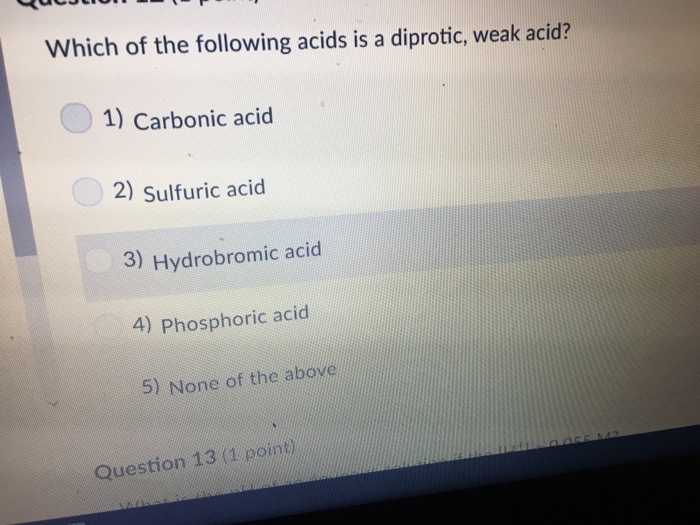
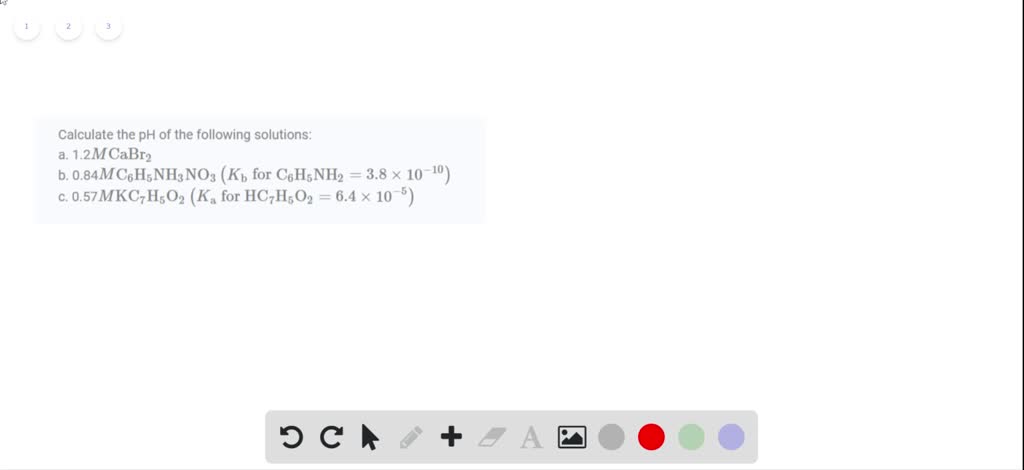
Which Of The Following Is A Diprotic Acid

