Why Does The Reactivity Decrease In Group 7
Hey there, fellow explorers of the universe, from the cosmic dust in the furthest galaxies to the humble atoms right here in our kitchens! Ever wondered about those super-cool, super-reactive elements that make up, well, a surprising chunk of our world? We're talking about the halogen family, also known as Group 7 of the periodic table. Think fluorine, chlorine, bromine, iodine, and astatine – the gang's all here! They're the life of the chemical party, always eager to jump into a reaction. But here's a quirky little secret: as you go down this group, things start to chill out a bit. Their reactivity, that fiery spark, actually starts to decrease. It's like watching a rock star gradually mellow out into a wise, contemplative artist. Let's dive into why this happens, with a vibe as relaxed as your favorite Sunday morning. Grab a coffee, settle in, and let's unravel this atomic mystery.
So, what's the big deal with reactivity anyway? In simple terms, it's an element's eagerness to gain or lose electrons to achieve a stable, happy electron configuration, usually by pairing up with other elements. Imagine electrons as little social butterflies, always looking for a dance partner to feel complete. For halogens, they're just one electron short of a full outer shell, making them incredibly keen to snatch one from anyone nearby. This makes them, especially at the top of the group, incredibly powerful oxidizers – the ultimate electron thieves!
Fluorine, perched at the very top of Group 7, is the undisputed champion of electron-grabbing. It's so aggressively reactive that it can even snatch electrons from noble gases, elements that are famously unreactive! Think of fluorine as the "Karen" of the periodic table, demanding what it wants with absolute conviction. It's so potent that it's rarely found in its pure form in nature; it's always busy bonding with something else. This intense reactivity is crucial for things like creating non-stick surfaces (hello, Teflon!) and powering rocket fuels – definitely not your average kitchen ingredient, but undeniably powerful!
Must Read
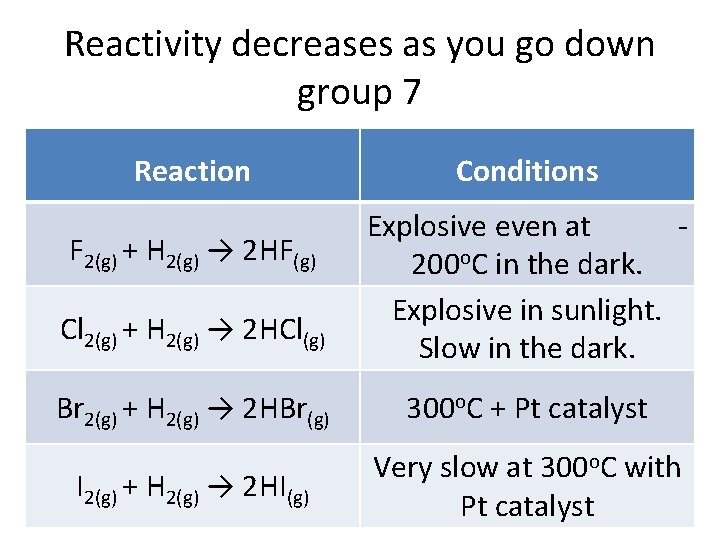
Chlorine, the next in line, is probably the most familiar to us. It's the active ingredient in bleach and swimming pool sanitizers, keeping things clean and germ-free. It's still super reactive, but just a tad less enthusiastic than fluorine. It’s like the slightly more sensible sibling who’s still up for adventure but also appreciates a good book. Chlorine’s widespread use highlights its balance of reactivity and manageability. We harness its power for sanitation and industrial processes, but we also take precautions because, well, it's still a strong character!
As we move further down, we meet bromine, iodine, and astatine. Bromine is a reddish-brown liquid that’s less volatile than chlorine but still packs a punch. It's used in flame retardants and some pharmaceuticals. Think of bromine as the moody artist, intense and expressive but perhaps not quite as universally applicable as chlorine. Iodine, the familiar purple solid, is essential for human health. Our thyroid glands need iodine to function properly, making it a vital nutrient. If you've ever heard about adding iodine to salt, that's to prevent widespread deficiency. It’s the caring elder in the group, providing essential support rather than dominating every interaction. It’s also used as an antiseptic – remember those iodine tinctures from childhood first-aid kits? That sting was its way of saying, "I'm here to help!"
