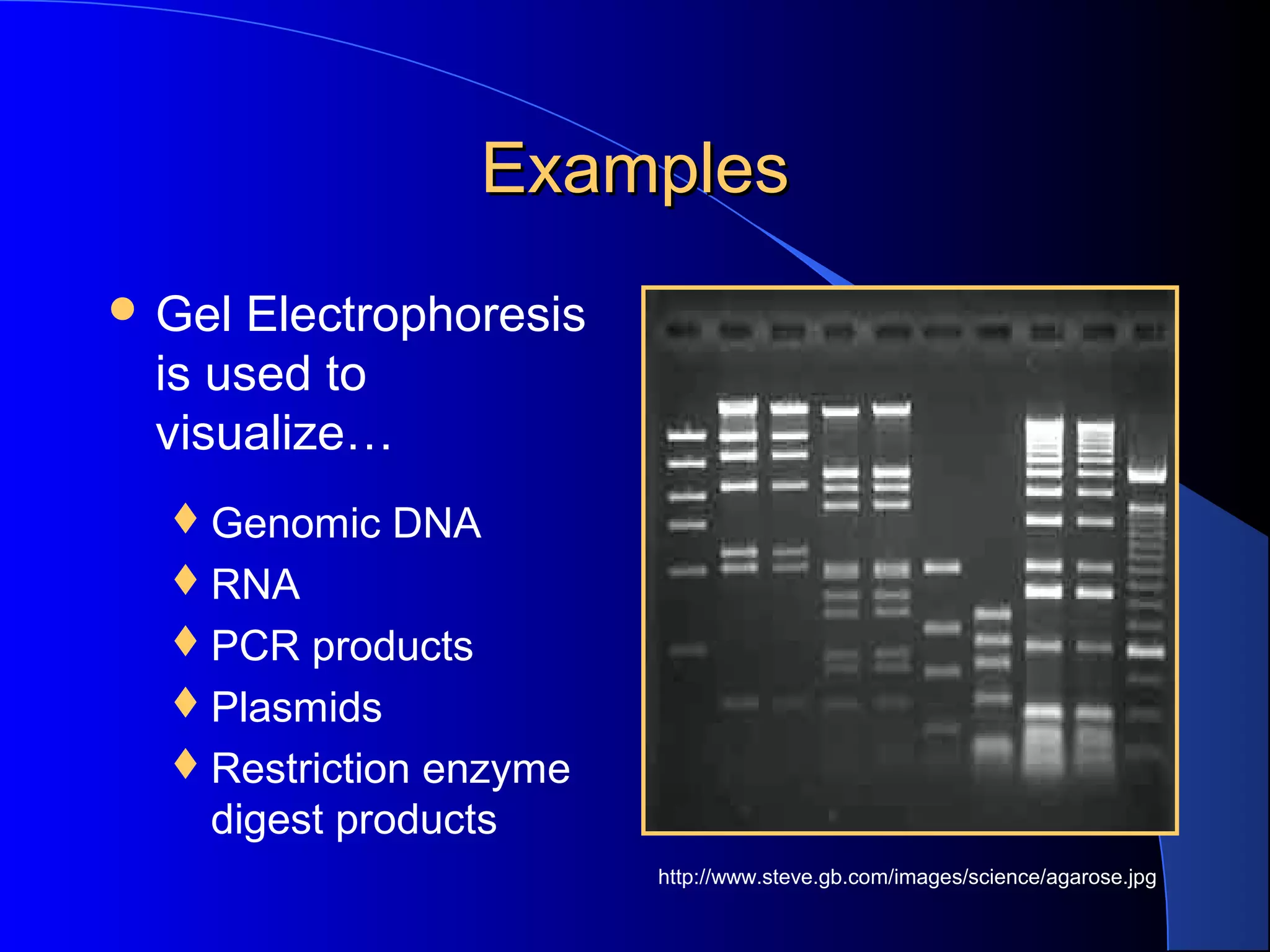
Why Is Glycerol Used In Gel Electrophoresis
Ever found yourself staring at a gel electrophoresis setup, perhaps in a movie or a particularly exciting biology documentary, and wondered, "What's that syrupy stuff doing in there?" You know, that slightly viscous liquid that sometimes makes an appearance? Well, get ready to have your mind gently blown, because we're about to dive into the wonderful world of glycerol in gel electrophoresis, and trust me, it's way cooler than it sounds!
