Which Phrase Describes A Valence Electron
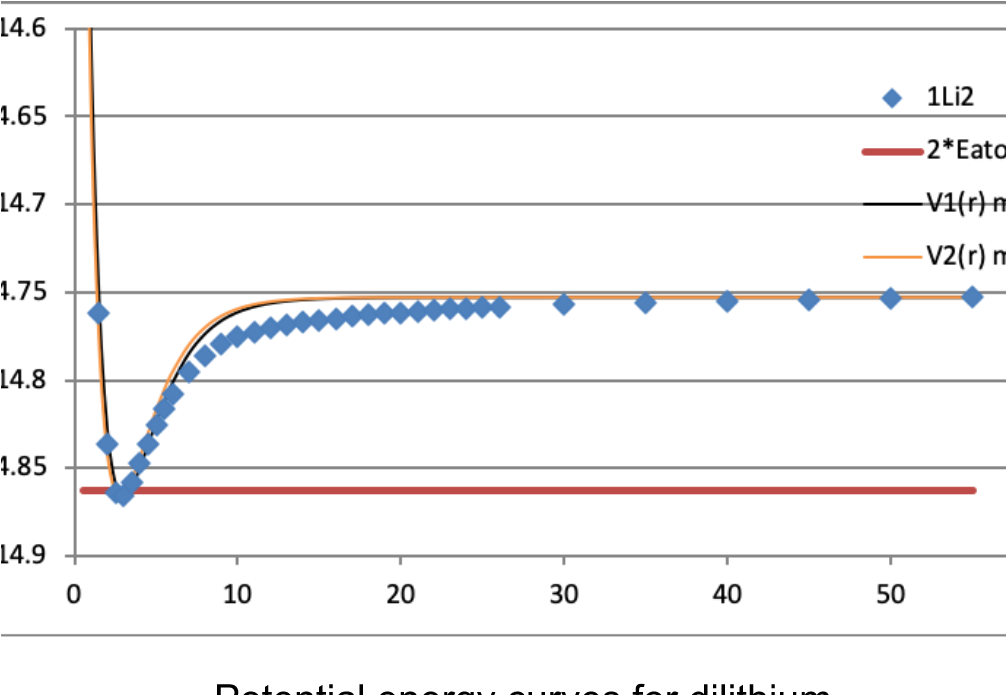
Have you ever wondered what makes things, well, things? Like why water is wet and fire is hot? It all boils down to tiny, zippy little characters dancing around inside atoms. And there's one special kind of these characters that gets all the attention.
These are the valence electrons. They're like the rock stars of the atomic world, the ones who get to go out and play. They’re the ones making all the connections, the ones that really shape how atoms interact. It’s where all the exciting chemistry happens!
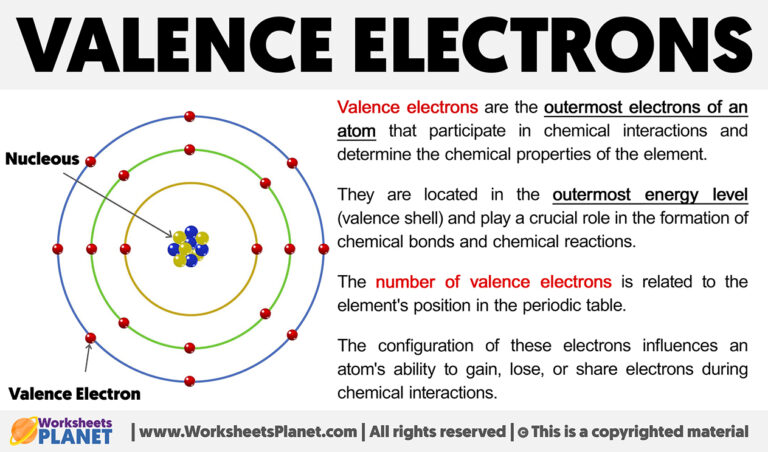
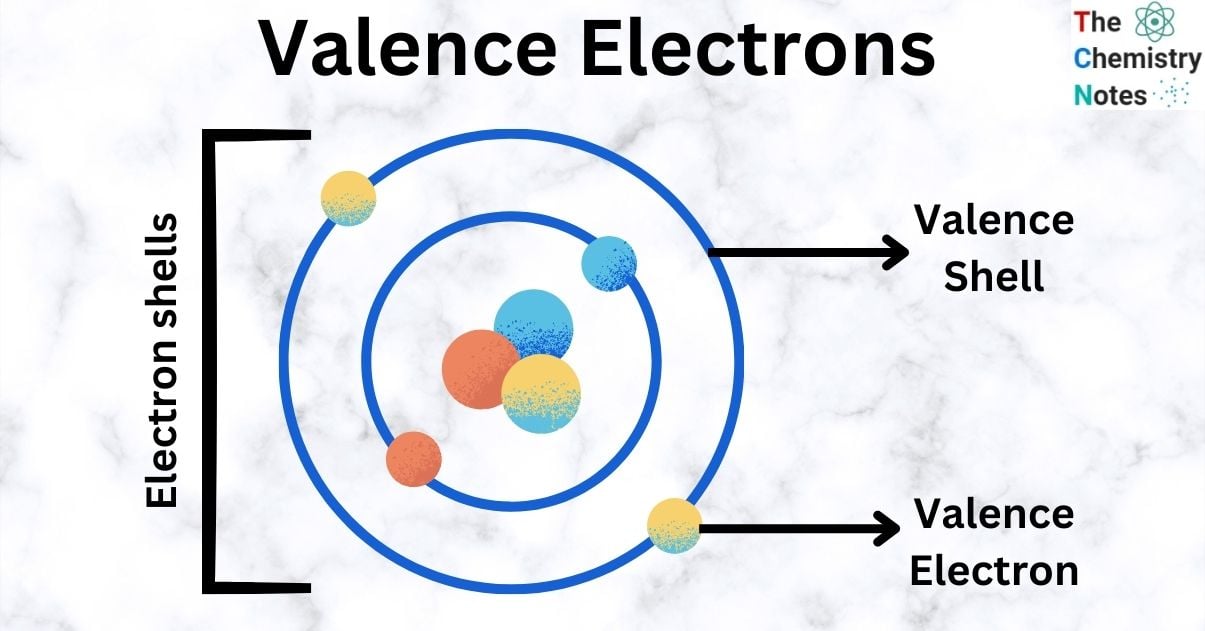
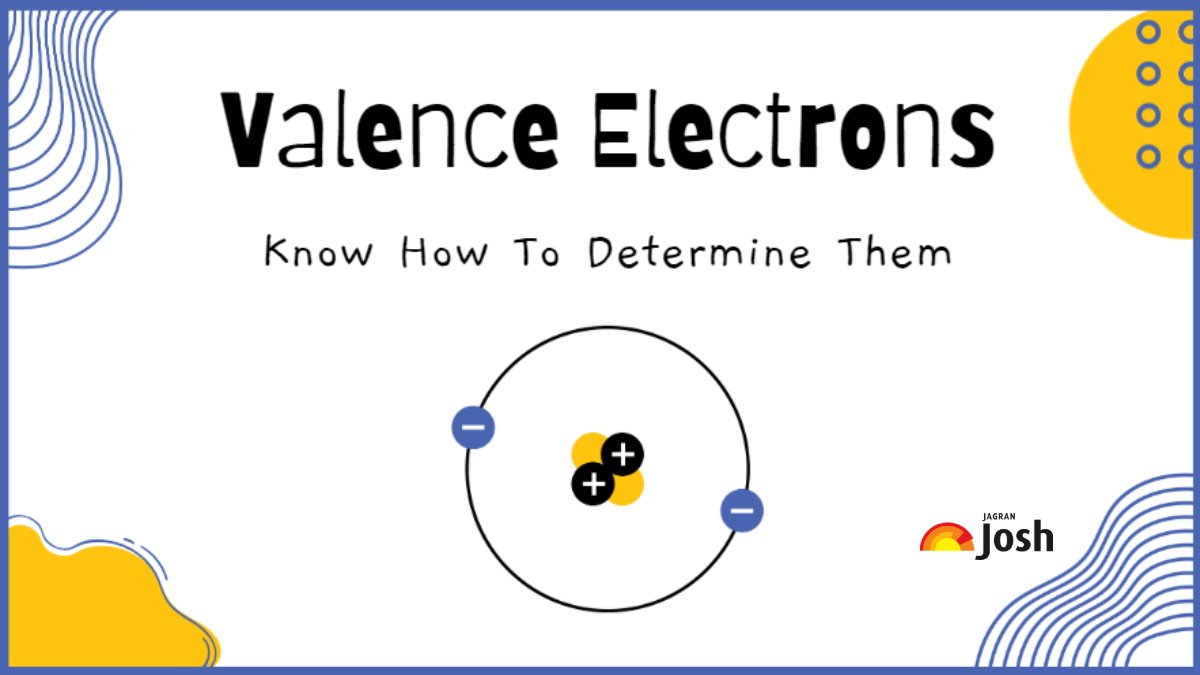
