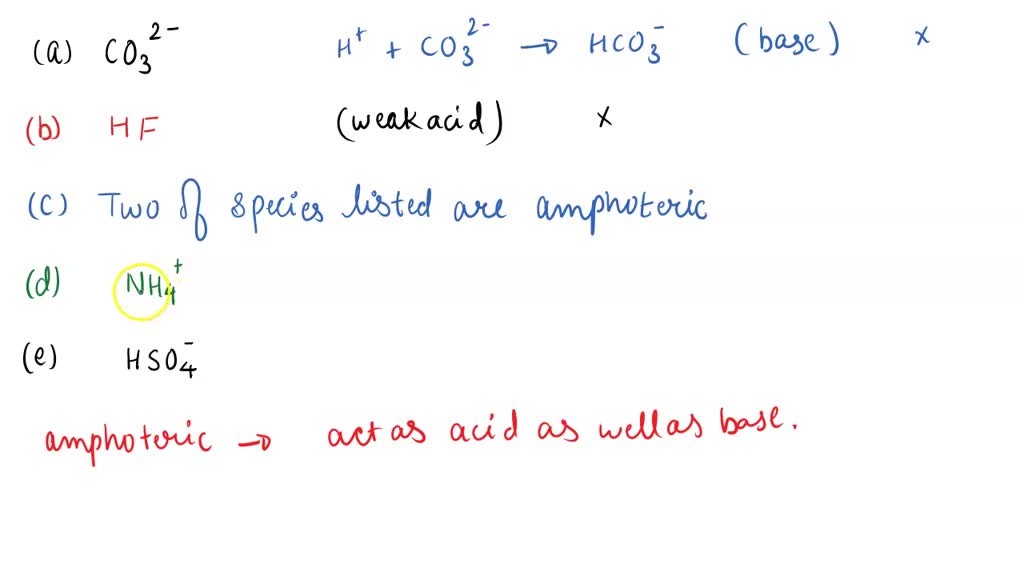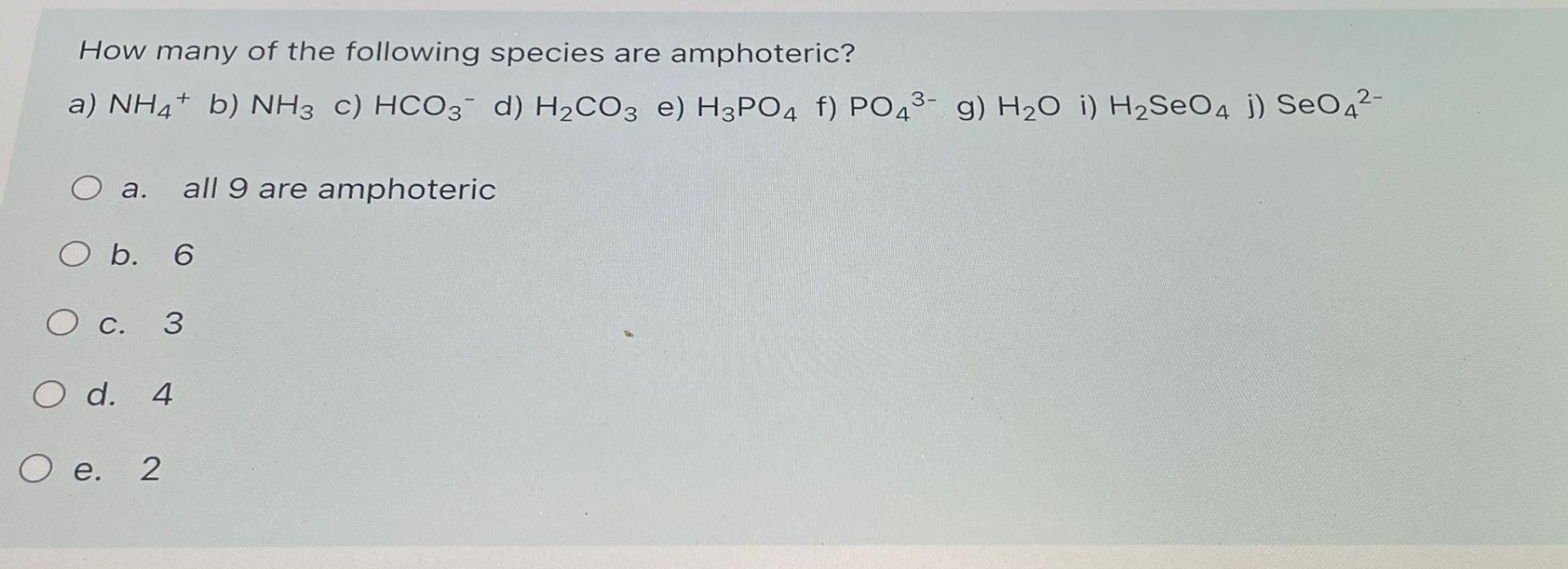Which Of The Following Species Is Amphoteric
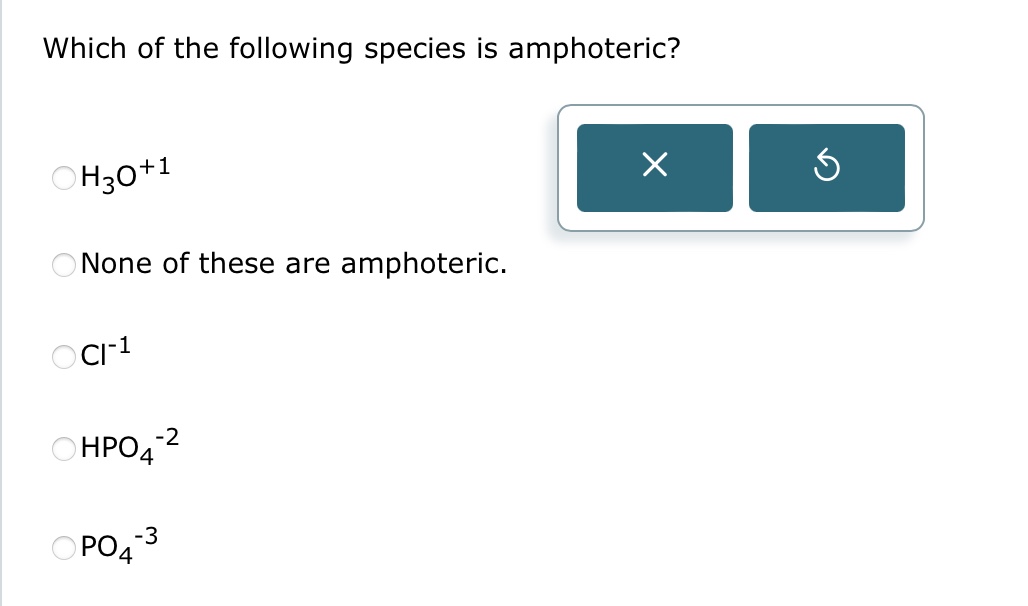
You know, I was fiddling around in the kitchen the other day, trying to whip up this fancy soufflé that, let's be honest, looked more like a deflated pancake. My friend, who's a whiz in the lab (and also happens to make a killer soufflé, what are the odds?), popped by. She took one look at my culinary disaster and then at the slightly murky water I'd used to rinse some spinach. "Ah," she said, with that knowing smirk, "you've got some interesting chemical reactions cooking going on there."
It got me thinking about chemistry, and how sometimes, things aren't quite as straightforward as you'd expect. Like, sometimes a substance decides to be two things at once, depending on who it's hanging out with. Sounds a bit like a teenager, right? "I'm a rebel!" to their parents, but "I'm a sweetheart!" to their friends. Well, in the wild and wonderful world of chemistry, we have a term for that: amphoteric. Fancy word, I know. Stick with me!
So, what exactly is amphoteric? Basically, it means a substance can act as either an acid or a base. Mind. Blown. It’s like a chemical chameleon! Acids, as you probably remember from those slightly terrifying high school chemistry classes, tend to donate protons (those little positively charged guys). Bases, on the other hand, are usually the proton acceptors. They're like the welcoming committee for those protons.
Must Read
But an amphoteric species? It's got the best of both worlds. It can be the generous giver when the situation calls for it, and the eager receiver when needed. It’s all about context, my friends. Just like how you wouldn't wear your pajamas to a job interview (unless it’s a very specific kind of job interview, I suppose), a chemical species’ behavior is dictated by its surroundings.
Now, the question that’s probably buzzing in your head is: "Which of the following species is amphoteric?" Because, let’s be real, this isn’t just some abstract philosophical debate. This is about knowing your stuff, whether you're cramming for an exam, trying to impress a date with your scientific prowess (highly recommended, by the way), or just genuinely curious about the world around you. So, let's dive in and explore some common amphoteric suspects.
