Which Statement Best Describes An Electron
Hey there, science curious friends! Ever wondered about the tiny, zippy things that make up, well, everything? Today, we're diving into the wild world of electrons. Think of them as the universe's ultimate party starters. They're everywhere! Seriously, you're probably made of billions of them right now. Mind. Blown.
So, which statement best describes this elusive little critter? It's a bit like asking to describe a really hyper squirrel. They're not just one thing, are they? They're a whole bunch of amazing, slightly baffling characteristics rolled into one. Let's break it down, shall we?
Electrons: The Tiny Titans of the Atomic World
First off, electrons are super small. Like, ridiculously small. Imagine taking a grain of sand and then making it even tinier. And then tinier again. We're talking way, way, way down there. They're so small, in fact, that we can't really "see" them in the way you see a bouncy ball. It's more like we infer their presence, like noticing a ghost's handprint on a dusty table.
Must Read
And they're negatively charged. This is a biggie. Think of positive and negative like best frenemies. They're attracted to each other. Protons, for instance, are their positively charged buddies in the center of an atom. Electrons orbit around them, kinda like planets around a sun, but much more chaotic and less predictable.
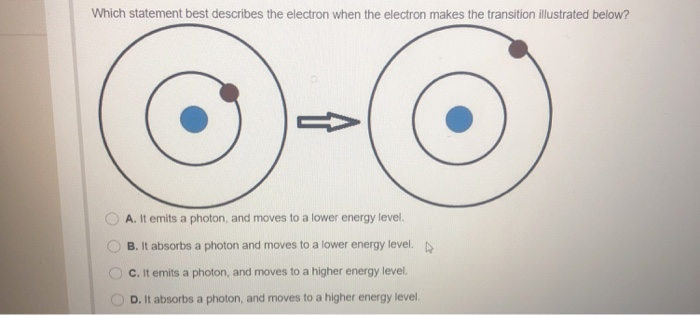
Speaking of orbits, here's where things get deliciously weird. Electrons don't just zip around in neat little circles. Oh no. They exist in these fuzzy regions called orbitals. Imagine a cloud of probability. The electron is somewhere in that cloud, but pinpointing its exact location at any given moment is a fun game of "where's Waldo?" for physicists.
The Quantum Conundrum: Not Your Average Ball
This is where the fun really kicks in! Electrons are quantum particles. What does that even mean? Well, it means they don't always behave like tiny little marbles. Sometimes they act like waves! Yep, they can be a particle and a wave at the same time. It's called wave-particle duality, and it's one of those mind-bending concepts that makes physics so darn cool.
Think about it. If you throw a ball, it's a ball. If you send a ripple across water, it's a wave. An electron can be both! It's like a magician who can turn into a puff of smoke and then solidify back into a rabbit. Except, you know, with less top hats and more abstract mathematical equations.
And don't even get me started on uncertainty. Heisenberg's Uncertainty Principle tells us we can't know both the exact position and the exact momentum of an electron at the same time. The more precisely you know where it is, the less you know about how fast it's moving, and vice versa. It's a cosmic game of "guess who?"
Electrons: The Movers and Shakers of Chemistry
But here's the truly exciting part for us everyday humans: electrons are the secret sauce of chemistry. They're the ones who get together, form bonds, and create all the molecules that make up everything you see, touch, and taste. Without electrons, there'd be no water, no air, no chocolate chip cookies (and that, my friends, is a true tragedy).
When atoms want to hang out, it's usually the electrons doing the talking. They're passed around, shared, and grabbed in all sorts of chemical reactions. Think of them as the social butterflies of the atomic world, always mingling and making connections.
They're responsible for electricity, too! When electrons get moving in a directed flow, that's electricity zipping through wires. So, that light bulb that just turned on? Thank the electrons for their energetic dash.
A Quirky Little Detail: Spin!
And as if being a wave, a particle, and an elusive quantum object wasn't enough, electrons also have this thing called spin. It's not like they're physically spinning like a top, but they have an intrinsic angular momentum. They can have "spin up" or "spin down." It's another one of those quirky properties that make them so fascinating.
Imagine two electrons in the same orbital. They have to have opposite spins. It's like a cosmic rule, the Pauli Exclusion Principle, saying "one must be up, and one must be down!" It's a bit like a dance, where partners always have to face opposite directions. This rule is fundamental to how atoms are structured and, therefore, how matter itself is organized.
So, What Statement Truly Captures an Electron?
Given all this, which statement best describes an electron? It's definitely not just "a tiny thing." It's a whole lot more!
A good statement would probably be something along the lines of: "An electron is a fundamental, negatively charged subatomic particle that exhibits wave-particle duality, exists in probabilistic orbitals, and is the primary driver of chemical bonding and electrical phenomena."
But that's a bit of a mouthful, right? For a more fun, casual vibe, we could say:

"It's the universe's most energetic and indecisive party animal. It's a tiny negatively charged bit of 'stuff' that's sometimes a particle, sometimes a wave, and always up for a chemical reaction!"
Or even better:
"It's the ultimate multitasker! This super-tiny, negatively charged particle is responsible for everything from electricity to the bonds that hold your coffee mug together, all while existing in a fuzzy, uncertain state that makes physicists scratch their heads (in a good way!)."
The beauty of electrons is that they challenge our everyday intuition. They force us to think differently about reality. They're not just building blocks; they're the dynamic, playful forces that bring the universe to life. So next time you flick a light switch or admire a beautiful crystal, take a moment to appreciate the incredible, quirky dance of the electrons!