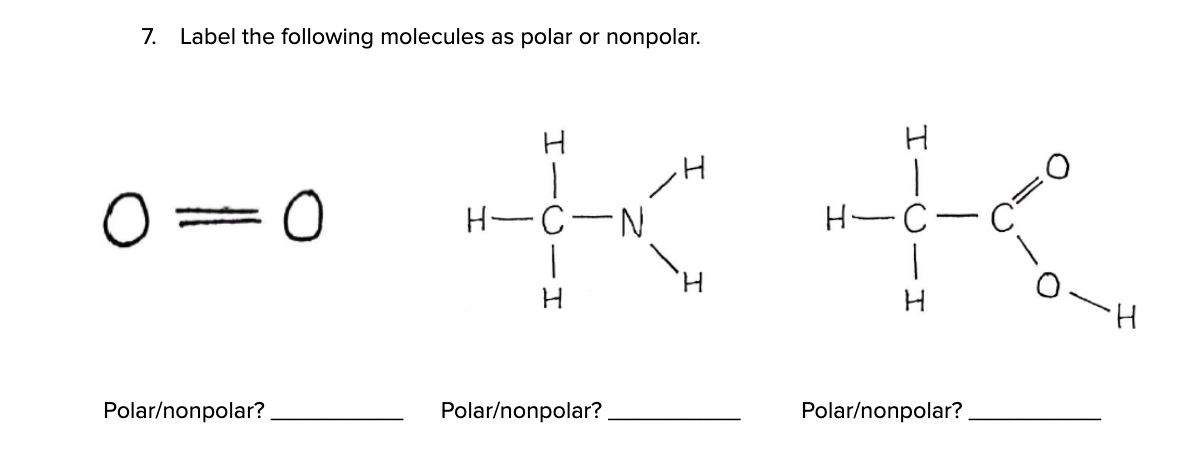
Which Of The Following Molecules Is Nonpolar

Hey there, science curious folks! Ever look at tiny little things, like the building blocks of everything around us, and wonder how they behave? Today, we're diving into the super-duper interesting world of molecules, those little champs that make up your morning coffee, the air you breathe, and even that fluffy cat you might be cuddling. We're going on a mission, a fun little treasure hunt to find out which molecule is the chillest of the bunch, the one that doesn't play favorites with its electrical charges. Get ready for some molecular merriment!
Imagine molecules like tiny, bustling neighborhoods. Some neighborhoods are like a perfectly organized street with houses all neatly lined up, sharing their resources equally. Others are a bit more… dramatic. They have houses that are a little bit more "popular" and attract all the attention, leaving other houses feeling a tad neglected. It's all about how the "stuff" inside these molecular houses is arranged. And that, my friends, is where our nonpolar friend shines.
So, what does it mean for a molecule to be nonpolar? Think of it like a perfectly balanced seesaw. Everyone's having a great time, no one's tipping over, and the weight is distributed evenly. In the world of molecules, this "weight" is actually electrical charge. A nonpolar molecule is like the ultimate peacemaker, where the electrical charges are spread out so evenly, nobody's really pulling harder than anyone else.
Must Read
Let's get our detective hats on and examine some suspects. We've got a lineup of potential molecular celebrities, each with their own unique personality. Our mission, should we choose to accept it (and we totally do!), is to identify the one that's the ultimate symbol of molecular harmony.
Suspect Number One: Water (H₂O)
Ah, water! The lifeblood of our planet, the reason we can make awesome soups, and the bane of any poorly sealed picnic basket. You probably know water well. It's that clear, refreshing stuff that comes out of your tap and makes those delightful little splashes when you jump in a pool. But when it comes to being nonpolar, water is a bit of a drama queen.
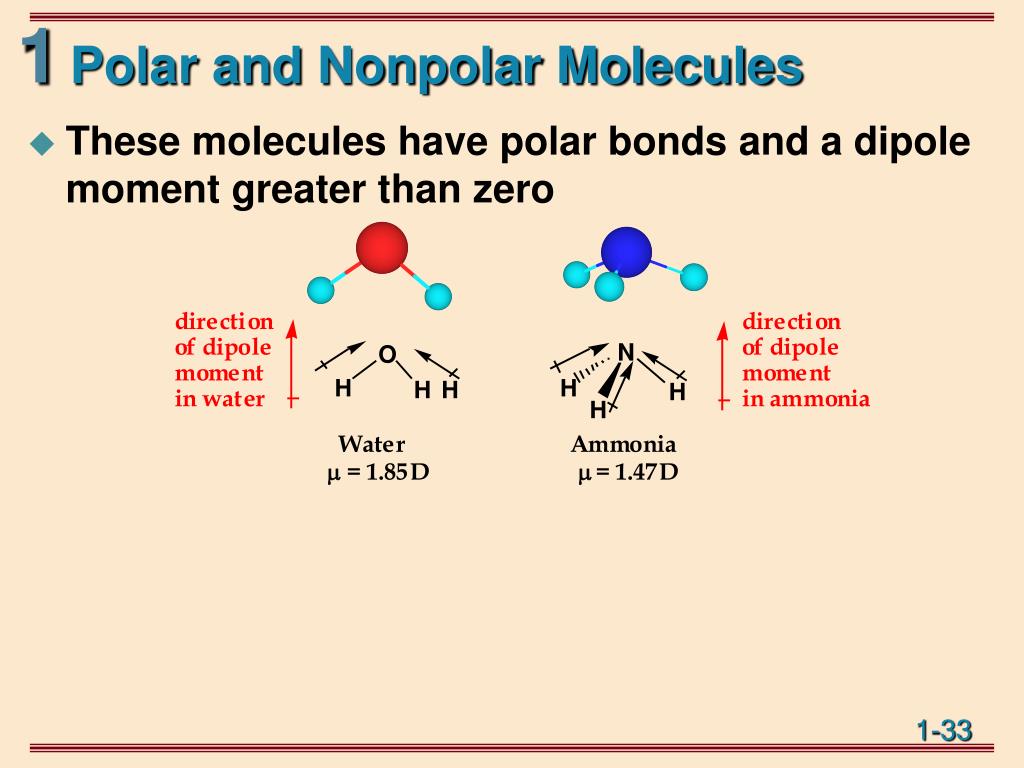
Water molecules are like a tiny, bent boomerang. They have a oxygène atom in the middle, which is like the popular kid at school, hogging most of the "attention" (which in molecule-speak, means electrical charge). Then you have two hydrogen atoms on either side, looking a little bit less popular. This uneven distribution of charge makes water a polar molecule. It’s like one end of the molecule is a little bit more “positive” and the other is a little bit more “negative.”
This polarity is actually why water is such an amazing solvent. It’s like a super-friendly partygoer that can get along with all sorts of other molecules, dissolving them like a charm. But for our nonpolar quest, water is unfortunately disqualified. It’s just a little too… charged up!
Suspect Number Two: Carbon Dioxide (CO₂)
Next up, we have carbon dioxide, or CO₂. You might know this one from the fizzy bubbles in your soda or the stuff we exhale after taking a big breath. It’s a pretty common molecule, buzzing around us all the time. So, is it the nonpolar champion we're looking for?
Let’s look at its structure. Carbon dioxide is like a perfectly straight stick. It has a carbon atom in the very center, and then two oxygen atoms, one on each end. Now, the oxygen atoms are a bit more "attractive" electrically than the carbon atom. However, here’s the magic trick!
Because the molecule is perfectly straight and the two oxygen atoms are on opposite sides, their "pull" on the electrical charges cancels each other out. It’s like having two equally strong people pushing on opposite ends of a door – the door doesn't move! The charges are perfectly balanced. Think of it like two friends having a tug-of-war, but they’re perfectly matched, so the rope doesn’t budge. This makes carbon dioxide a nonpolar molecule!

