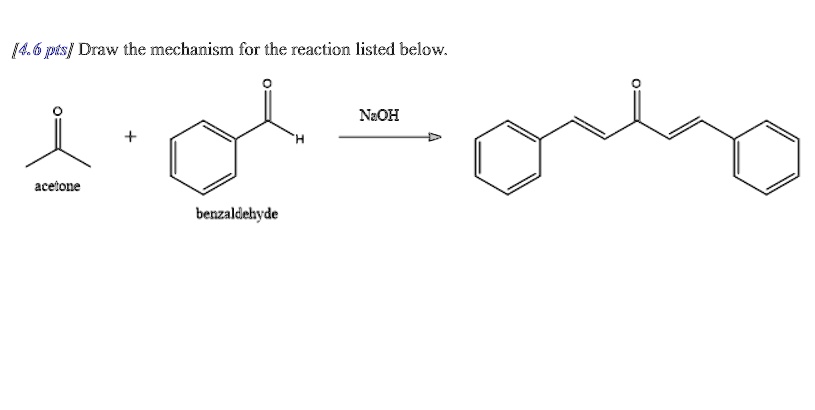
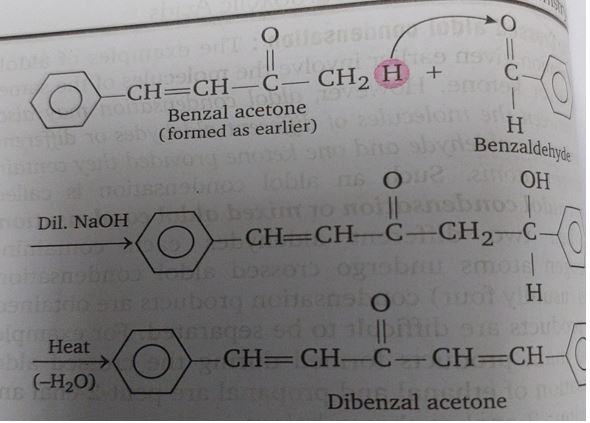
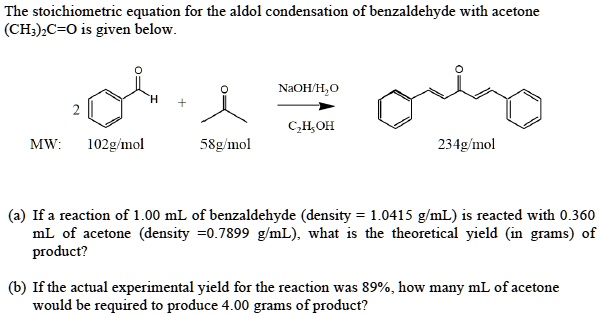
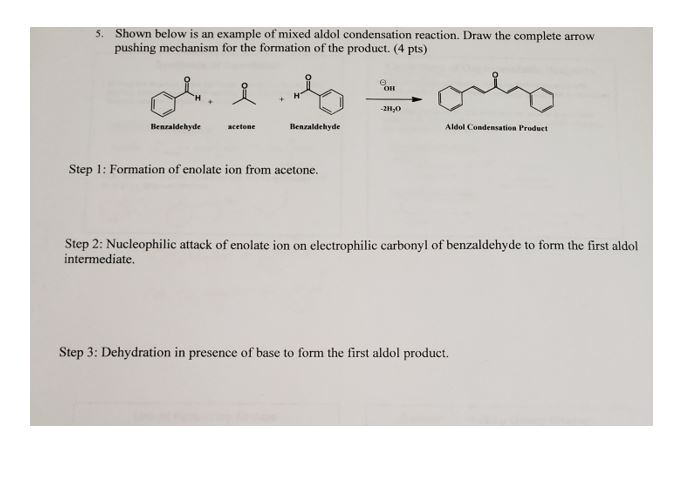
Why Does Acetone Enolate Attack Benzaldehyde
Alright, gather 'round, my fellow flavor enthusiasts and chemical voyeurs! Today, we're diving headfirst into a culinary-adjacent mystery that would make even Hercule Poirot scratch his magnificent mustache: why, oh why, does the humble acetone enolate have a fiery, almost passionate, fling with benzaldehyde?
