Why Is Racemic Mixture Not Optically Active

Ever wondered why some molecules are like perfectly balanced dancers, and others… well, not so much? We’re talking about something called chirality. It’s a fancy word, but think of it like your hands. Your left hand is a mirror image of your right hand, right? But you can’t quite put your left glove on your right hand. They’re similar, but not identical. That’s chirality in a nutshell for molecules.
Now, some molecules can exist in two "handed" forms. We call these forms enantiomers. They’re like those mirror-image hands. Super close, but flipped. And here’s where it gets interesting: these enantiomers can behave differently. Especially when it comes to something called plane-polarized light. Imagine light waves all wiggling in the same direction. It's neat and tidy. But when this special light passes through certain chiral molecules, it gets a little… jazzy. It rotates! One enantiomer might twist it one way, and its mirror image will twist it the exact opposite way. It’s like they have their own little dance moves for light.
This ability to twist light is what makes a molecule optically active. It’s like the molecule is showing off its unique personality to the light. Pretty cool, huh? It’s the reason why, in some cases, one form of a drug might work wonders, while its mirror image could be useless, or even harmful. We definitely want our medicines to be the right "handed" ones, don’t we?
Must Read
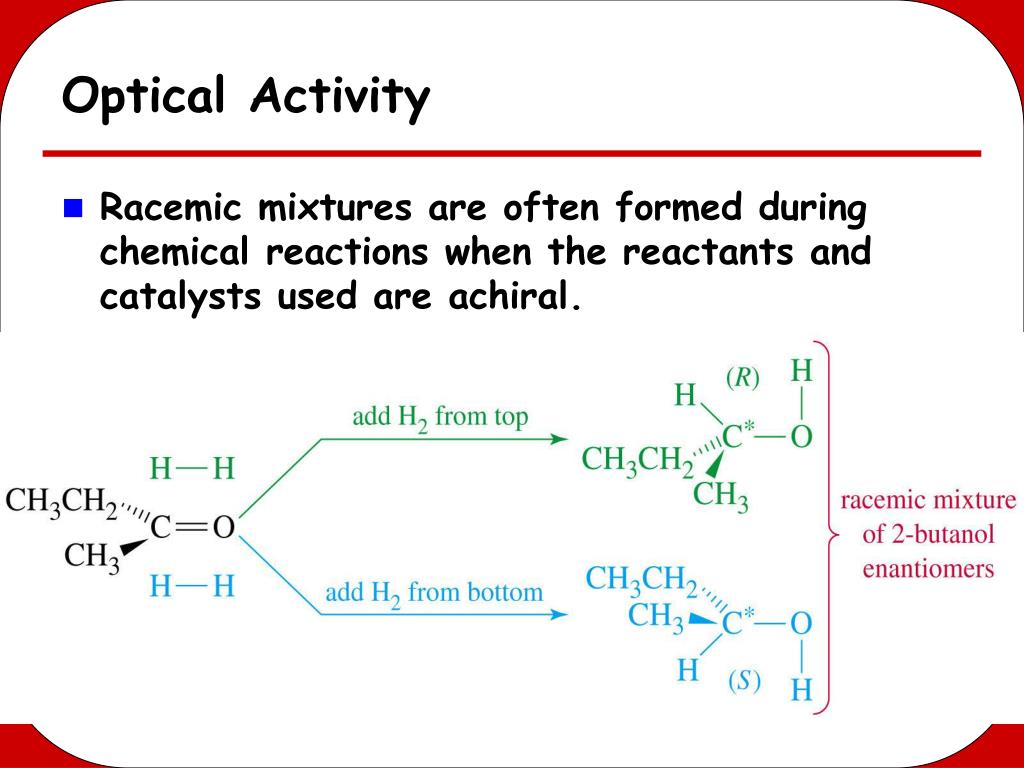
But then, there’s this thing called a racemic mixture. And let me tell you, this is where things get a bit… meh. A racemic mixture is basically a 50/50 blend of those two enantiomers. Think of it like having an equal number of left-handed gloves and right-handed gloves in a box. You’ve got both, all mixed up.
So, why is this perfectly balanced, 50/50 blend of chiral molecules not optically active? This is the question that keeps chemists up at night, probably while sipping lukewarm coffee. It’s like asking why a perfectly balanced seesaw doesn’t move. Or why a perfectly neutral joke doesn’t make anyone laugh (or cry). It’s just… balanced.

Here’s the slightly mind-bending part. Remember how one enantiomer twists the plane-polarized light one way, and the other enantiomer twists it the exact opposite way? In a racemic mixture, you have just as much of the "left-twisting" molecule as you do the "right-twisting" molecule. They’re in a standoff. A molecular tug-of-war.
So, if the left-twisting one tries to rotate the light clockwise, the right-twisting one is right there, trying to rotate it counter-clockwise with equal force. They cancel each other out! It’s like having a loud argument and a quiet apology happening at the same time. The net result? Silence. Or, in this case, no rotation of the light. The light goes through looking exactly as it did before, totally unfazed by the molecular party happening inside.

It’s the ultimate act of molecular indifference. They’ve got their own chirality, their own handedness, but when they team up equally, they decide to be boring together.
It’s almost a shame, really. All that potential for optical fancy-footwork, just… nullified. It's like buying a superhero costume and then deciding to stay home and watch TV. You’ve got the outfit, but you’re not using your powers.

This is why, in the world of chemistry, if you want to see that cool light-twisting effect, you need to have a sample that is enantiomerically pure. That means it’s either all one "handed" form, or at least has a significant excess of one over the other. If it’s a perfect 50/50 split, the optical activity takes a vacation. It’s on strike.
Think of it like this: imagine you’re at a party. If everyone is dancing wildly in different directions, it’s a lively scene. But if you have exactly half the people doing the Macarena and the other half doing the Hokey Pokey in perfect opposition, the whole dance floor just ends up looking… still. No net movement. The energy cancels out.
So, the next time you hear about a racemic mixture, you can nod knowingly. It’s the molecule that’s technically got it, but doesn’t show it. It's the Jekyll and Hyde who always end up at a truce, leaving the world of optical activity a little less sparkly. And maybe, just maybe, that’s okay. Sometimes, perfect balance is just… balanced. And that’s its own kind of (unpopular?) truth.