So, what's the big deal with glycerol, you ask? Is it just there for giggles? Absolutely not! While it might seem like a minor player, glycerol is actually a superstar in the world of gel electrophoresis, making your DNA- or protein-separating adventures a whole lot smoother, literally!
Let's break it down, shall we? Imagine you're trying to run some precious tiny molecules, like DNA fragments, through a gel. Think of the gel as a maze, a super-fine, intricate labyrinth designed to sort these molecules by size. Now, you want those molecules to dive into this maze in a nice, orderly fashion, right? You don't want them to just sort of drift in haphazardly.
Must Read
This is where our friend, glycerol, swoops in to save the day! Its primary role? To act as a density agent. Yep, you heard that right! Glycerol is a pretty dense liquid. Think of it like adding a little bit of heavy syrup to water – it makes the water heavier, or denser. When you add glycerol to your sample buffer (the liquid you mix your DNA or protein with before loading it onto the gel), you're essentially giving it some extra weight.
Why is this weight so important? Because when you load your samples into the wells of the gel, that extra density from the glycerol helps your samples sink right in! Without it, your precious DNA or protein samples might just float around on the surface, or worse, drift out into the buffer solution before they even get a chance to start their journey through the gel maze. Not ideal, is it?
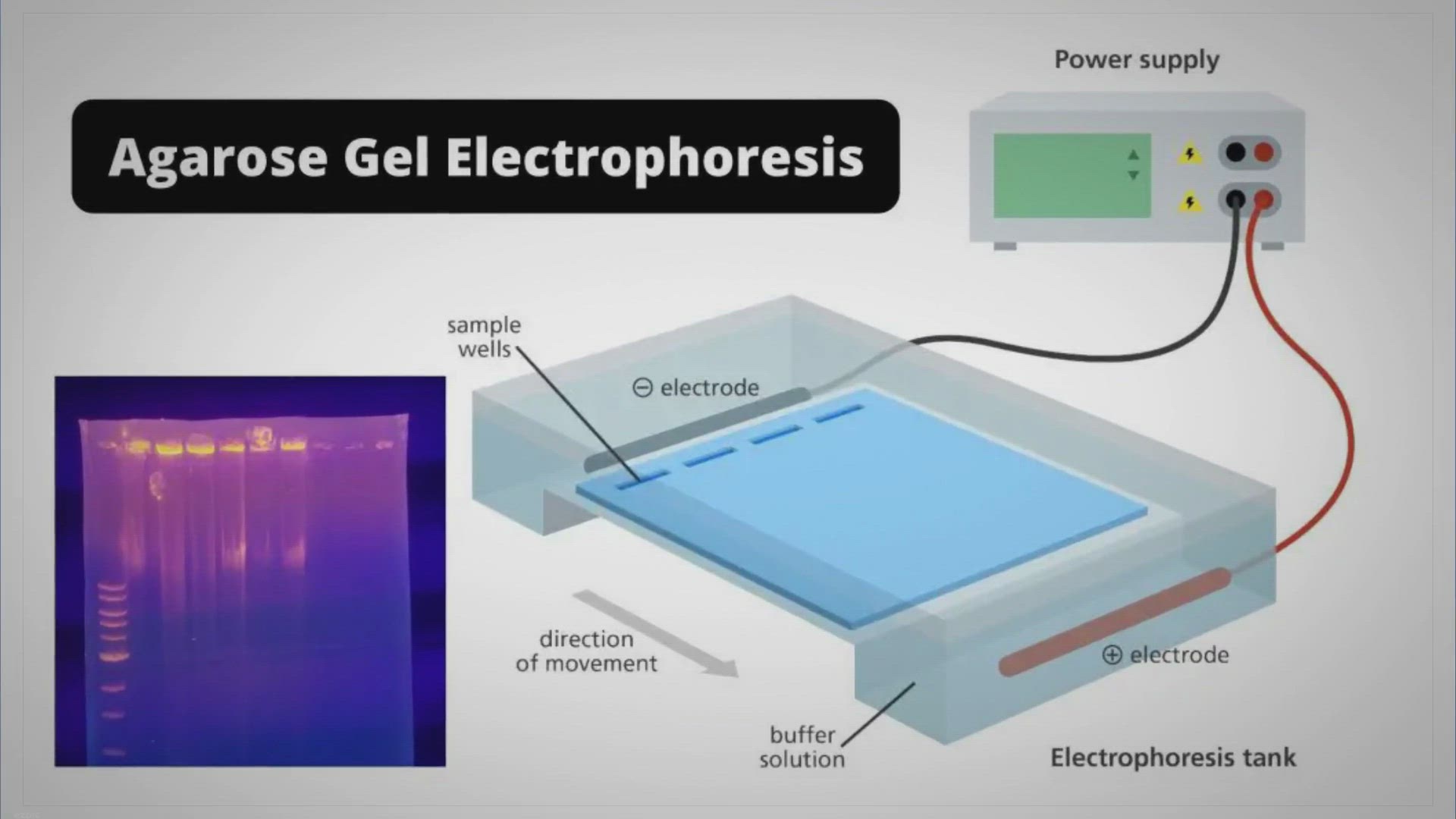
Think of it like trying to drop marbles into a glass of water. If you just plop them in, they might splash around a bit. But if you carefully place them, they sink nicely to the bottom. Glycerol helps your samples behave more like those carefully placed marbles, ensuring a clean and successful loading.
