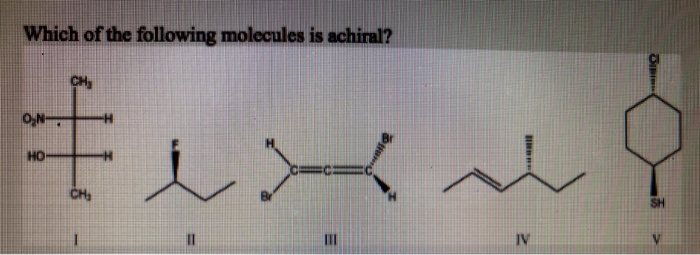
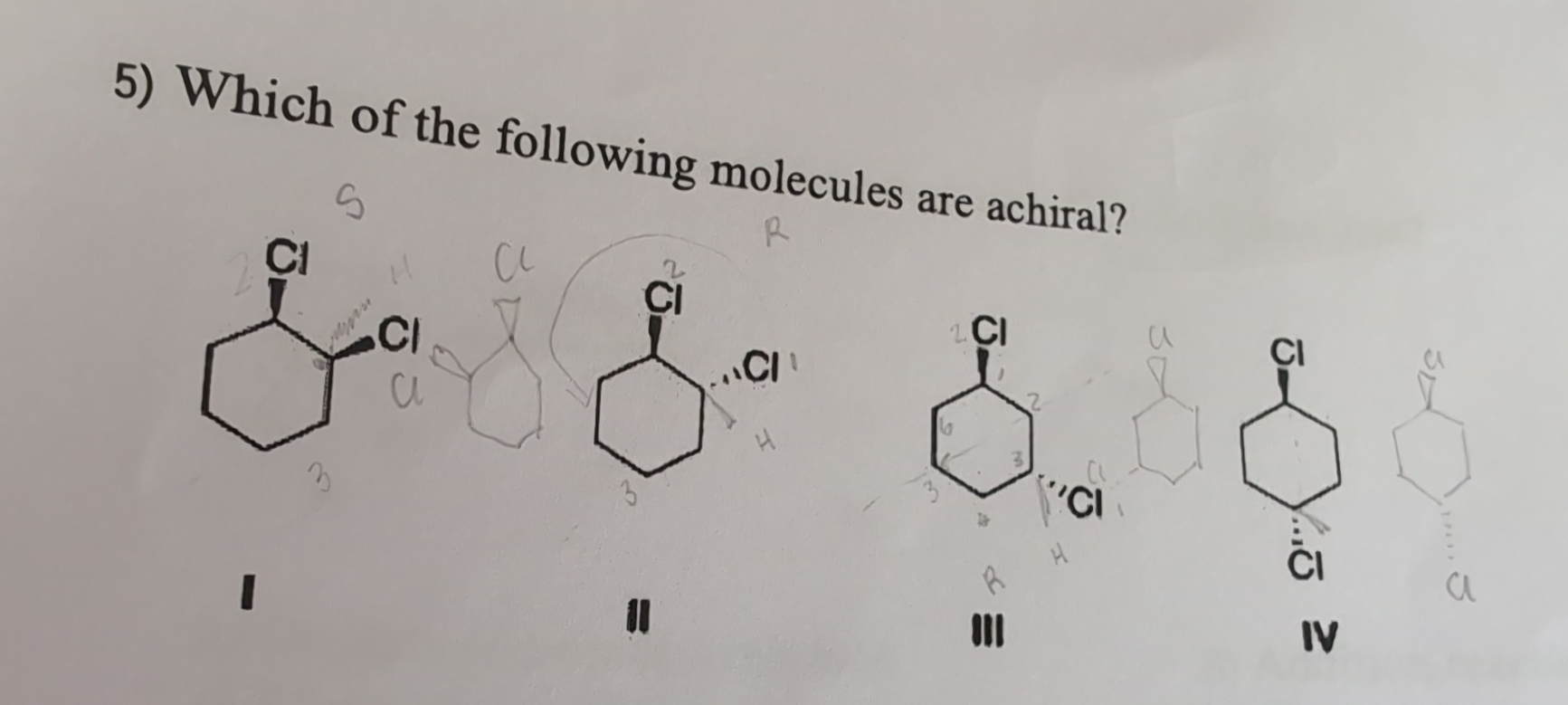
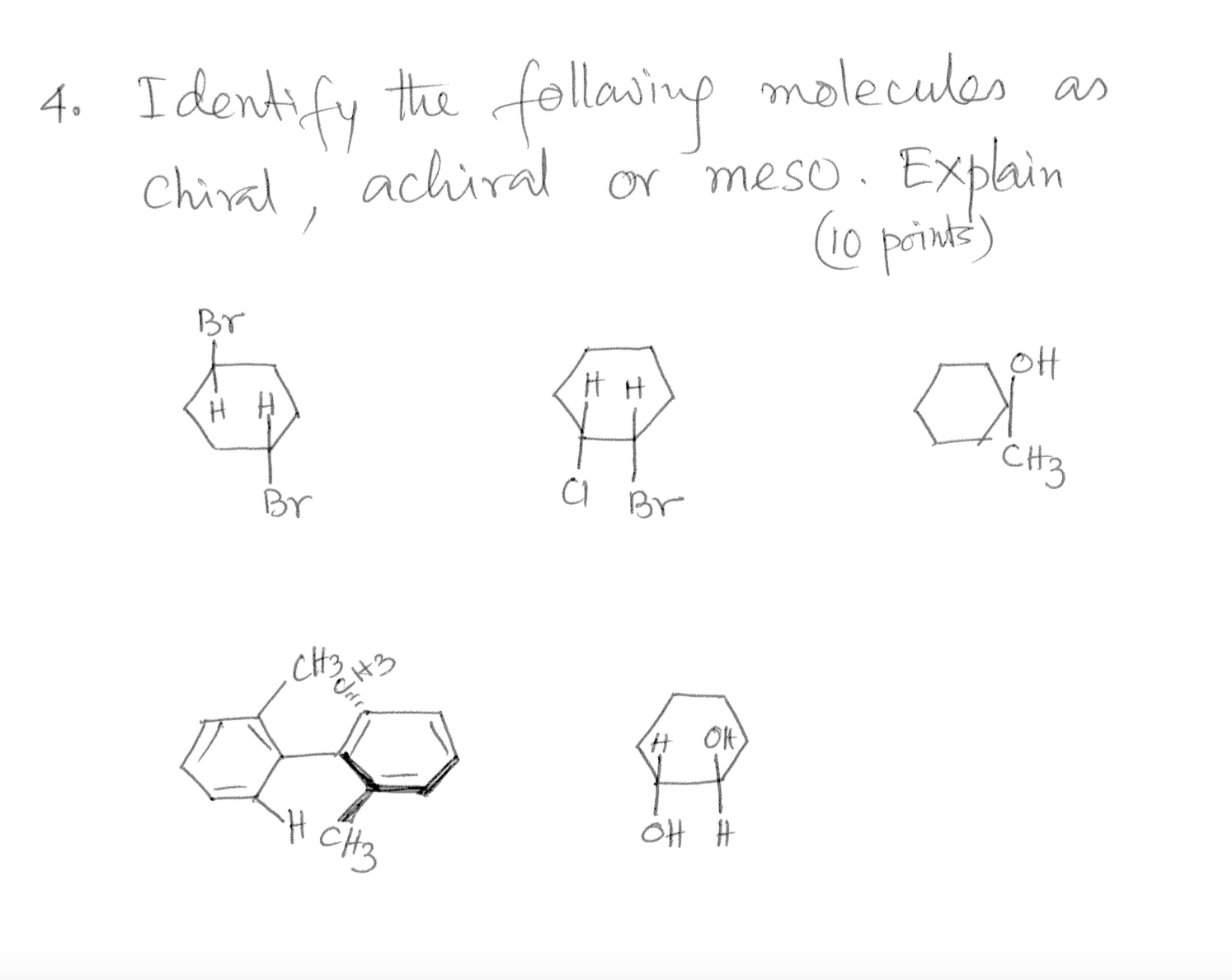
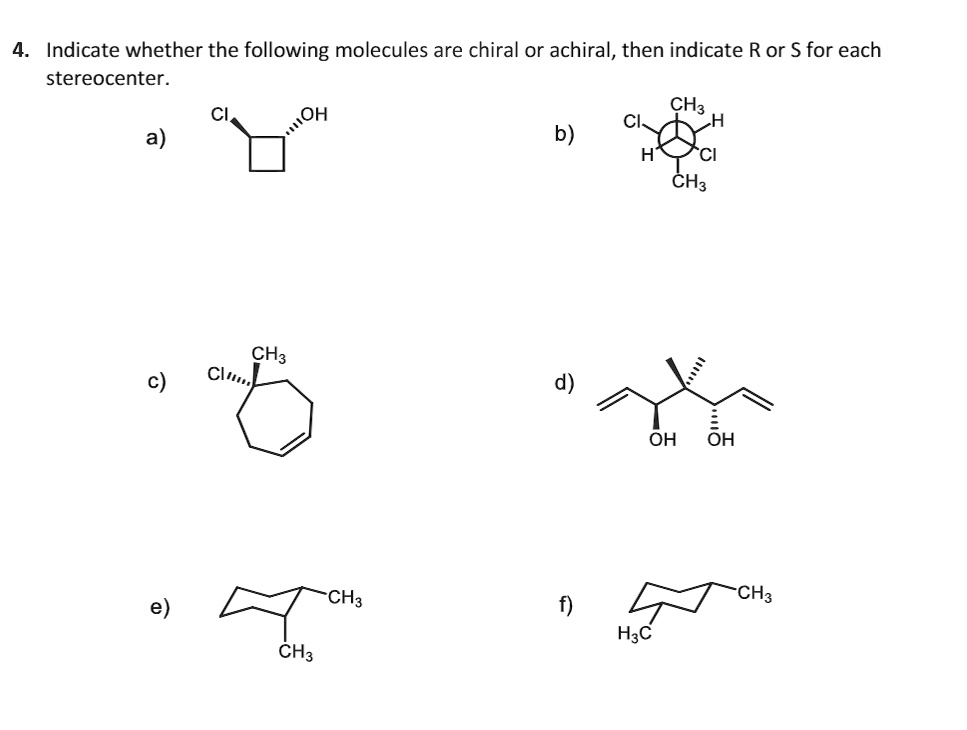
Which Of The Following Molecules Is Achiral
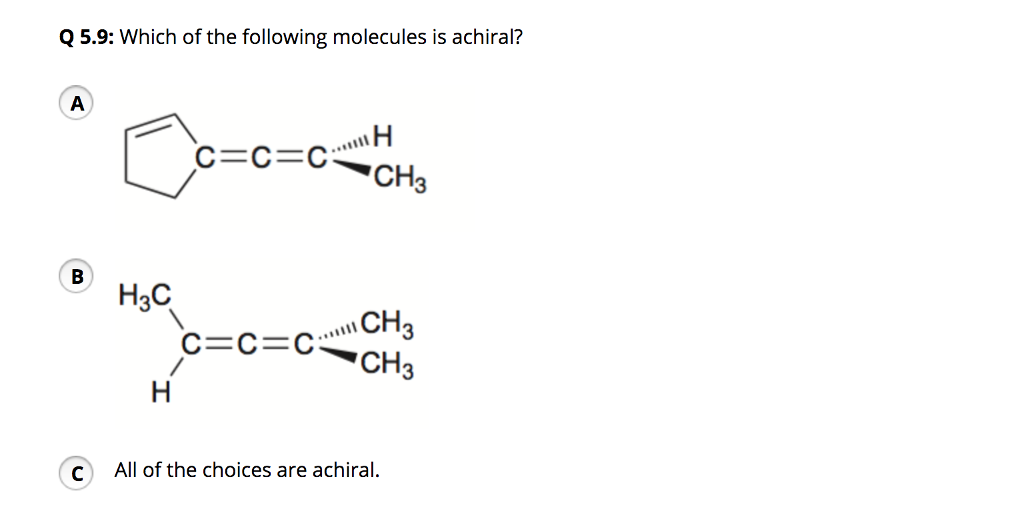
Hey there, fellow humans! Ever looked at something and thought, "Hmm, that's pretty neat," but then totally forgotten about it by lunchtime? We all do it! Our brains are like busy little hummingbirds, flitting from one interesting thing to the next. Today, though, I want to chat about something that might sound a tad science-y, but I promise we're going to make it as cozy and familiar as your favorite comfy chair. We're talking about molecules, and specifically, which ones are achiral. Stick with me, it’s actually pretty fun!