So, CO₂ is looking pretty good for the nonpolar crown. It’s symmetrical, it’s balanced, and it’s got that chill, even distribution of charge we’re searching for. It’s the epitome of molecular détente!
Suspect Number Three: Ammonia (NH₃)
Now, let’s meet ammonia, or NH₃. This molecule has a bit of a reputation for being… pungent. You might have smelled it in some cleaning products or noticed its distinctive odor in certain places. It’s a molecule with a strong personality, to say the least!
Ammonia is shaped a bit like a tiny pyramid or a tripod. It has a central nitrogen atom, which is like the bossy one, pulling a lot of the electrical charge towards itself. Then, you have three hydrogen atoms sitting at the base of the pyramid. This arrangement is super important.
Because the nitrogen atom is so much more "electrically attractive" and it’s all bunched up on one side of the molecule, the charges are NOT evenly distributed. It’s like having a whole bunch of magnets clustered on one side of a toy car. That side is going to be way more attractive to metal objects! This makes ammonia a decidedly polar molecule. It has a distinct positive end and a distinct negative end.

So, while ammonia is fascinating for other reasons (like its ability to dissolve in water and its important role in many biological processes), it’s not the nonpolar hero we’re hunting for. It’s too busy being electrically opinionated!
Suspect Number Four: Methane (CH₄)
Last but certainly not least, let’s consider methane, or CH₄. This is the main ingredient in natural gas, the stuff that heats your home and cooks your food. It’s a simple, humble molecule that’s incredibly abundant on Earth, and also out in space. Could this be our winner?
Methane has a structure that’s like a perfect, symmetrical tetrahedron. Imagine a pyramid with a flat base and a point at the top. At the very center is a carbon atom, and at each of the four corners of the tetrahedron is a hydrogen atom. This arrangement is key!
While the carbon atom does have a slightly stronger pull on electrons than the hydrogen atoms, the shape of the molecule is what saves the day. Because the hydrogen atoms are spread out perfectly evenly in this 3D tetrahedral shape, their individual pulls on the electrons effectively cancel each other out. It's like having four friends equally spaced around a table, each holding a string attached to a central object. No single friend can drag the object towards them because the pulls are perfectly balanced from all directions.

This perfect symmetry and the resulting even distribution of electrical charge make methane a truly nonpolar molecule. It’s the master of molecular equilibrium!
The Verdict!
So, after our little molecular investigation, which molecule emerged as the undisputed champion of nonpolarity? Drumroll, please! It’s a tie between our perfectly linear carbon dioxide and our symmetrically tetrahedral methane! Both of these molecules showcase that beautiful, even distribution of electrical charge that defines a nonpolar compound. They're the chillest, most balanced molecules in our lineup!
These nonpolar molecules are super important in their own right. They're great at dissolving other nonpolar substances, like oils and fats. That's why you might use a nonpolar solvent to get rid of a greasy stain – it's the nonpolar molecules working their magic on the greasy, also nonpolar, mess!
Isn't it amazing how the arrangement of just a few atoms can lead to such different behaviors? From the energetic interactions of polar molecules like water to the serene balance of nonpolar molecules like CO₂ and CH₄, the world of chemistry is full of fascinating stories. Keep looking around, and you'll see these molecular wonders everywhere!