can run the full, fancy respiration marathon. This involves a few stages, like glycolysis, the Krebs cycle, and the electron transport chain. It’s a whole production, with lots of steps and byproducts like carbon dioxide and water. It’s like a well-rehearsed play.
But what if there's no oxygen? Or not enough? This is where fermentation steps in, like a superhero without a cape, just a really good attitude. When oxygen is scarce, the cell can't complete the entire respiration process. It gets stuck. It's like trying to bake a cake but realizing you’re missing the oven! So, it switches gears. It says, "Okay, plan B! Let's do this quick thing instead!"

The first step, glycolysis, is common to both respiration and fermentation. This is where glucose gets broken down into a smaller molecule called pyruvate. It's like chopping up your ingredients. Glycolysis gives the cell a little bit of energy right away, a small snack while it figures out the next move. This is the initial energy boost that both pathways can access.
Now, here’s where the fork in the road appears. In respiration, pyruvate goes on a journey through the oxygen-loving stages. But in fermentation, without oxygen, pyruvate needs a different fate. It can't just hang around. It needs to be dealt with so that glycolysis can keep happening and the cell can get its tiny energy fixes.
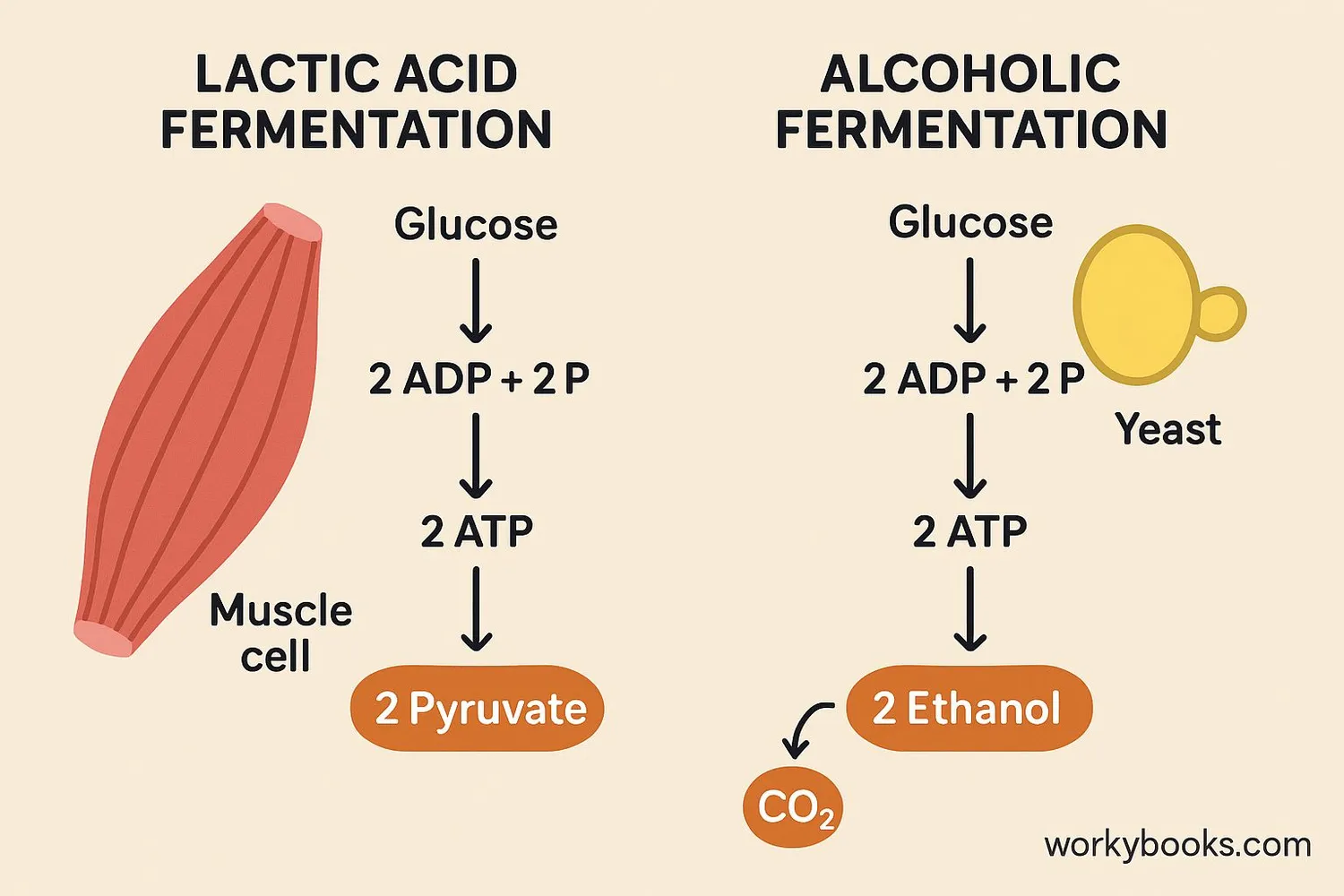
This is where the "fun" really starts. There are different types of fermentation, and they’re like different dance styles. The most famous ones are lactic acid fermentation and alcoholic fermentation.
Think about when you do a really intense workout, like sprinting or lifting heavy weights. Your muscles suddenly need a TON of energy, super fast. Sometimes, the oxygen supply can't keep up with the demand. So, your muscle cells might resort to lactic acid fermentation. Pyruvate gets converted into lactic acid. This process regenerates a molecule called NAD+, which is essential for glycolysis to continue. It's like a quick reset button. That burning sensation you feel in your muscles? That's partly the lactic acid! It’s the cell’s way of saying, "Phew, that was a close one, but we got some energy!"
Then there’s alcoholic fermentation. This is the one our friend, yeast, is famous for. Yeast, when it’s hungry and doesn’t have much oxygen, takes pyruvate and turns it into ethanol (that’s the alcohol in drinks!) and carbon dioxide. That carbon dioxide is what makes bread rise and gives fizzy drinks their bubbles! It’s like the yeast is having a little party and letting off some steam (and gas!). It's pretty cool that these tiny organisms are responsible for so many of the delicious things we enjoy.

So, why would a cell choose this less energy-packed route? It’s all about survival and speed. When oxygen is limited, fermentation is the best option for getting some energy, some ATP, rather than none at all. It’s like getting a little bit of cash when you can’t get the full paycheck. It keeps the essential functions going.
It's also a way for cells to adapt to their environment. Think about deep sea creatures or bacteria living in muddy bogs where oxygen is scarce. They've evolved to thrive on fermentation! They’ve perfected their kitchen dance-offs.
So, next time you’re enjoying a slice of sourdough bread, or perhaps a refreshing beer, take a moment to appreciate the humble act of fermentation. It’s a testament to the incredible adaptability and ingenuity of life at its smallest scale. It's a less glamorous, but undeniably vital, process that keeps the energy party going, even when the main ballroom is closed. It's a quirky, energetic solution that shows even in the tiniest of worlds, there's always a way to keep dancing!




:max_bytes(150000):strip_icc()/cellular_respiration-8fcc3f1ad3e54a828dabc02146ce4307.jpg)
