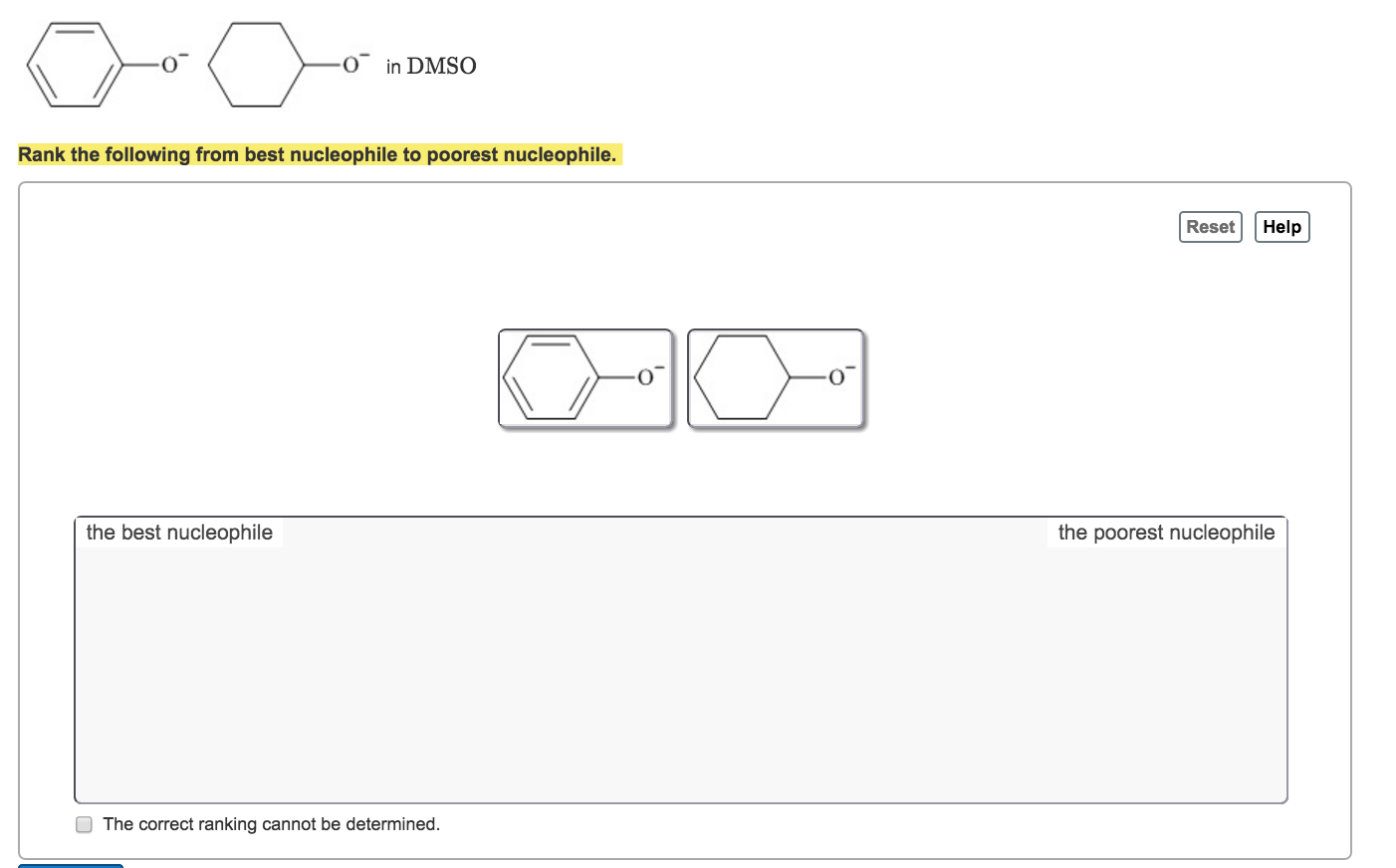
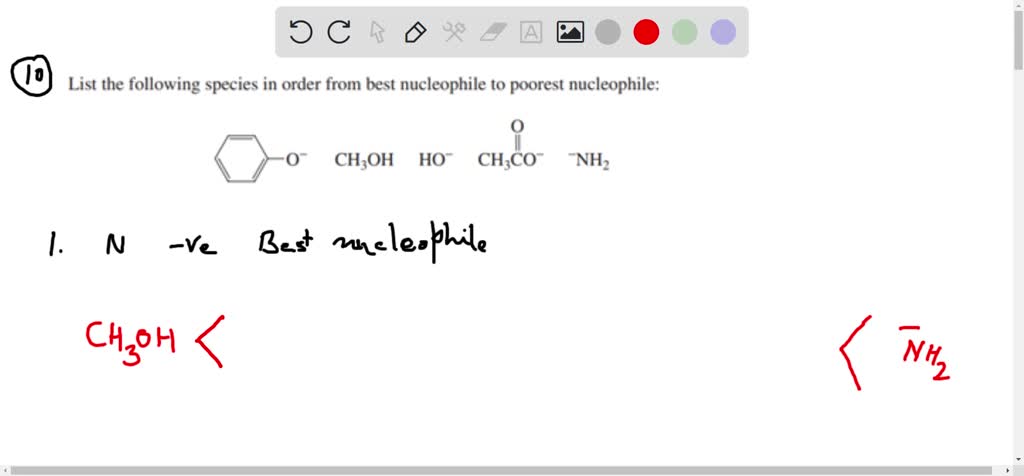
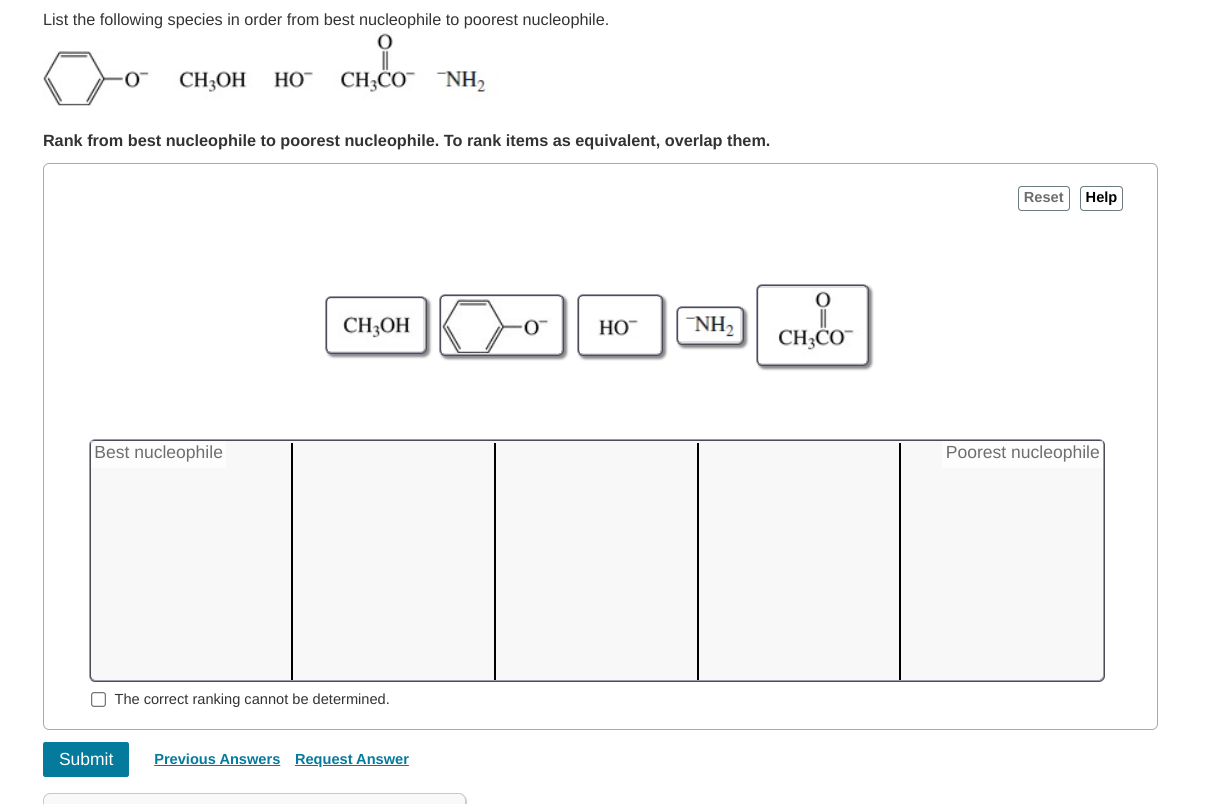
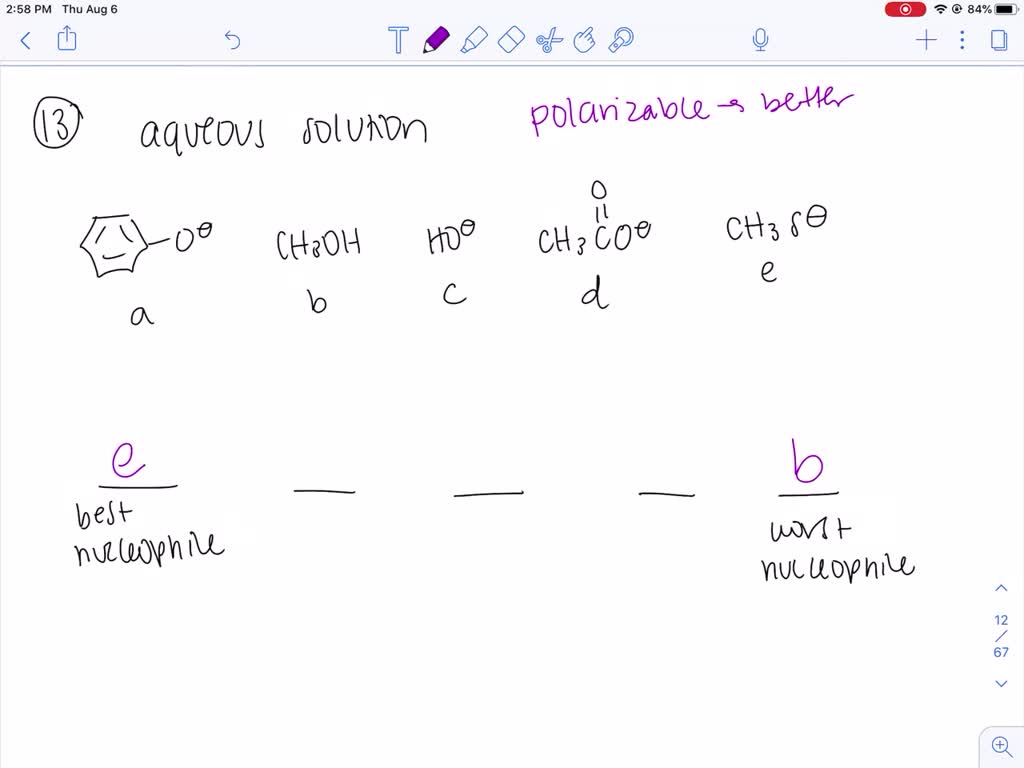
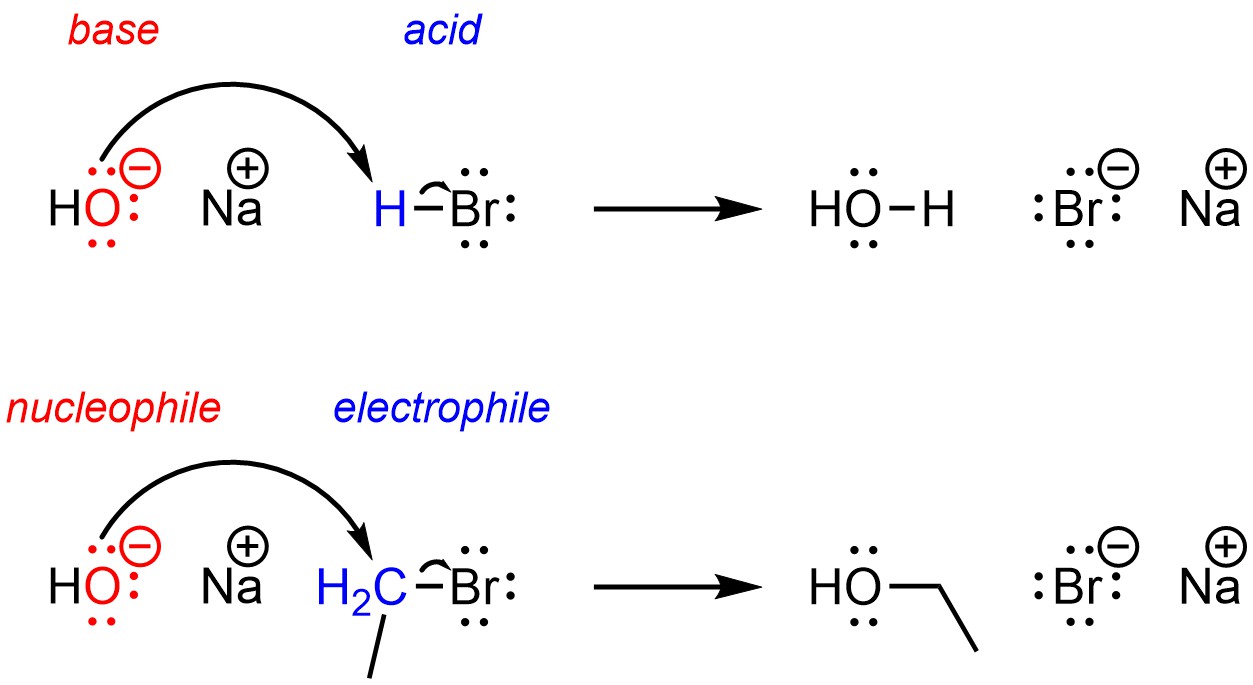
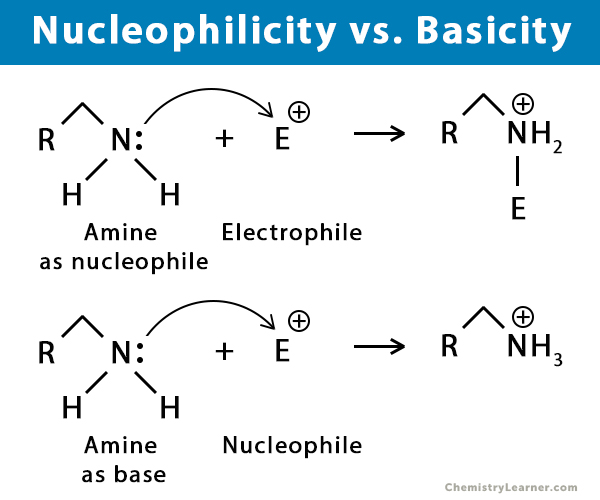
Which Of The Following Is Not A Nucleophile
Have you ever found yourself staring at a blank canvas, a lump of clay, or a pile of yarn, feeling that familiar itch to create something, anything? That creative spark, that urge to bring something new into the world, is a truly wonderful thing. And if you're looking for a fun, accessible, and surprisingly insightful way to explore your artistic side, you might be surprised to learn that understanding a little bit of science can actually be a fantastic jumping-off point. Today, we're going to dive into a question that might seem a little niche, but trust us, it has more creative potential than you might think: Which of the following is not a nucleophile?
