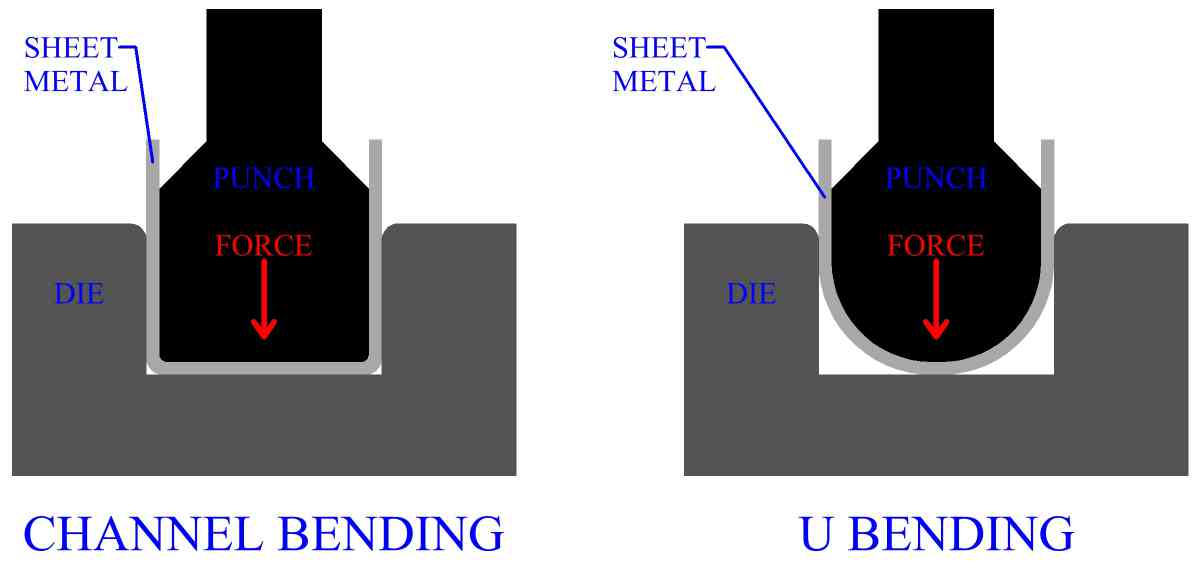
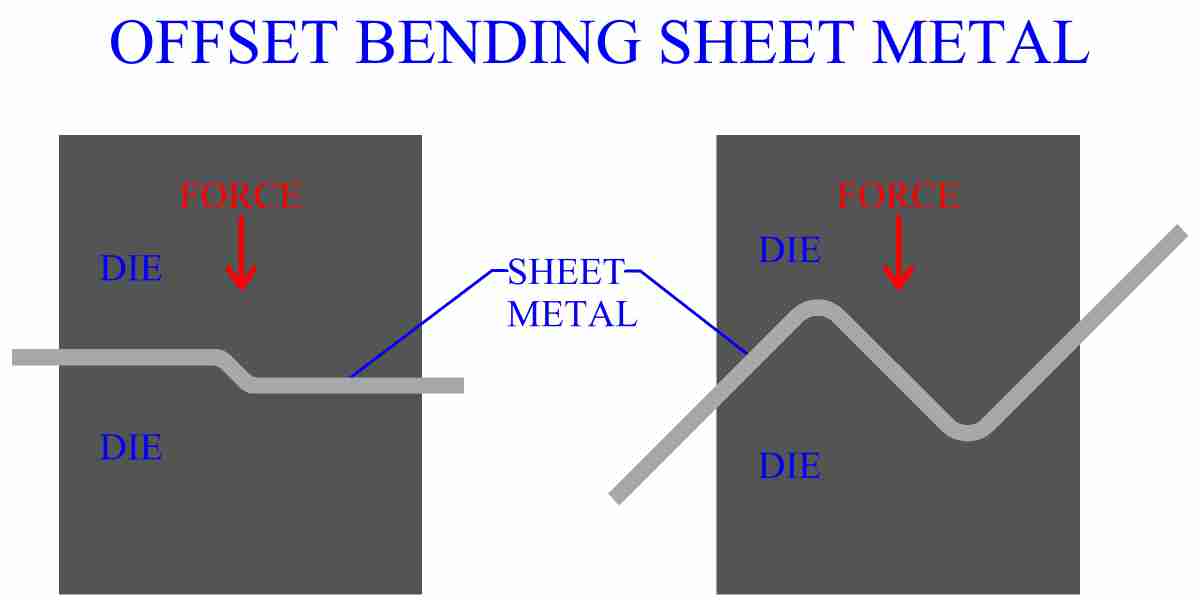
Why Can Metals Be Bent And Shaped

Ever wondered how a delicate piece of jewelry gets its intricate curves, or how a sturdy bridge can gracefully span a vast distance? It all comes down to a fascinating property of metals: their ability to be bent and shaped. It's a concept that touches everything from the tools we use to the art we admire, and understanding it can spark a whole new appreciation for the world around us. Think of it as unlocking a secret superpower of everyday objects!
So, why can metals be so easily molded? The secret lies deep within their atomic structure. Unlike brittle materials like glass, which break when pushed too far, metals have a unique arrangement of atoms. Imagine tiny, tightly packed spheres. In metals, these spheres are held together by what we call a "sea of electrons." These electrons aren't tied to any specific atom but can move freely between them. This is the key!
When you apply pressure to a metal, instead of snapping, these atoms can actually slide past each other. The "sea of electrons" acts like a lubricant, allowing the atomic layers to shift and rearrange without breaking the bonds completely. It's like rearranging a deck of cards – you can slide them around without tearing them. This characteristic is known as ductility and malleability. Ductility refers to a metal's ability to be drawn into wires, while malleability is its ability to be hammered or rolled into thin sheets.
Must Read
The practical implications of this property are absolutely enormous. Think about everyday items: the aluminum foil you use to wrap leftovers, the copper wires that power your electronics, the steel beams that form the skeleton of skyscrapers, and even the horseshoe on a horse's hoof. All of these owe their existence to the bendability and shapability of metals. In education, this principle is a cornerstone of physics and materials science. Students learn about crystal structures, atomic bonding, and the forces that govern the behavior of matter. It’s a fundamental concept that bridges abstract scientific ideas with tangible, real-world applications.

Exploring this further is easier than you might think. You can observe it directly with simple materials. Take a paperclip, for instance. Try bending it back and forth repeatedly. You'll notice it becomes easier to bend in the same spot, and eventually, it will break. This is because repeated bending causes the atomic layers to shift and lock in place, making further movement difficult. Or, consider a piece of aluminum foil. You can crumple it, fold it, and reshape it countless times. You might also notice how different metals behave differently. A piece of soft copper wire is far easier to bend than a hardened steel nail.
For a slightly more involved exploration, you could look at how blacksmiths have shaped metal for centuries. Watching videos of them hammering and molding glowing-hot iron demonstrates these principles in action. They use heat to make the metal more pliable, allowing them to exploit its natural tendency to bend and shape. So, the next time you pick up a metal object, take a moment to appreciate the incredible science that allows it to be so versatile. It’s a testament to the elegant simplicity and remarkable power hidden within the atomic world.