Now, I know what you're thinking. "Acetone? Benzaldehyde? Isn't that the stuff that smells like nail polish remover and slightly alarming almonds?" And you'd be partially right! Acetone, our unsung hero of clean-up and paint stripping, is surprisingly versatile. And benzaldehyde? Well, that's the aromatic heart of cherries and almonds, the subtle whisper of marzipan. They seem about as compatible as a cat and a vacuum cleaner.
But chemistry, my friends, is a twisted mistress. She loves to throw together the unlikeliest of couples and then marvel at the sparks that fly. And in this particular love story, our protagonists are none other than the enolate of acetone and the charmingly electropositive benzaldehyde.
Must Read
The Awkward Introduction: Acetone's Dramatic Makeover
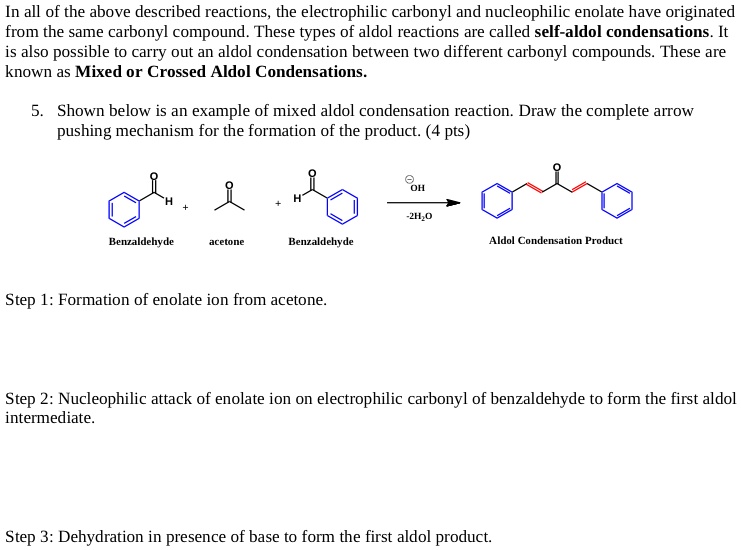
First, let's talk about acetone. In its natural state, it's like a perfectly content couch potato. It's got these two little hydrogens attached to a carbon, which is also connected to the two carbonyl groups. Think of it as a very stable, slightly boring molecule. Utterly unadventurous.
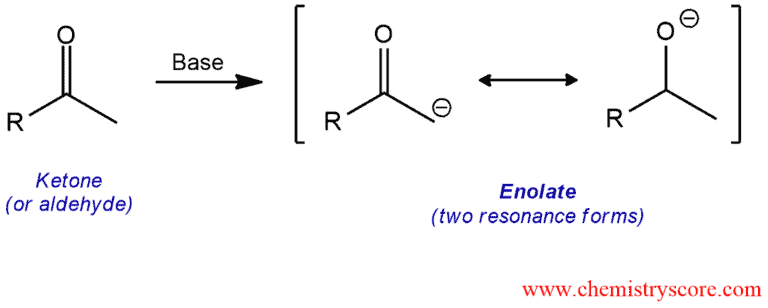
But then, the mischievous base comes along. This base, let's call it a “wingman” for our story, is like that friend who whispers in your ear, "Go on, you can do it! Just be a little… different." This wingman base is strong enough to pry one of those hydrogen atoms right off acetone. Poof!
And what do we get? Not a sad, de-hydrogenated acetone. Oh no! We get something far more exciting: the enolate. This isn't just any old ion; it's a resonating superstar. Imagine a molecule doing a dazzling figure-skating routine, its negative charge sliding back and forth between the oxygen and the carbon. This makes the carbon atom, which used to be all shy and attached, suddenly very, very electron-rich. It's practically begging for something positive to cuddle with.
Enter Benzaldehyde: The Allure of the Electrophilic
Now, let's cast our gaze upon benzaldehyde. This fella is a bit more… refined. It's got a benzene ring, which makes it look all sophisticated and important. But the real magic happens at its carbonyl group (that C=O bit). The oxygen atom, being a bit of a diva, hogs all the electrons. This leaves the carbon atom in the carbonyl group feeling rather electron-deficient. It's like the popular kid at the party who everyone wants to talk to, but who feels a bit overwhelmed.
So, our benzaldehyde carbon is basically wearing a sign that says, "Looking for some positive attention!" It's an electrophile, a lover of electrons, a magnet for anything negative.