The Usual Suspects: Common Amphoteric Substances
When chemists talk about amphoteric substances, a few names pop up again and again. They’re the rockstars of this particular chemical party. Think of them as the Swiss Army knives of the molecular world.
Water (H₂O): The Ultimate Amphoteric MVP
Yep, you guessed it. That ubiquitous, life-giving stuff we’re all made of? Water is a prime example of an amphoteric compound. How does it pull off this double-act? Well, in the presence of a stronger acid, water can actually accept a proton and become a hydronium ion (H₃O⁺). Think of it like this: the acid is really throwing its weight around, and water, being polite, says, "Sure, I'll take that!"

But then, in the presence of a strong base, water can play the acid role. It can donate a proton, leaving behind a hydroxide ion (OH⁻). This is where the base is the dominant personality, and water decides to be the helpful one, giving up its proton to keep the peace. It's a delicate dance, isn't it? And water, bless its simple little molecule, is a natural at it.
This ability of water to act as both an acid and a base is crucial for so many chemical reactions, especially those happening in aqueous solutions. It's the reason why pure water has a tiny, tiny bit of acidity and a tiny, tiny bit of basicity – it’s auto-ionizing! Wild, right? So next time you’re sipping on some H₂O, give it a nod of respect for its diplomatic skills.
Metal Oxides: The Not-So-Simple Solids
Okay, moving on from the liquid lifeblood of Earth to solids. Certain metal oxides are also known for their amphoteric behavior. This is where things get a little more specific, so pay attention! We’re not talking about all metal oxides, just a select few. These tend to be oxides of metals that are in intermediate oxidation states, often those found in the p-block of the periodic table.
Examples include things like aluminum oxide (Al₂O₃), zinc oxide (ZnO), and lead(II) oxide (PbO). These guys are pretty cool because they can react with both acids and bases. For instance, aluminum oxide will happily react with a strong acid like hydrochloric acid (HCl) to form aluminum chloride and water. That’s its acidic side showing.

But then, if you throw in a strong base, like sodium hydroxide (NaOH), aluminum oxide does something different. It reacts to form a complex ion, like sodium tetrahydroxoaluminate. It’s essentially acting as an acid here, reacting with the base. It’s a fascinating duality. It’s like they have this built-in flexibility, making them valuable in various industrial processes where you might need to dissolve or react with both acidic and basic impurities.
Metal Hydroxides: The Watery Cousins
Similar to their oxide counterparts, some metal hydroxides also exhibit amphoteric behavior. These are basically metal oxides that have reacted with water. Again, it's usually those metals with intermediate oxidation states.
Think about aluminum hydroxide (Al(OH)₃) and zinc hydroxide (Zn(OH)₂). They behave in a very similar fashion to their oxide relatives. With acids, they’ll act like bases, forming salts and water. With strong bases, they'll act like acids, dissolving to form complex ions.
It’s important to remember that not all metal hydroxides are amphoteric. Strong bases like sodium hydroxide (NaOH) are strictly bases, and metal hydroxides of highly electropositive metals (like potassium hydroxide, KOH) are also strongly basic. The amphoteric ones are the ones that are less strongly ionic and have a bit more "character," if you will.

Certain Amino Acids: The Building Blocks of Life
Now, for something completely different! Even some organic molecules can be amphoteric. Take amino acids, for example. These are the fundamental building blocks of proteins, and they have a very special structure that allows them to act as both acids and bases.
An amino acid has an amino group (-NH₂) and a carboxyl group (-COOH) attached to the same central carbon atom. The amino group is basic (it can accept a proton), and the carboxyl group is acidic (it can donate a proton). So, depending on the pH of the solution, the amino acid can shift its behavior.
In an acidic solution, the amino group will likely accept a proton. In a basic solution, the carboxyl group will likely donate a proton. This means amino acids can exist in different charged forms, which is super important for how proteins fold and function. It's another testament to the incredible versatility of molecules, even the ones that make us, well, us!