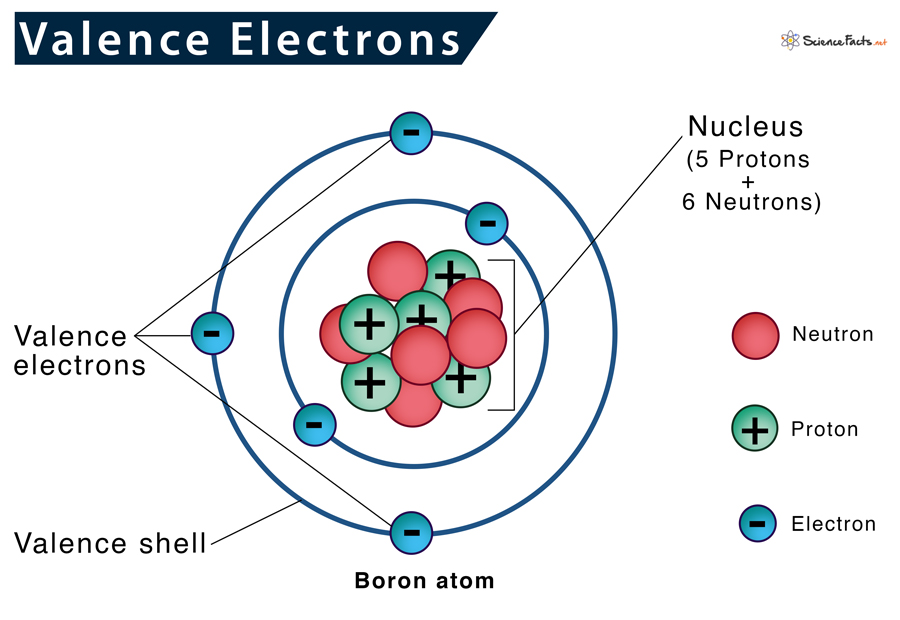
Imagine an atom is like a cozy little house. It has different rooms, and the electrons live in these rooms, which we call shells. The electrons closest to the center are like the ones living in the basement – nice and safe, but not much going on.
Must Read
But the valence electrons? They live in the outermost rooms, the ones right by the front door. They're the ones who can easily peek outside, interact with neighbors, and make new friends. This is what makes them so incredibly special and important.
So, which phrase really nails what a valence electron is all about? Think about it. What do these outer electrons do? They are the ones involved in the action, the ones that allow atoms to stick together. They are the key players in forming all sorts of amazing stuff.
It’s like a cosmic handshake. The valence electrons are the hands that reach out. They’re the ones that allow atoms to bond, to join forces, and to create everything we see and touch. This ability to connect is their superpower.
If you had to pick just one phrase, it would likely highlight their position and their job. They are on the outside, ready for action. They are the bonding electrons, the ones that facilitate chemical reactions.
Think about it! Without these little guys, nothing would ever combine. We wouldn't have water to drink, air to breathe, or even the screens you're looking at right now. They are the fundamental architects of our material world.