The Inevitable Encounter: When Two Worlds Collide
And what happens when our electron-rich, slightly desperate acetone enolate meets the electron-deficient, attention-seeking benzaldehyde? It's a chemical love story for the ages!
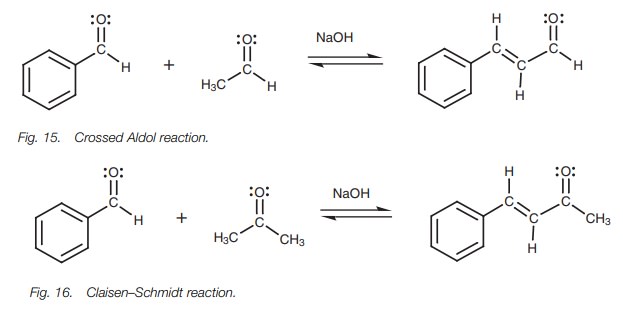
The enolate, with its abundant electrons practically buzzing with excitement, sees the positively-charged carbon of benzaldehyde and thinks, "Well, hello there, handsome!" It’s like a duck spotting a particularly juicy worm. There’s an immediate, undeniable attraction.
The enolate, like a bold suitor, rushes in and attacks the benzaldehyde carbon. This is a nucleophilic attack, where the electron-rich enolate (the nucleophile) gives a generous donation of its electrons to the electron-poor benzaldehyde carbon (the electrophile). It’s a beautiful exchange, a transfer of wealth, a true meeting of minds… or in this case, electron clouds.
The Aftermath: A Not-So-Simple Embrace
This initial attack isn't just a fleeting handshake; it’s a full-on embrace. The carbon-carbon bond forms, creating a brand new, larger molecule. The enolate is no longer just a sassy acetone fragment; it's now intimately connected to the fancy benzaldehyde.
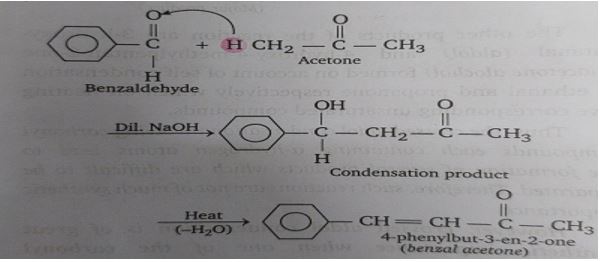
But it doesn't stop there! After this initial union, things get even more interesting. That oxygen that was so selfish with electrons in benzaldehyde? It ends up getting a proton (a little H+ buddy) from somewhere. And the oxygen from the original acetone carbonyl? It also gets a proton.
The result? We get a molecule with a new carbon-carbon bond, and we often end up with a new functional group: an alkoxide, which then gets protonated to form a beta-hydroxy carbonyl compound. It’s like the happy couple is moving in together and starting a beautiful chemical family.
Why THIS Pair? The Magic of Electronegativity and Resonance
So, what makes this specific pairing so special? It’s a dance of forces! Acetone, when it becomes an enolate, is a phenomenal nucleophile. Its negative charge is stabilized by resonance, meaning it’s not some fleeting anger; it’s a well-supported, readily available electron source. Think of it as having a consistently strong handshake.

Benzaldehyde, on the other hand, is a prime electrophile. That benzene ring pulls electron density away from the carbonyl carbon, making it wonderfully susceptible to attack. It’s like a perfectly ripe piece of fruit, just waiting to be plucked.
This isn't a situation where one molecule is trying too hard or the other is playing coy. They are perfectly matched, each providing what the other needs. It’s a chemical soulmate situation, complete with dramatic reunions and the formation of something entirely new and, dare I say, beautiful.
Beyond the Café: The Real-World Yumminess
And the best part? This isn't just some abstract chemical concept confined to dusty textbooks. This fundamental reaction, the aldol reaction, is the backbone of so many incredible things! It's how chemists build complex molecules, like pharmaceuticals, fragrances (that's where the almond smell comes in handy!), and even materials that make our lives better.
So, the next time you catch a whiff of cherry-scented shampoo or admire a beautifully crafted piece of plastic, remember the humble acetone enolate and the charming benzaldehyde, locked in their eternal chemical embrace. It’s a tale of attraction, electron transfer, and the creation of something truly wonderful. Who knew nail polish remover and almond extract could be such good matchmakers?