So, what's behind this gradual dialing-down of the halogen hype train?
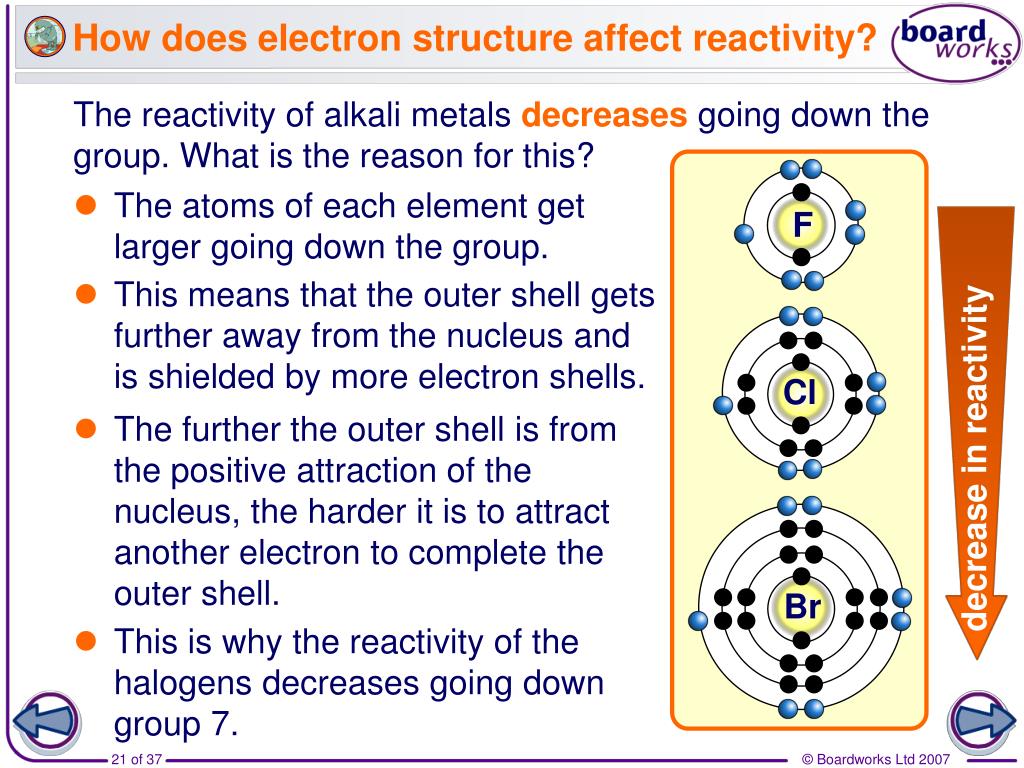
It all comes down to two main players: atomic size and electron shielding. Picture an atom as a tiny solar system. The nucleus is the sun, and the electrons are the planets orbiting it. Now, imagine that as you go down Group 7, the "solar system" gets bigger. The nucleus remains the same positive charge (carrying the protons), but you're adding more electron "shells" or energy levels. These extra shells act like cosmic insulation, creating a barrier between the positively charged nucleus and the outermost electrons.
This is where electron shielding kicks in. The inner electrons, those closer to the nucleus, effectively "shield" the outer electrons from the nucleus's strong pull. Think of it like trying to hear someone whispering across a crowded room – the more people (inner electrons) between you and the whisperer (nucleus), the fainter the sound (attraction) becomes for those on the edges (outermost electron).
Consequently, the positive charge of the nucleus has a weaker grip on the outermost electron as you descend the group. For a halogen, the ultimate goal is to grab an electron to fill its outer shell. If the nucleus's pull on its own outer electrons is already weakening, it's going to be less effective at attracting new electrons from other atoms. It's like trying to persuade someone to join your very exclusive club when your own membership perks are becoming less appealing.