So, what in the world does "achiral" even mean? Imagine you have a glove. You know, the kind you wear when it's chilly outside, or maybe when you’re gardening? If you hold up your left glove, and then try to place your right glove perfectly on top of it, they’re never quite going to match up exactly, right? They're mirror images, but they're also different. They’re like twins who look alike but have completely opposite personalities. That's kind of what being chiral means for molecules. They're mirror images of each other, but you can't superimpose them perfectly. They're like a pair of hands – your left hand and your right hand are mirror images, but you can’t stack them perfectly on top of each other so all your fingers line up. One is always going to be off.
Now, an achiral molecule is the opposite. Think about a simple, plain ol' ball. If you have a ball, and you hold up its mirror image, and then you try to put them together, poof! They match up perfectly. It doesn't matter how you turn it; it's the same. An achiral molecule is like that ball – it’s symmetrical. It doesn't have that "handy" quality of being a mirror image that can’t be superimposed. It’s like a perfectly round pizza; no matter how you slice it, it’s still a pizza!
Must Read
Why should we even care about whether a molecule is like a glove or like a ball? Well, it turns out these little molecular differences can have a huge impact on our lives, especially when it comes to things we eat, drink, and even the medicines we take. Our bodies are incredibly sophisticated machines, and they often interact differently with chiral molecules depending on which "hand" they are. It’s like trying to put on a shoe: a left shoe only fits comfortably on your left foot, right? Your body is the same way with certain molecules.
Let’s dive into some examples, shall we? Think about lemons. The fresh, zesty smell of a lemon comes from a molecule called limonene. Now, limonene actually exists in two forms, a left-handed version and a right-handed version. The right-handed limonene smells like lemons, which is pretty awesome for lemonade! The left-handed limonene, on the other hand, smells more like oranges. So, even though they’re the same basic molecule, the slight difference in their 3D arrangement makes them smell completely different. How cool is that? It’s like having two identical keys that unlock totally different doors – one opens your house, the other opens your neighbor's!
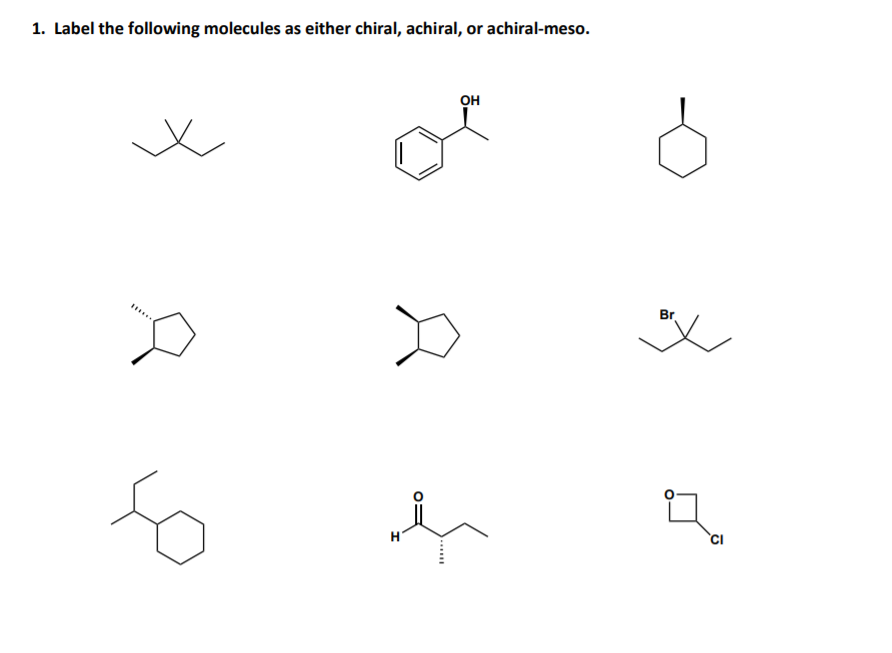
Now, what about achiral molecules? These are the molecules that don't have this handedness. They’re the steady, reliable ones. A perfect example of an achiral molecule is something super common: water. H₂O. You know, the stuff that makes up about 70% of you and me? Water is achiral. It's perfectly symmetrical. You can flip it, turn it, do whatever you want; it’s always going to look like water. This is why water is such a fantastic solvent – it can interact with all sorts of other molecules without getting picky about their handedness.
