Why Do Giant Covalent Structures Have High Melting Points
Alright, settle in, grab your latte, because we're about to dive into the wonderfully weird world of giant covalent structures and why they’re basically the culinary titans of the melting point world. You know those moments when you try to melt something, and it’s like, "Nah, man, I'm good"? That's usually our cue that we're dealing with one of these stubborn giants.
Think about it. Most things you try to melt, like, say, a chocolate bar on a hot summer day, surrender pretty quickly. They go from solid to goo faster than you can say "brain freeze." But then you’ve got stuff like diamond, graphite, or even sand (which is basically silicon dioxide, fancy name for that gritty stuff), and they’re like, "You want me to melt? You melt first, buddy!"
So, what’s the deal? Why are these guys so incredibly, ridiculously, stubbornly resistant to becoming a liquid puddle? It all comes down to something called a giant covalent structure. Now, "giant" is a bit of an understatement. We're talking about structures that are essentially one massive, uninterrupted network of atoms, all happily (or maybe not-so-happily, depending on their perspective) bonded together. It's like one giant, interconnected family reunion that never, ever ends.
Must Read
Imagine trying to break apart a huge, really old family. Not just a few cousins, but like, the entire clan, from great-great-grandparents to the newest arrival, all linked together by unbreakable vows (or in this case, covalent bonds). You'd need some serious muscle, right? And that, my friends, is precisely what’s happening at the atomic level.
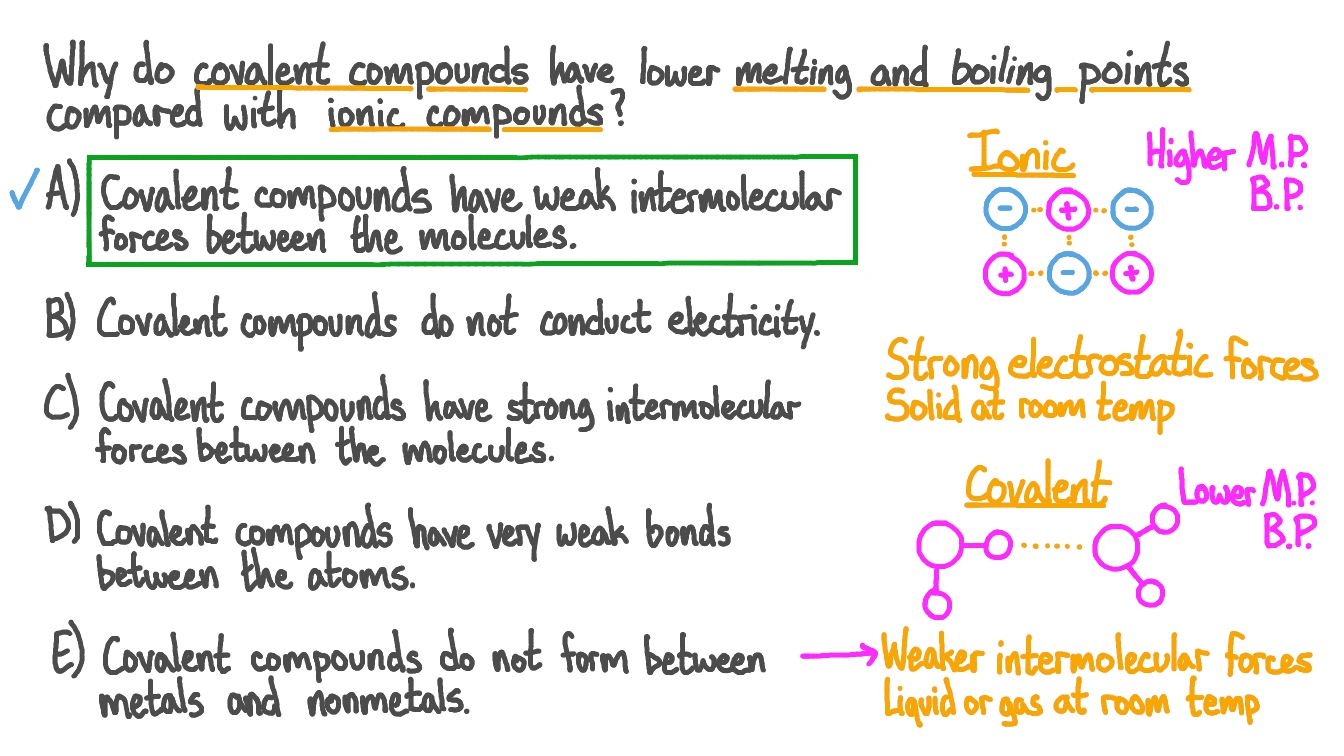
In a typical simple molecular compound, like water (H₂O) or sugar, the molecules are like little individual cars on a highway. They're bonded to each other, sure, but the forces holding these cars together (intermolecular forces, for the science nerds in the back) are relatively weak. A little bit of heat, and bam! The cars start zipping around independently, and you’ve got yourself a liquid.

