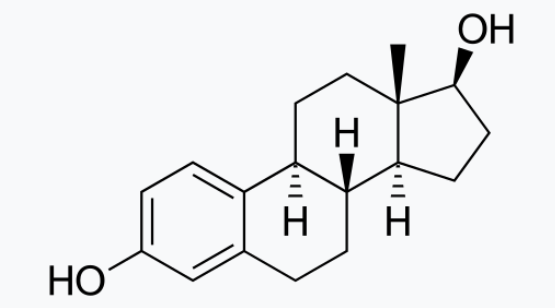
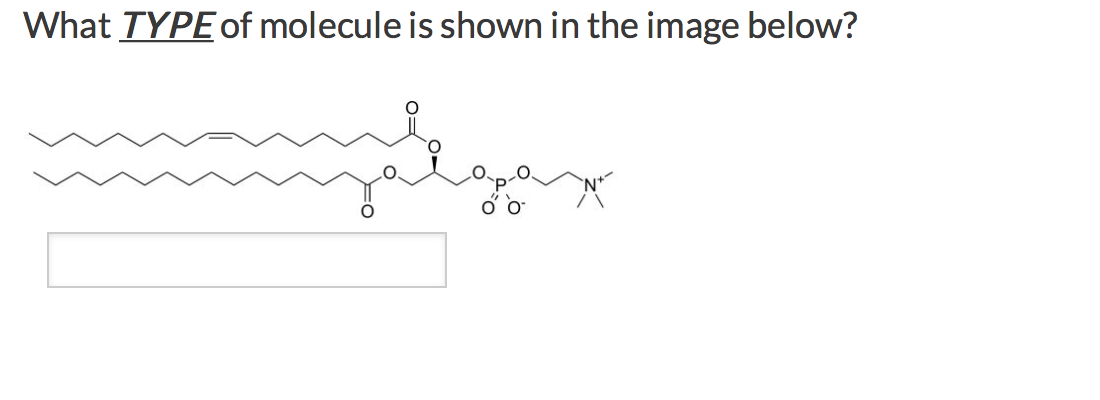
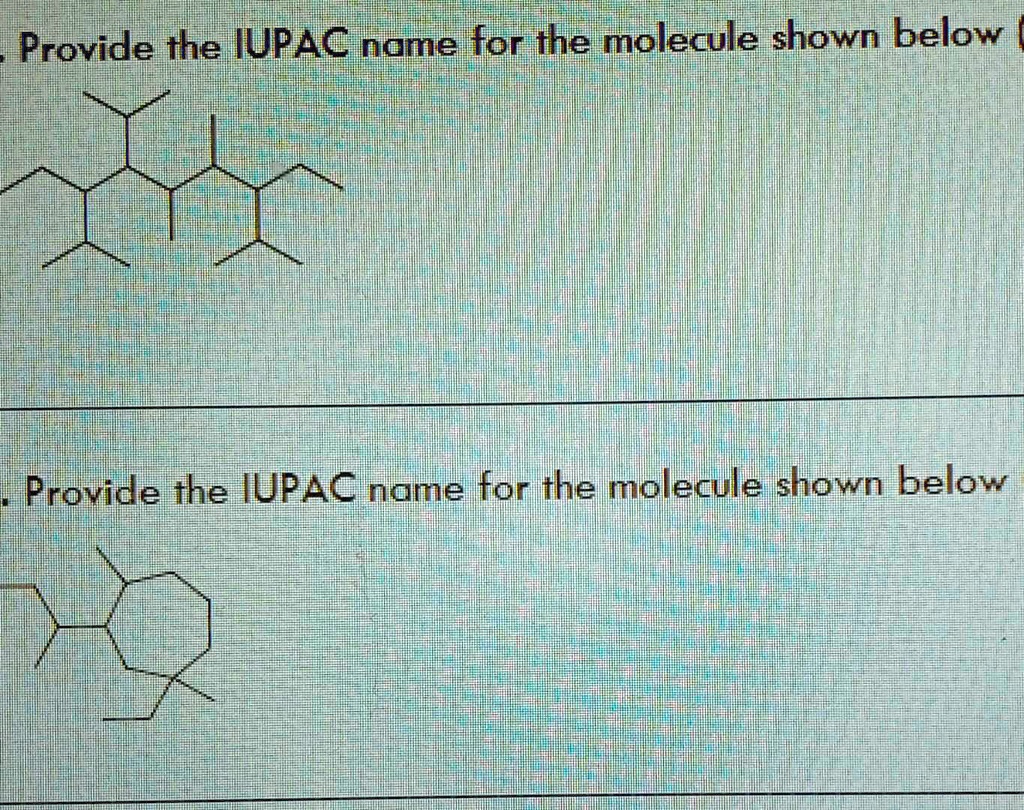
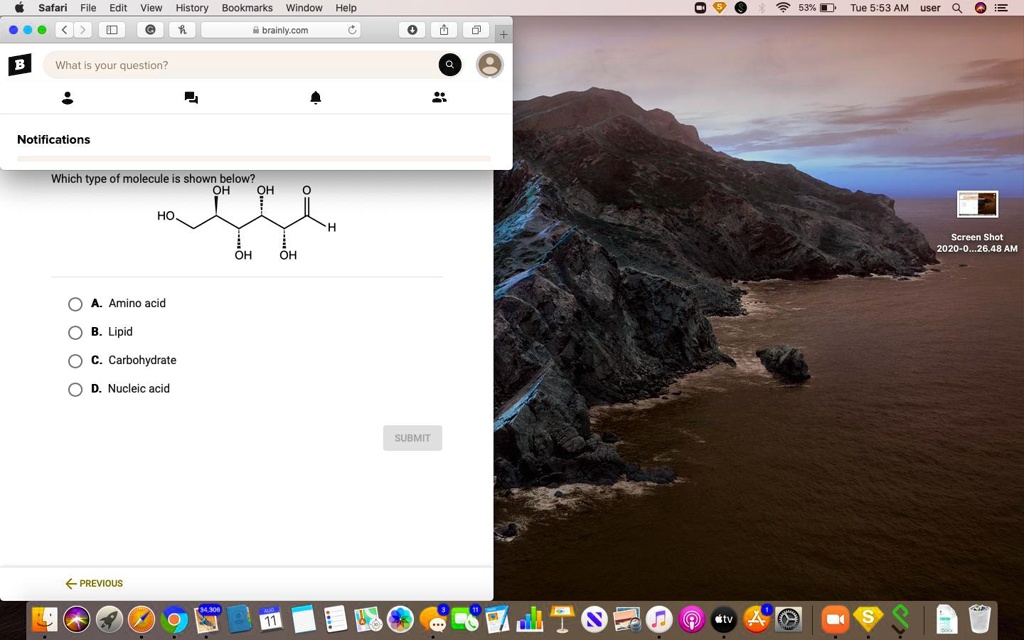
Which Type Of Molecule Is Shown Below Apex
Hey there! So, we're diving into the wonderfully weird world of molecules today, and I've got a little puzzle for you. Imagine we're just chilling, maybe with some coffee or tea, and I slide this picture across the table. It's a molecule, right? But what kind of molecule is it? Big question, I know! It's like asking, "What's for dinner?" when you've got a whole pantry staring you down. But fear not, we're gonna break it down, piece by piece, and figure this thing out together. Think of me as your friendly neighborhood molecule whisperer.
So, you're looking at this thing. What's the first thing that pops into your head? Does it look, I dunno, important? Like it's got a job to do? Because, let me tell you, every molecule, even the little shy ones, has a purpose. It's their whole raison d'être, you know? They're not just sitting around looking pretty. They're out there, doing their molecular thang. And this one, this particular specimen we're eyeballin', is definitely got some serious business going on. It's not some flimsy, easily-distracted molecule. Oh no. This one is built for... well, for whatever it's built for!
Let's get down to the nitty-gritty, shall we? When we're talking about molecule types, we're basically categorizing them based on what they're made of and how they're put together. It's like sorting your LEGOs – you've got the basic bricks, the wheels, the little minifigures, right? Well, molecules are kinda like that, but way more complex and, dare I say, infinitely cooler. And this one here? It's giving off some serious vibes. It's not your average, everyday, "oh, that's just water" kind of molecule. This one has character.