Making Your Gel Run Smoothly (Literally!)
But glycerol's talents don't stop at just getting your samples into the gel. It also helps to ensure that your samples migrate through the gel in a nice, tight band. This is super important for getting clear, sharp bands when you visualize your results. Fuzzy bands? Nobody wants those!
When your DNA or protein molecules start their journey through the gel, driven by an electric current, they naturally tend to spread out a bit. This is especially true for your samples that are loaded first. However, glycerol’s presence in the sample buffer can help to maintain the integrity of the sample band as it enters the gel. It essentially helps to keep the molecules clustered together, preventing them from diffusing too quickly.
So, that little bit of syrup-like substance is actually helping to keep your experiment from turning into a blurry mess. Pretty neat, huh? It’s like a little helper, whispering to your molecules, "Stay together, team! We've got a maze to conquer!"
A Little Bit of Shine, Too?
While its primary functions are density and band integrity, glycerol can also add a touch of aesthetic appeal to your gels. When you're working with clear gels, the glycerol can give your sample wells a nice, shiny appearance. It’s a small detail, perhaps, but in the sometimes-serious world of lab work, those little visual cues can be surprisingly satisfying!
Think about it: you're meticulously preparing your samples, loading them with care, and then you see those beautifully defined, slightly glistening wells. It’s a small victory, a sign that things are on track and that your experiment is set up for success. It’s the little things that make the scientific journey more enjoyable!

And let’s not forget that glycerol is a humectant, meaning it attracts and retains moisture. While this might not be its main gig in gel electrophoresis, it can contribute to keeping the gel itself from drying out too quickly during the run. This can be particularly helpful in longer electrophoresis experiments where evaporation could become an issue.
Is It Always Necessary?
Now, you might be wondering if every gel electrophoresis protocol absolutely demands glycerol. The short answer is: not always. Some very specific protocols might opt for other density agents or might be designed in such a way that glycerol isn't strictly required. However, for the vast majority of standard DNA and protein gel electrophoresis applications, especially for beginners, you'll find glycerol to be an incredibly useful and common component of your loading buffer.
It’s one of those unsung heroes in the lab. You might not always consciously think about it, but its presence makes a significant difference in the reliability and clarity of your results. It’s like the quiet friend who always has your back, ensuring everything runs smoothly behind the scenes.

So, next time you see glycerol in your gel electrophoresis setup, give it a little nod of appreciation. It's not just some random ingredient; it's a carefully chosen tool that helps you unlock the secrets hidden within your DNA or proteins. It’s a facilitator of scientific discovery, a tiny but mighty contributor to the accuracy of your experiments.
Learning about these seemingly small details can actually make the entire process of scientific investigation more engaging and less intimidating. It demystifies the techniques and shows you that even the most complex experiments are built on a foundation of clever, practical solutions. Understanding why things are done a certain way empowers you and makes you a more confident and curious scientist, even if you're just experimenting in your own home!
So, keep asking those questions! Keep digging a little deeper into the "why." Because every answer you find, whether it’s about glycerol in gels or the intricate dance of molecules, is a step towards a more fascinating and inspiring journey of learning. The world of science is full of these little wonders, just waiting for you to discover them!