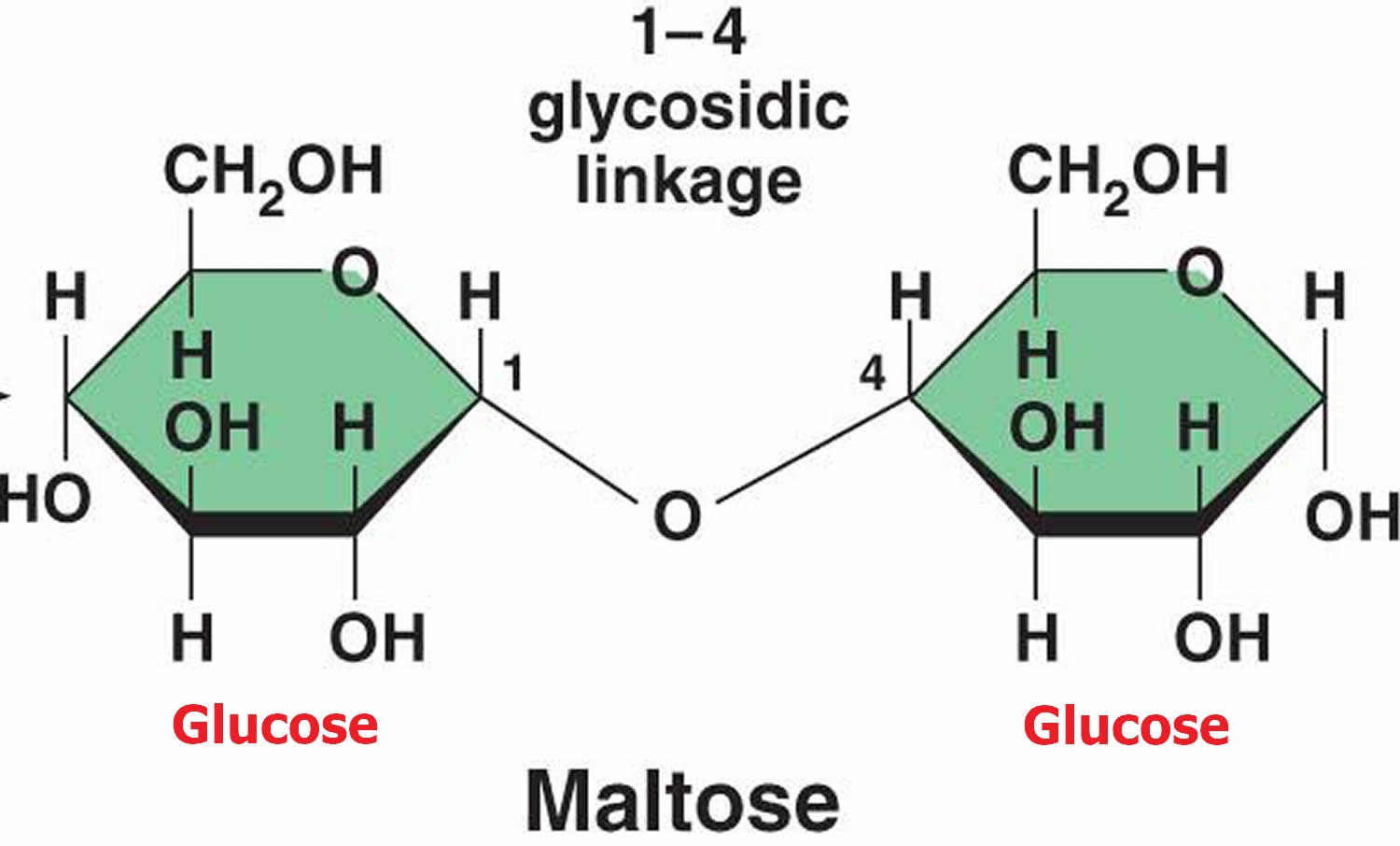
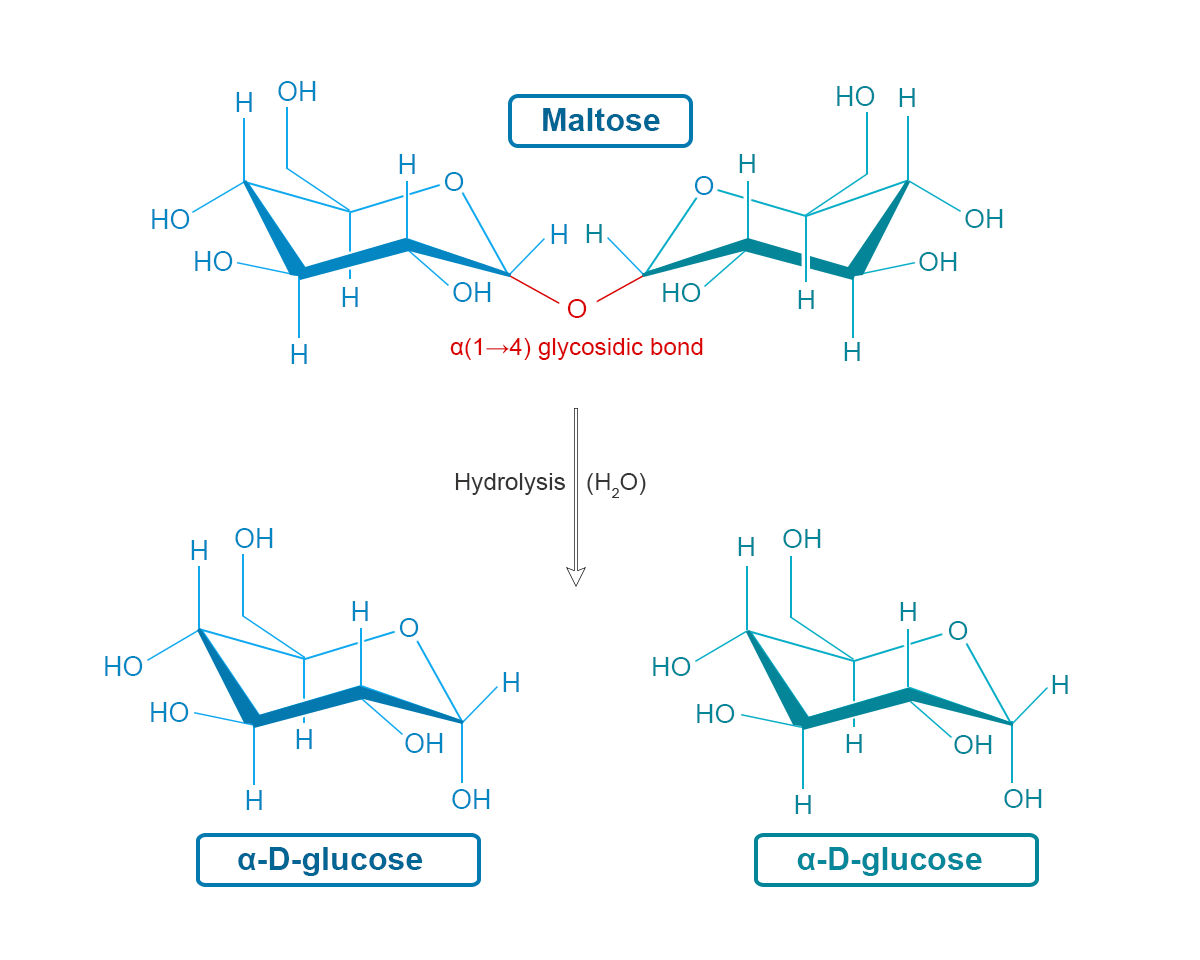
Why Does Maltose Have Both A And B Anomers

Ever wondered about the sweet, comforting taste of maltose? That's the sugar that makes malted milkshakes and those delicious caramel candies so wonderfully chewy. But this sugar has a little secret, a kind of dual personality that’s actually quite charming when you think about it.
Imagine maltose as a molecule that decided it couldn't quite make up its mind about how it wanted to look. It’s like a person who has two favorite outfits and sometimes wears one, and sometimes wears the other, depending on their mood. This indecision is what gives maltose its special ability to exist in two different forms, known as anomers.
These two forms are playfully named alpha (α) anomer and beta (β) anomer. Think of them as fraternal twins – they’re related, share a lot of the same "genes," but have distinct differences. These differences are subtle, but they're enough to make each anomer a unique character in the sugary world.
Must Read
The key to understanding this is to look at a very special atom in the maltose molecule, a carbon atom that’s a bit of a show-off. This carbon atom is like the "pivot point" for maltose’s split personality. It's the place where the molecule can choose to arrange its attached groups in one of two ways.
In the alpha anomer, a particular group (often represented by an -OH, a hydroxyl group) sticks out in a certain direction. It’s like one twin always prefers to stand on the left side of a photograph. This arrangement is super important for how maltose behaves when it's involved in chemical reactions.
Then there’s the beta anomer. This is where that same special carbon atom decides to arrange things differently. The hydroxyl group flips to the other side. It’s the other twin, who insists on standing on the right.
