Which Statement Best Describes A Compound

Hey there, my fellow curious minds! Ever found yourself staring at a science textbook, feeling like you're deciphering ancient hieroglyphs written by a very confused alien? Yeah, me too. Today, we're diving into the wonderful world of chemistry, and specifically, we're going to tackle a question that might sound a tad intimidating but is actually super straightforward: Which statement best describes a compound? Don't worry, we're not going to get bogged down in a million complex formulas. Think of this as a friendly chat, maybe over some (hypothetical, sadly) cookies and milk, where we unravel this mystery together.
So, what is this mysterious "compound" we keep hearing about? Imagine you have two or more ingredients, right? Like, salt and pepper for your fries (don't judge my taste!). Or maybe flour and sugar for some delicious baking. These ingredients are distinct on their own. You can still see the grains of salt, and you can definitely tell the difference between the fluffy flour and the sparkly sugar. They're like roommates, hanging out in the same bowl, but still very much themselves. That's sort of how elements work in chemistry. You've got your good ol' building blocks like Hydrogen (H), Oxygen (O), or Carbon (C). They're the OG's, the single ingredients.
Now, here's where things get interesting. What happens when these elements decide to get really close? Like, hold-hands-forever, can't-be-separated-without-a-chemical-reaction close? That, my friends, is a compound! It's not just a mixture where things are hanging out together; it's a whole new substance formed when two or more different elements chemically bond together in a fixed ratio. Think of it like baking. You can't just throw flour and sugar into a bowl and call it a cake. You gotta mix 'em, bake 'em, and when you're done, you've got something entirely new and delicious. That cake is your compound!
Must Read
Let's break this down a bit more. When elements form a compound, they lose their individual identities, in a way. They combine in a specific, predictable way. It's like Romeo and Juliet, but with atoms. They unite, and the result is a brand new entity with completely different properties. Salt, for example. You know, that yummy stuff that makes your popcorn sing? That's a compound called sodium chloride. It's made from sodium (Na), which is a highly reactive metal that can explode in water (yikes!), and chlorine (Cl), which is a poisonous gas (double yikes!). Pretty scary stuff on their own, right? But when they get together and form sodium chloride, they become something completely safe and essential for life. Mind. Blown.
So, if we were to look at a statement that best describes a compound, what would it be? It's got to capture that idea of elements combining chemically to form something new. It's not just about them being in the same place; it's about them forming a strong, unbreakable bond (until a chemical reaction breaks it, of course – nothing is that permanent, except maybe my craving for pizza).

Let's consider some options, like little quiz questions designed to make you think. Imagine you see these statements:
Option 1: "A substance made of two or more different elements physically mixed together."
Hmm, "physically mixed." This sounds like our cookie dough before it goes into the oven. You can still see the chocolate chips, the flour, the sugar. They're together, but they haven't fundamentally changed. This describes a mixture, not a compound. So, this statement is a big fat NOPE for compounds. It's like saying your homework is a compound because all the papers are in one folder. Nope, still just individual papers.
Option 2: "A substance made of only one type of atom."
This sounds familiar! We talked about this. These are your elements. Gold is an element, made only of gold atoms. Helium is an element, made only of helium atoms. They're the pure, unadulterated building blocks. So, this statement is also a miss for our compound quest. It's like saying a single chocolate chip is a compound. It's just a chocolate chip!

Option 3: "A substance made of two or more different elements chemically bonded together in a fixed ratio."
Ooh, this one is looking good! "Chemically bonded" – that's the key phrase! It tells us these elements aren't just chilling together; they've formed a strong connection. "In a fixed ratio" – that's important too. It means you won't find water molecules with three oxygen atoms and one hydrogen atom (that would be H₂O₃, which sounds… bubbly and probably not in a good way). Water is always H₂O, two hydrogens and one oxygen. This consistency is a hallmark of compounds. This statement? It's hitting all the right notes. It's like saying a perfectly baked cake is a compound – the ingredients are all bound up and transformed into something new and wonderful!
Let's think of another example. Water (H₂O). We know it's made of hydrogen (H) and oxygen (O). Are they just hanging out? Nope! They're chemically bonded. And it's always a 2:1 ratio of hydrogen to oxygen. Try to imagine trying to separate those hydrogens from that oxygen without a chemical reaction – it’s not going to happen easily. You can't just strain them apart like spaghetti. You'd need a special process, like electrolysis, to break those bonds.

Consider carbon dioxide (CO₂), the stuff we breathe out (and that plants are so fond of). It's made of carbon (C) and oxygen (O). They're chemically bonded. And it’s always one carbon atom for every two oxygen atoms. If you tried to just physically mix carbon and oxygen, you’d just have… well, carbon and oxygen floating around. But when they bond chemically, BAM! You've got CO₂, a gas with totally different properties.
So, the statement that best describes a compound is definitely the one that highlights the chemical bonding and the fixed ratio of different elements. It's this chemical union that creates something with novel properties, something distinct from its original components.
Think about it this way: if you're making lemonade, you're mixing lemon juice, water, and sugar. That's a mixture. You can still taste the tartness of the lemon, the sweetness of the sugar, and the coolness of the water. They're all there, coexisting. But if you were to somehow chemically combine hydrogen and oxygen in that fixed 2:1 ratio, you wouldn't get something that tastes like lemon or sugar. You'd get water. Entirely new properties, entirely new substance. Cool, huh?
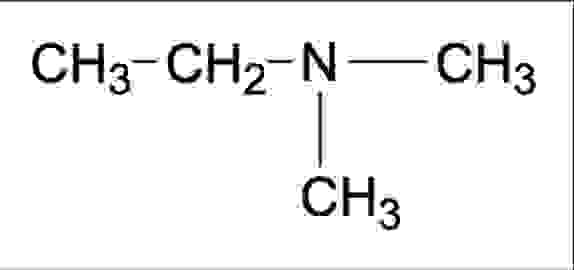
It's really this transformation that makes compounds so fascinating. They're like the ultimate alchemical transformation, but it’s real science! The elements come together, lose their individual quirks, and create a brand new entity with its own unique personality. It’s the chemistry equivalent of a superhero team-up, where the individual powers combine to create something even more powerful and distinct.
So, when you're faced with a question about compounds, just remember those key words: elements, chemically bonded, and fixed ratio. If a statement includes those ideas, chances are you've found your winner! It’s about more than just being together; it’s about becoming one, in a chemical kind of way.
And here's the truly awesome part. The universe is built on these fundamental principles. Every breath you take, every bite you eat, every star you gaze at – it's all a spectacular dance of elements forming compounds. From the simplest water molecule to the incredibly complex molecules that make up life itself, compounds are the unsung heroes of the cosmos. So, the next time you hear the word "compound," don't be intimidated. Think of it as the universe's way of creating new and exciting things from its basic building blocks. You’ve totally got this! Go forth and conquer the world of chemistry, one compound at a time!