Must Read
Now, before we get too deep, let's talk about what we're even seeing. We've got these little spheres, right? And they're all connected by sticks. Super simple visual, but oh boy, it's packing a lot of information. Each sphere represents an atom. Think of them as the building blocks. And the sticks? Those are the bonds, the connections that hold the atoms together. It's like a molecular handshake, but way more permanent. These bonds are the glue that keeps the whole party from falling apart. And trust me, in the molecular world, things can get pretty wild and woolly.
So, what kind of atoms are we dealing with here? That's the first clue, the big opener in our molecular mystery. We're not seeing a whole lot of variety in terms of the types of spheres, which is already telling us something. If we had a whole rainbow of different colored spheres, we'd be looking at a much more diverse and, frankly, chaotic molecule. But this one? It seems to be keeping things relatively consistent. This suggests a certain level of simplicity in its construction, at least from an elemental perspective.
And the way these atoms are arranged, the shape it's taken on, that's another huge giveaway. Molecules aren't just randomly tossed together; they have specific structures. Think of a house. You can have the same number of bricks, but you can build a shed or a mansion, right? It's all about the arrangement. And this molecule? It's not a flat, boring disc. It has some depth, some dimensionality. It's got a bit of a three-dimensional swagger to it.
Now, let's consider the typical suspects in the molecular world. We've got your inorganic molecules, which are usually things like salts and acids, and then we've got your organic molecules. And this one, the one we're staring at, is screaming "organic!" Why? Well, for starters, organic molecules are basically the backbone of life. They're the ones that make up your body, your food, the very air you breathe (well, not all of it, but a good chunk!). And they almost always contain carbon. Lots and lots of carbon.
See those little spheres? If they represent carbon atoms, and they seem to be the primary players here, then we're definitely leaning towards the organic side of the fence. Carbon is like the ultimate social butterfly of the atomic world. It can bond with itself in all sorts of amazing ways, forming long chains, rings, and all sorts of intricate structures. It's the master architect of organic chemistry. Without carbon, life as we know it? Not happening. It's a pretty big deal, honestly.
And if it's organic, what kind of organic molecule are we looking at? There are tons of categories, of course. We've got carbohydrates, lipids, proteins, nucleic acids. That's like the A-list of organic molecules. They're the ones doing all the heavy lifting in biological systems. But there are also smaller, simpler organic molecules that act as building blocks or important intermediaries.
Let's look closer at the structure. Do you see any repeating units? Sometimes molecules look like little train cars hooked up together. That can be a sign of a polymer, which is just a long chain of repeating smaller molecules. But this one doesn't immediately scream "long repeating chain" to me. It looks a bit more contained, more like a distinct unit.
What about those little spheres that aren't carbon? If we're assuming it's organic, then we're likely to see some hydrogen atoms, maybe some oxygen, nitrogen, or sulfur. These other elements are like the supporting cast, adding different properties and functionalities to the molecule. They can change its reactivity, its solubility, its overall behavior. It's like adding spices to a dish; they completely transform the flavor.
Now, the question is, what's the specific type? Without labels on the spheres, it's a bit of a guessing game, but we can make educated guesses based on common molecular structures. If you're seeing a lot of carbon atoms linked together in a ring or a straight chain, and then some hydrogens attached, you're probably looking at a hydrocarbon. That's the simplest form of organic molecule, really. Just carbon and hydrogen. Think of fuels like methane or propane. They're hydrocarbons.
But if there are other atoms involved, like oxygen, we might be looking at something like an alcohol, which has a hydroxyl group (-OH). Or a carboxylic acid, with a carboxyl group (-COOH). These little functional groups, as chemists call them, are like the molecule's personality traits. They dictate how it interacts with the world. It's like giving someone a quirky habit or a specific talent; it defines them.
Let's zoom in (metaphorically, of course, since we can't actually zoom in on a static image!). Do you see any double bonds? Those are where two pairs of electrons are shared between atoms, making the bond stronger and the molecule behave a little differently. Single bonds are like a casual nod; double bonds are more like a firm handshake, and triple bonds are like a full-on molecular hug. They add a whole other layer of complexity and reactivity.