How to Spot an Amphoteric Species
So, you're faced with a list of chemical species. How do you figure out which one is the amphoteric rockstar? Here are a few clues:
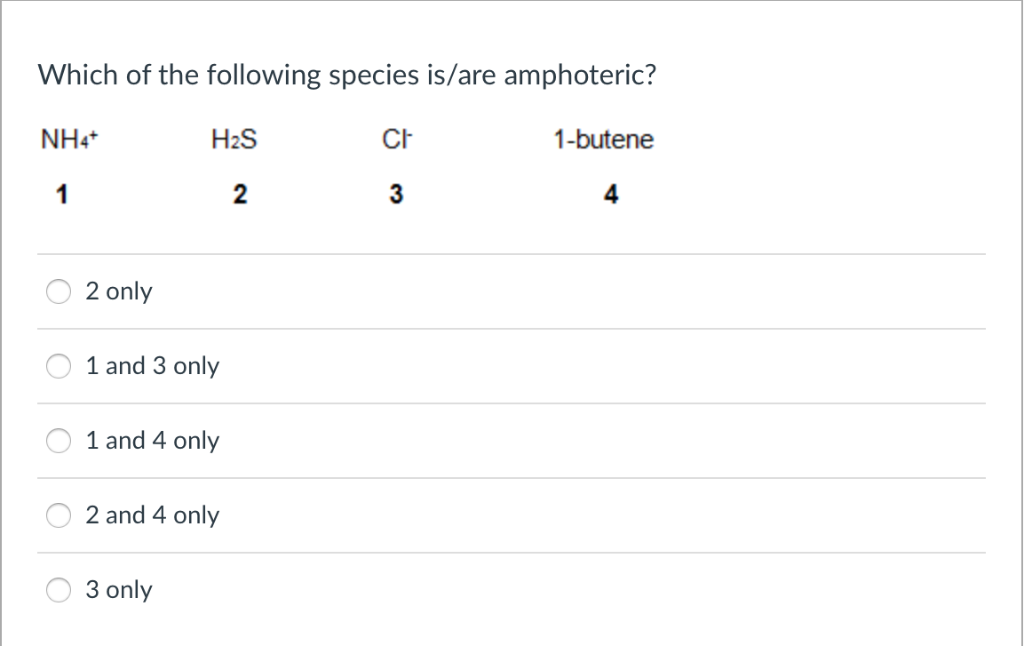
- Look for the 'middle ground' metals: As we touched on, amphoteric oxides and hydroxides often involve metals like aluminum, zinc, lead, tin, and chromium. They aren't the super reactive alkali metals or the noble, unreactive ones. They're somewhere in the middle, allowing for that dual personality.
- Consider the structure: For organic molecules like amino acids, look for functional groups that can either accept or donate protons. The presence of both an acidic group and a basic group on the same molecule is a huge hint.
- The ultimate test: reaction with both acids and bases: The definitive characteristic is the ability to react with both a strong acid and a strong base. If a substance only reacts with acids or only with bases, it's not amphoteric. It’s the willingness to engage with both ends of the pH spectrum that makes it special.
It's not always immediately obvious from just looking at a chemical formula. Sometimes you need to know a bit about the typical behavior of certain elements or functional groups. It's a bit like being a detective – you gather clues and put the pieces together.
Why Does It Even Matter?
You might be thinking, "Okay, cool, some molecules can be two things. So what?" Well, it matters a surprising amount! This amphoteric nature is fundamental to many chemical processes:
- Buffer solutions: Many buffer solutions, which are crucial for maintaining stable pH in biological systems and chemical reactions, rely on amphoteric substances or pairs of acidic/basic species.
- Industrial applications: The ability of metal oxides and hydroxides to react with both acids and bases makes them useful in processes like purification, extraction, and manufacturing. Think about how they might be used to remove impurities or to facilitate a specific reaction.
- Biological systems: As mentioned with amino acids, amphoteric behavior is vital for the intricate workings of life. Proteins, enzymes, and cellular processes all depend on molecules that can adapt their behavior.
It’s this adaptability that makes chemistry so fascinating. It’s not rigid; it’s dynamic. Substances are constantly interacting and influencing each other, and the amphoteric ones are the ultimate diplomats in this molecular world.
A Final Thought (and maybe a quiz question!)
So, to circle back to that original, unstated question: Which of the following species is amphoteric? Without a list to choose from, I can’t give you a definitive answer. But hopefully, by now, you have a much better idea of what to look for! If you see water, aluminum oxide, zinc oxide, or an amino acid on a list, you're on the right track.
Remember, it's about the ability to act as both an acid and a base. It's about being flexible, adaptable, and a true chemical all-rounder. Chemistry can be a bit like a puzzle, and understanding concepts like amphoterism is like finding a key piece that unlocks a whole new level of understanding. Keep exploring, keep questioning, and who knows, maybe your next kitchen experiment will involve a little less pancake and a little more chemistry!