This subtle twist in arrangement might seem tiny, but it has big consequences. It affects how these maltose molecules interact with each other and with other molecules. Think of it like two different handshakes – one might be a firm grip, the other a more relaxed clasp.
So, why does maltose bother having both anomers? Well, it turns out that having these two forms gives maltose a lot of flexibility. It's like having a multi-tool; you can tackle different tasks with it.

One of the main reasons is for storage and energy release. When plants like barley store energy in the form of starch, they use maltose as a building block. The different anomers can be linked together in slightly different ways, creating complex structures that store a lot of sugar.
When our bodies need energy, enzymes come in to break down these starches. These enzymes are like skilled chefs who know exactly how to snip apart the chains of maltose. Some enzymes are picky eaters; they prefer to work with one anomer over the other.
Having both alpha and beta anomers means that maltose can be broken down by a wider variety of these enzyme "chefs." It’s like having a pantry stocked with two different types of keys, ensuring that you can always unlock the energy when you need it. This ensures a reliable and efficient energy supply.
This adaptability also plays a role in how maltose is used in food production. The subtle differences in how the anomers interact can affect texture, sweetness, and how the sugar behaves during cooking and baking. That perfect caramel chewiness? A little bit thanks to these dual anomers.
It’s also a testament to the elegance of nature. Molecules don’t always have to be simple and straightforward. Sometimes, having a bit of variation, a touch of duality, makes them more robust and versatile.
Think of it as a molecular dance. The alpha anomer might waltz in one pattern, while the beta anomer performs a slightly different jig. Both dances are beautiful and serve a purpose in the grand ballroom of biochemistry.
The cool part is that these anomers are constantly interconverting in solution. They’re like little acrobats, flipping back and forth between their alpha and beta forms. This dynamic equilibrium is crucial for many biological processes.
It's not just about survival; it's about being good at what you do. Maltose, with its two faces, is exceptionally good at being a source of energy and a building block for larger carbohydrates. It’s a molecule that’s ready for anything.
So, the next time you enjoy a malted treat, take a moment to appreciate the subtle complexity of the maltose within. It’s not just sugar; it’s a fascinating molecule with a charming duality, always ready to be what’s needed.
The story of maltose’s anomers is a little reminder that even the simplest things in life can have hidden depths and intriguing personalities. It’s a tiny piece of the amazing molecular world that makes our food taste so good.
It’s a heartwarming thought, isn’t it? That something as common as sugar has this kind of sophisticated adaptability. It’s a quiet strength, a molecular adaptability that ensures its vital role.
The alpha and beta anomers are not just abstract chemical concepts; they are the reason why maltose can efficiently participate in the grand cycle of energy and sustenance. They are tiny heroes in the microscopic world.
So, embrace the anomers! They are the secret sauce, the little twists that make maltose the sweet and reliable sugar we know and love. It’s a lesson in how variation can lead to strength and versatility.
This dual nature isn’t a flaw; it’s a feature. It’s what allows maltose to be a key player in so many biological processes. It’s a testament to evolution’s clever design.
It makes you wonder what other everyday things have such surprising molecular secrets. Perhaps your favorite cookie has a sugar with an even more complex story. The world of food science is full of these delightful discoveries.
Ultimately, the presence of both alpha and beta anomers in maltose is a beautiful illustration of molecular diversity. It’s a reminder that even in the simplest molecules, there's a rich and dynamic chemistry at play.
So, next time you savor that malty flavor, give a little nod to the alpha and beta anomers. They’re the unsung heroes behind that delicious sweetness. They’re the reason maltose is so adaptable and so essential.
It's a delightful dance of atoms, a chemical ballet that ensures your enjoyment. And isn't that just the sweetest thing? The molecules themselves are working to make life a little bit tastier and more energetic for us.
The flexibility that these anomers provide is paramount for efficient energy metabolism. It's like having multiple tools in a toolbox, each suited for a different task in breaking down or building up sugars. This redundancy ensures that energy is always accessible.
This duality ensures that maltose can engage with a variety of biological machinery. It's not a one-trick pony; it's a versatile performer ready to meet the demands of living systems. This adaptability is key to its widespread use in nature.
So, the next time you enjoy a malty beverage or a caramel candy, remember the fascinating duality of maltose. It’s a testament to the elegant complexity that underlies the simple pleasures of life.
It's a little bit of molecular magic that makes our food delightful and our bodies energized. The story of alpha and beta anomers is a sweet one indeed. It's a scientific detail that adds a layer of appreciation for the foods we love.