The arrangement also matters. Is it a linear molecule, all stretched out in a line? Or is it branched, with side chains coming off the main structure? Or is it cyclic, forming a ring? Each of these arrangements leads to different properties. A ring structure can be quite stable, while a branched structure might be more reactive. It's like comparing a tightly wound ball of yarn to a loose, sprawling spiderweb. Both are made of string, but they're very different.

So, if this molecule is predominantly carbon and hydrogen, and it's got a relatively simple, non-repeating structure, and it doesn't have any particularly complex functional groups that we can readily identify just by looking at it, then it's highly likely to be a hydrocarbon. They are the fundamental building blocks of so many other organic molecules. They're the unsung heroes of organic chemistry.
But what if it's a bit more complex? What if it has oxygen atoms in there? If you see a carbon atom double-bonded to an oxygen atom, and then also single-bonded to another atom (which could be another carbon, or an oxygen, or even a hydrogen), you might be looking at a carbonyl group. This is a key feature in two important classes of organic molecules: aldehydes and ketones. They're like cousins in the organic family tree.
If that carbonyl group is attached to a hydroxyl group, then you've got a carboxylic acid. And these guys are, well, acidic! They can donate a proton, which is a big deal in chemical reactions. They're the ones that can make things a bit sour, like vinegar.
Now, let's think about the size. Is this a small molecule, something you can hold in your hand (if it were a physical object, of course)? Or is it a giant macromolecule, something that would stretch for miles if you laid it out? Based on the representation, it looks like it's not an outrageously huge molecule. It's not a whole protein or a DNA strand. It seems to be a more manageable size, a distinct, self-contained unit.
So, if we're sticking with the idea that it's organic, made primarily of carbon and hydrogen, and it's a relatively small, distinct unit, then we're likely looking at something that falls under the broader umbrella of a simple organic molecule. This could be anything from a simple alkane to an alkene, or perhaps even a cyclic hydrocarbon.
The crucial thing to remember is that the specific arrangement and types of atoms dictate the molecule's identity. It's like a secret code, and chemists are the ones who can crack it. They look at the patterns, the connections, and the elemental makeup, and they can tell you exactly what the molecule is and what it does. It's pretty mind-blowing when you think about it.
Without more specific information, like labels on the atoms or context about its origin or function, pinpointing the exact type of molecule can be tricky. It's like trying to identify a person from a blurry silhouette. You can get a general idea, but the fine details are missing. However, based on common visual representations and the general appearance of being composed of interconnected spheres, and assuming it's not an inorganic salt or ion, the most likely broad category is indeed an organic molecule.
And if we were to refine that further, looking for common patterns in such simplified diagrams, it often represents a hydrocarbon or a simple derivative thereof. Think of it as the foundation upon which more complex organic structures are built. These are the workhorses of organic chemistry, the ones that make all the other fancy stuff possible. So, while we might not be able to name it definitively as, say, "benzene" or "ethanol" without more clues, we can confidently say it's in the organic family. It's got that distinct carbon-based backbone that screams "organic!" loud and clear.
So, there you have it! A little journey into the world of molecular identification. It's all about looking at the building blocks, how they're connected, and the overall structure. This particular molecule, with its interconnected spheres, is definitely sporting an organic vibe. It's not something you'd find in a rock, for example. It’s got that life-giving, carbon-based signature. And that, my friends, is a pretty darn good starting point for understanding what type of molecule we're looking at! Pretty cool, right? Now, about that coffee...


![[Solved]: What is the molecule shown below (note that lone p](https://media.cheggcdn.com/media/26e/26eba3f6-eca6-4f28-91db-57567be3cdaf/IMG_0963.png)
![[ANSWERED] 66 2 points In the molecule shown below atom X is most - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20220518205927802635-3645535.jpg?h=512)
![[GET ANSWER] Check the box under each structure in the table that is an](https://cdn.numerade.com/ask_images/8bd0b554f48e44948cf1ba00bafee6a7.jpg)