Now, before you picture yourself hunched over a chemistry textbook, let's demystify this. A nucleophile, in the simplest of terms, is something that "loves" to donate electrons. Think of it as a generous giver, always ready to share. Understanding this concept, even in a lighthearted way, can unlock a whole new perspective for artists, hobbyists, and even the curious casual learner. For artists, it's about understanding the give and take of reactions – the way colors blend, how materials interact, or even the flow of inspiration itself. Hobbyists can find it a fun way to add a unique angle to their projects, perhaps incorporating scientific themes or visual metaphors. And for casual learners, it’s a gentle introduction to fascinating scientific ideas in a context that’s anything but dry.
Imagine, for instance, applying this idea to abstract painting. You could create a piece where vibrant, electron-rich colors (your nucleophiles) are drawn to and interact with more "electron-deficient" areas on the canvas, perhaps cooler tones or textured spaces. Or consider sculpture: a sculptor might envision the way two different materials, each with its own electron-donating or accepting tendencies, fuse and react. Even in fiber arts, you could explore the interaction of dyes and fabrics as a form of "nucleophilic" attraction. The beauty lies in the versatility of the concept – it can be interpreted visually, thematically, or metaphorically across a vast range of creative endeavors.
Must Read
Ready to give it a try at home? It's simpler than you might think! Start by choosing a medium you enjoy. Grab some paints and explore how different colors seem to "attract" each other on your palette or canvas. You don't need to be a chemist to see how some colors blend seamlessly, almost as if they're donating their essence to create something new. Or, try working with two different types of clay. Notice how they stick together, or how one might slightly alter the texture of the other. You could even use this as inspiration for storytelling, creating characters who are either generous givers (nucleophiles) or those who readily accept (electrophiles, the electron acceptors!). The key is to play and observe.
Ultimately, the enjoyment comes from the aha! moment. It’s in discovering that everyday phenomena, even something as seemingly scientific as chemical reactions, can be a source of endless creative inspiration. It’s about seeing the world with a little more curiosity, a little more wonder, and a lot more potential. So, the next time you’re pondering that artistic challenge, remember our friendly nucleophiles – the givers, the connectors, the ones who initiate change. They might just be the catalyst you need for your next masterpiece!