But with giant covalent structures, it’s not like individual cars. It’s more like one ginormous monster truck made of Lego bricks. Every single Lego brick (atom) is firmly attached to its neighbors with incredibly strong chemical bonds. We’re talking about covalent bonds, those super-strong partnerships where atoms actually share electrons. It’s like they’re holding hands, not letting go, no matter what.
These covalent bonds are the superglue of the atomic world. They require an enormous amount of energy to break. When you try to melt a giant covalent structure, you’re not just trying to get the molecules to slide past each other. Oh no. You’re trying to break those incredibly strong covalent bonds that hold the entire network together. It's like trying to pull apart a concrete wall brick by brick using only your fingernails. Good luck with that!
Let’s take diamond as our star example. Diamond is pure carbon, and each carbon atom is bonded to four other carbon atoms in a tetrahedral arrangement. It’s a 3D lattice, like a super-strong, perfectly constructed crystal. It’s so strong, in fact, that it's used in drill bits and saw blades. If it's tough enough to cut through rock, you better believe it's tough enough to resist your measly Bunsen burner.

The melting point of diamond is around 3,550°C (6,422°F). To put that in perspective, that's hotter than the surface of many stars! You could probably boil the ocean multiple times over before you even get a tiny speck of diamond to change its state. It’s like telling a brick wall it's time for a spa day and expecting it to relax.
Then there’s graphite, also made of carbon, but with a different arrangement. It’s like a layered cake, where each layer is a sheet of carbon atoms arranged in hexagons. These layers are held together by weaker forces than the covalent bonds within the layers. This is why graphite can scratch things and is used as pencil lead – the layers slide off easily. But to melt the entire graphite structure, you’d still need to break those strong covalent bonds within each layer, which is still a monumental task. Its melting point is around 3,652°C (6,606°F) – even hotter than diamond, which is a bit of a plot twist!

Think of it like this: imagine you have a bunch of tightly knit families living in interconnected houses. If the houses are made of really strong, glued-together bricks (covalent bonds within the structure), it’s going to take a lot of effort to knock down even one house, let alone the whole neighborhood. You need to apply a tremendous amount of heat energy to provide enough "oomph" to overcome the strength of those bonds.
It's this continuous, interconnected network of strong covalent bonds that gives these materials their incredible stability and, consequently, their sky-high melting points. There are no weak points, no easily breakable links. It’s all or nothing, and "nothing" is usually the outcome when you’re just trying to get something to melt for your science experiment.
So, next time you’re marveling at a diamond’s sparkle or using a pencil, remember the epic battle happening at the atomic level. It’s a testament to the sheer power of covalent bonding, and a rather amusing reminder that some things just refuse to be easily persuaded to change their state. They’re the ultimate stubborn old timers of the chemical world, and we can only admire their unwavering resolve. Now, who wants another coffee? Because I’m guessing you’re not melting this café down any time soon!