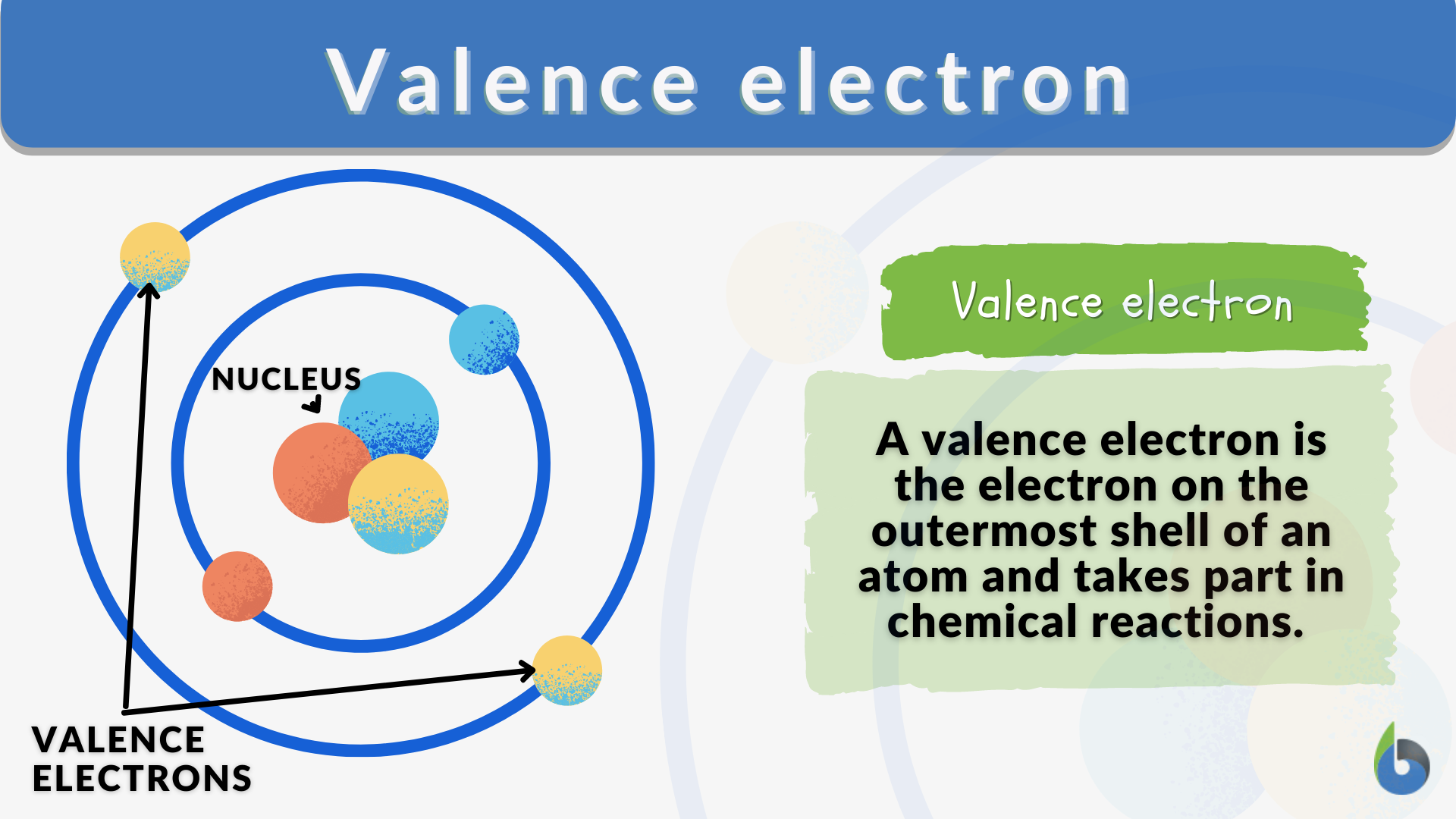
The phrase that best describes a valence electron is one that points to their outward-facing nature and their role in creating connections. They are the outermost electrons. This simple description hints at so much more.