Another critical factor is atomic radius. As we've established, atoms get bigger as you go down a group. Fluorine is a tiny atom, with its single valence electron (the one it wants to gain) very close to the nucleus. This means the nucleus's attractive force is very strong, making fluorine super eager to snatch that electron. Bromine, on the other hand, is a much larger atom. Its valence electron is much further from the nucleus, experiencing less of that direct pull. It's like trying to catch a frisbee when you're standing right next to the thrower versus when you're a good distance away – the closer you are, the easier it is to interact.
This decreasing attraction means that the energy required to remove an electron from the halogen atom (ionization energy) actually increases as you go down the group. Conversely, the energy released when a halogen atom gains an electron (electron affinity) generally decreases (with a slight anomaly for fluorine). This is a bit counterintuitive, isn't it? You’d think a bigger atom would be more willing to accept another electron. But it's precisely because the outer electron is so far away and shielded that it doesn't "feel" the nucleus's welcoming embrace as strongly.

The Cultural Significance of Halogens: More Than Just Chemistry Class
These elements aren't just abstract concepts in textbooks; they've woven themselves into the fabric of our lives and cultures. Chlorine, as we mentioned, is the superhero of clean water, making modern public health possible. Imagine a world without safe, chlorinated tap water – a return to widespread waterborne diseases. It's a chemical unsung hero, quietly protecting millions.
Iodine's role in preventing goiter and cretinism is another profound cultural impact. For centuries, iodine deficiency was a silent epidemic in many regions, leading to significant health problems. The simple addition of iodine to table salt, a widespread public health intervention, dramatically improved cognitive development and overall well-being in countless communities. It's a brilliant example of how chemistry can directly uplift societies.
Bromine has also had its moments, both good and bad. Historically, bromine compounds were used as sedatives, leading to its nickname "the truth serum" in some circles – though its effectiveness and ethical implications are debatable, it certainly captured the public imagination. More practically, its use in flame retardants has saved countless lives by slowing the spread of fires in homes and public spaces, a testament to its chemical prowess in enhancing safety.

Even fluorine, the fierce electron thief, has a gentler side. It’s added to toothpaste and municipal water supplies (in carefully controlled amounts) to strengthen tooth enamel and prevent cavities. So, while it's busy powering rockets, it's also helping us maintain those pearly whites – a duality that’s quite remarkable!
Fun Little Facts to Brighten Your Day
Did you know that fluorine gas is so reactive, it can spontaneously combust with materials that normally wouldn't burn, like glass and even stainless steel, under certain conditions? It's like a chemical wildfire!
Bromine was discovered accidentally by a young German chemist in 1826 who was experimenting with seaweed ash. He was reportedly so annoyed by a spilled sample that he threw it out, only to notice later that it had reacted with the glass container! Talk about a happy accident!
Iodine was first identified in 1811 by a French chemist who was making gunpowder. He noticed a beautiful purple vapor when heating seaweed ash, which was quite a show!

Astatine is one of the rarest naturally occurring elements on Earth. It's estimated that there's less than an ounce of it in the Earth's entire crust at any given time. It's the ultimate mystery element, playing hide-and-seek with scientists!
The name "halogen" itself comes from Greek words meaning "salt-former." This is because when halogens react with metals, they typically form salts!
A Moment of Reflection
It's fascinating, isn't it? This subtle dance of attraction and repulsion, of size and shielding, dictates how these elements interact. And it’s a pretty good metaphor for life itself. We all have our own "outer shells," our own ways of attracting or repelling those around us. Sometimes, the closer we are to the "nucleus" of our core values, the more strongly we can influence those around us. But as we expand our "shells" – through experiences, learning, and growing older – our immediate pull might lessen, replaced by a more nuanced understanding and a different kind of influence. We learn to shield ourselves from negativity while still being open to connection, just like the halogens that mellow out but never lose their fundamental nature.
So, the next time you see a swimming pool shimmering, or brush your teeth with fluoride toothpaste, or even just marvel at the vibrant colors of a sunset (yes, there are halogens in the atmosphere too!), take a moment to appreciate the intricate, and sometimes surprisingly mellow, world of Group 7. Their decreasing reactivity isn't a sign of weakness, but a testament to their evolutionary journey down the periodic table, a journey that has made them essential, in their own unique ways, to the world we inhabit.