Another simple, achiral molecule is carbon dioxide, CO₂. It’s linear and symmetrical. Think of it like a perfectly straight ruler. No matter which way you orient it, it's still a ruler. This symmetry is why carbon dioxide behaves the way it does in our atmosphere and in our bodies. It doesn't have different "sides" that our biological systems would react to differently.
So, how do we figure out which molecules are achiral? In the lab, scientists look for a special kind of symmetry. The most common reason a molecule is not achiral (meaning it's chiral) is if it has a chiral center. This is usually a carbon atom that's bonded to four different groups. Imagine a carbon atom as a happy little guy in the center, and he's got four very different friends holding his hands. This makes him asymmetric, and thus, his molecule will likely be chiral. If, however, that carbon atom has two identical friends holding its hands, then the molecule gains some symmetry and becomes achiral. It’s like our carbon friend having two best buddies who are exactly alike – it evens things out!
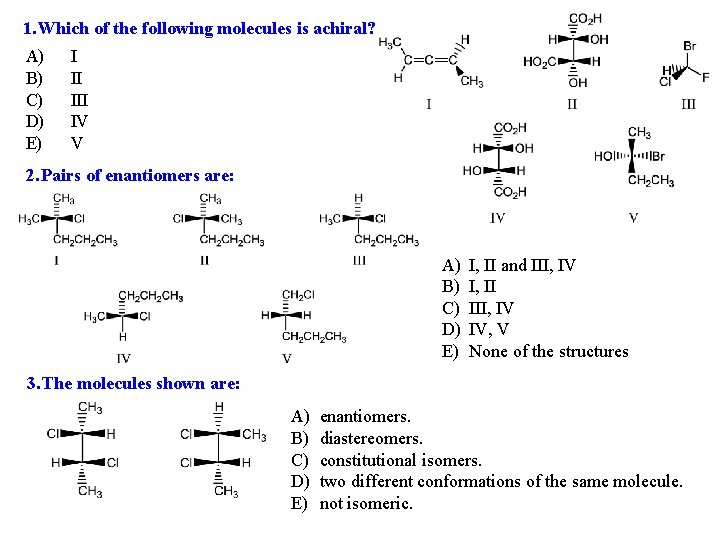
Think about a simple molecule like methane, CH₄. The carbon is in the middle, and it's bonded to four hydrogen atoms. Since all the hydrogen atoms are the same, methane is achiral. It’s like a perfectly balanced scale. No matter what you do, it stays balanced. It’s symmetrical and can’t be distinguished from its mirror image.
Another common achiral molecule you might encounter in your kitchen is ethanol, C₂H₅OH – that’s the alcohol in your favorite beverage! The carbon atoms in ethanol don't each have four different things attached. This lack of a chiral center makes ethanol achiral. It’s like a simple chair; it has symmetry that means you can’t really have a "left" or "right" version that’s fundamentally different in how it interacts with other things in a symmetrical way.

Now, let's bring it back to why you should care. Imagine you're feeling a bit under the weather, and you need a pain reliever. Many drugs are chiral molecules. Sometimes, only one of the two mirror-image forms of a drug works effectively to relieve your pain. The other form might be inactive, or worse, it could have unwanted side effects. This is why drug companies spend a lot of time and money making sure they produce the correct chiral form of a drug. It’s like making sure you get the right key for the medicine cabinet, not one that looks similar but opens the broom closet!
On the flip side, achiral molecules are often the workhorses of chemistry. Because they are symmetrical, they tend to be more predictable and can often be used in larger quantities without worrying about different "versions" interacting in unintended ways. Water, as we mentioned, is the ultimate achiral solvent. Many common chemicals used in manufacturing, like simple acids and bases, are also achiral. They’re the reliable building blocks of the chemical world.
So, the next time you hear the word "achiral," don't let it scare you off. Just picture a perfectly symmetrical ball, a drop of water, or a straight ruler. These are molecules that don't have that "handedness" problem. They are the symmetrical ones, the ones that are the same no matter how you look at them. They’re the unsung heroes of many processes, from making our bodies function to creating the products we use every day. And in a world full of complex and sometimes tricky chiral molecules, it’s nice to know we have these wonderfully simple and symmetrical achiral molecules keeping things grounded and predictable!