Why is this so entertaining? Because it’s the secret language of the universe! It's like a hidden code that explains why things behave the way they do. These tiny particles are the unsung heroes of chemistry.
The fact that something so small can have such a massive impact is truly mind-boggling. It's a reminder of the intricate beauty of nature. And it all starts with these amazing valence electrons.
When you hear "valence electron," think of them as the social butterflies of the atom. They are the ones who venture out, who mingle, and who form the bonds that hold everything together. They are the ambassadors of the atomic realm.
The phrase "outermost electrons" is pretty spot-on. It tells you where they are. But it also implies what they can do. Being on the outside means they are available for interaction.
They are the electrons involved in chemical bonding. This is their primary, most exciting function. They are the ones that make atoms interact and form molecules. It’s where the magic truly happens.
So, the phrase that describes a valence electron perfectly should capture both their location and their crucial role. They are the electrons in the outermost shell. This is the key to understanding atomic behavior.

Why is this so special? Because these electrons determine how an atom will react. They dictate whether an atom will be eager to share, eager to take, or perfectly content on its own. It’s their personality, revealed by their outer layer.
Imagine each atom as a tiny, unique character. The valence electrons are like their expression, their stance, their open hand or clenched fist. They are the outward signs of their atomic intentions.
Consider the element oxygen. It has valence electrons that make it incredibly reactive. It’s always looking to grab electrons from other atoms, which is why we need it to breathe and why fire needs it to burn. It’s all about those outer electrons.
Or think about the noble gases, like helium or neon. They have a full set of valence electrons, which makes them very stable and unreactive. They are the introverts of the atomic world, content to stay by themselves.
So, a phrase that encapsulates this is vital. It's not just about where they are, but what that means. It’s about their potential for interaction. The phrase bonding electrons really hits the nail on the head.
These are the electrons that participate in forming chemical bonds. They are the bridge between atoms. Without them, atoms would just float around independently, like ships without sails.

This is what makes chemistry so fascinating! It’s all about how these valence electrons, these outermost electrons, decide to connect. It’s a dance of attraction and repulsion, of sharing and taking.
The phrase reactive electrons also works wonderfully. It emphasizes their ability to initiate change. They are the sparks that ignite chemical reactions. They are the catalysts for creation.
It’s the outward-facing nature of these electrons that makes them so crucial. They are the ones that are accessible, the ones that can interact with the outside world. They are the atom's interface with everything else.
And this is why understanding valence electrons is so important. It’s the key to unlocking the secrets of matter. It's how we understand why different substances behave in such diverse ways.
So, the phrase you’re looking for is one that speaks to their position and their function. They are the electrons on the periphery. They are the ones involved in forming the connections that create our world.
Think of them as the atom's handshake. The valence electrons are the hands that reach out to other atoms. They are the ones that allow for the formation of molecules, of compounds, of everything.
The phrase that best describes a valence electron is one that is both simple and profound. It points to their location and their role. They are the electrons responsible for chemical bonding.
This phrase captures their essence perfectly. It tells you where they are (responsible) and what they do (chemical bonding). It’s the core of their identity and their significance.
It's like knowing the secret handshake of the universe. When you understand valence electrons, you start to see the world differently. You see the connections, the interactions, the constant dance of atoms.
And all of this amazing complexity, all of this wonder, starts with a few tiny particles on the outside of an atom. They are the ultimate connectors, the architects of reality.
So, the next time you hear about valence electrons, remember their special role. They are the outermost, reactive, bonding electrons. They are the ones making it all happen. Isn’t that just the coolest thing?
The phrase that truly captures the essence of a valence electron is one that highlights its outward-facing position and its critical role in forming connections. They are, in essence, the electrons that drive chemical reactions.
It’s their location and their purpose that makes them so fascinating. They are the atomic pioneers, venturing out to form the bonds that build everything around us. It’s a truly remarkable concept.
So, if you're looking for that perfect phrase, remember the valence electrons. They are the ones on the edge, ready to make a difference. They are the connectors of the atomic world. And that’s what makes them so incredibly special and endlessly entertaining to learn about.