Ever found yourself staring at a science question and feeling like you're trying to decipher alien hieroglyphics? You know, the kind that throws around words like "monoprotic" and "diprotic" and suddenly your brain just checks out and starts humming elevator music? Well, buckle up, buttercup, because we're about to demystify this whole "diprotic" business in a way that's so easy, you'll be high-fiving your pet goldfish (if you have one, and even if you don't, it's still a win!).
Imagine you're at a party, and there are snacks. Lots and lots of snacks. Now, some of your friends are super generous. They'll hand out one chip at a time. That's like a monoprotic acid. It's got one "thing" it can give away, like a single, precious sprinkle of fairy dust. Nice, but a little stingy, right?
But then, you've got the real champions. These are your party heroes. They don't just offer one chip; oh no! They've got a whole bowl, and they're practically throwing handfuls of chips your way. They're double-dipping… well, not literally (ew!), but they're offering up two of something. These, my friends, are our diprotic acids!
Must Read
Think of it like this: acids are like tiny little chemists who love to share. They've got these special "proton" passengers they like to give away. A monoprotic acid has only one proton to offer. It's like a single-serving soda. Once you've had it, that's it for that particular fizzy delight.
But a diprotic acid? Oh, they're like the buffet of protons! They've got not one, but TWO protons just itching to be shared. They're twice as generous! They're practically doing a little happy dance, just waiting to bestow their proton gifts upon the world (or, more accurately, upon something that needs them). It's like getting a whole two-liter bottle of your favorite soda – twice the fun, twice the fizz!
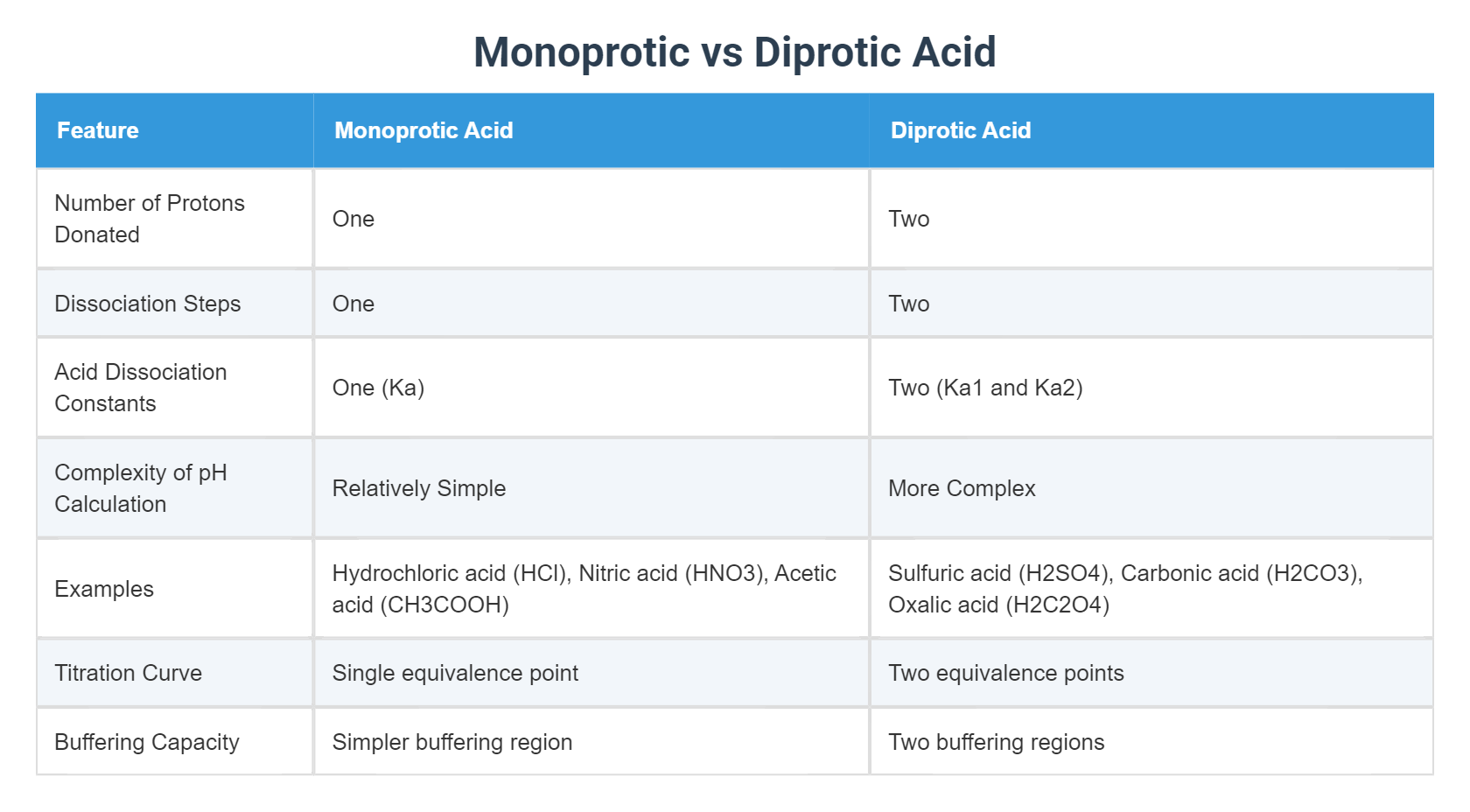
So, when you see one of these fantastic diprotic acids waltzing onto the scene, remember they're not messing around. They're bringing their A-game, offering up their proton treasures in two glorious installments. They're the rockstars of the acid world, the double-feature movie, the all-you-can-eat pancake breakfast of chemical compounds!
Now, let's get down to the nitty-gritty. Which of the following is a diprotic acid? This is where the fun really begins! Imagine you're picking out your favorite flavors at an ice cream shop. You've got your classic vanilla (maybe a monoprotic, who knows!), your adventurous rocky road (definitely a complex one!), and then… you spot it. The double-scoop special. That's our diprotic acid!
Let's consider a few hypothetical (and entirely made-up, for maximum silliness) examples. You see:
Hydrochloric acid (HCl): This little guy is like that friend who only ever has one joke. It's got one proton to give. Pure and simple. Monoprotic!

SOLVED: Lab 6: Titration of an Unknown Diprotic Acid Introduction Sulfuric acid (H₂SO₄): BAM! This one is a proton-giving powerhouse! It’s got not one, but TWO protons ready to roll. It’s like getting two free tickets to your favorite concert! Diprotic!
Nitric acid (HNO₃): Similar to hydrochloric acid, this one's a one-trick pony in the proton department. One and done. Monoprotic!

Diprotic Acid Definition and Examples Phosphoric acid (H₃PO₄): Now this one's a triple threat! It's the friend who not only tells three jokes but also brings snacks, a playlist, and a karaoke machine. It's got three protons to give. Triprotic!
See how that works? It’s all about how many of those special "proton" passengers the molecule is willing to share. A diprotic acid is the one that says, "Here, have one! No, wait, have another one too! I'm feeling generous today!" It’s like getting a surprise bonus, a buy-one-get-one-free deal on protons!
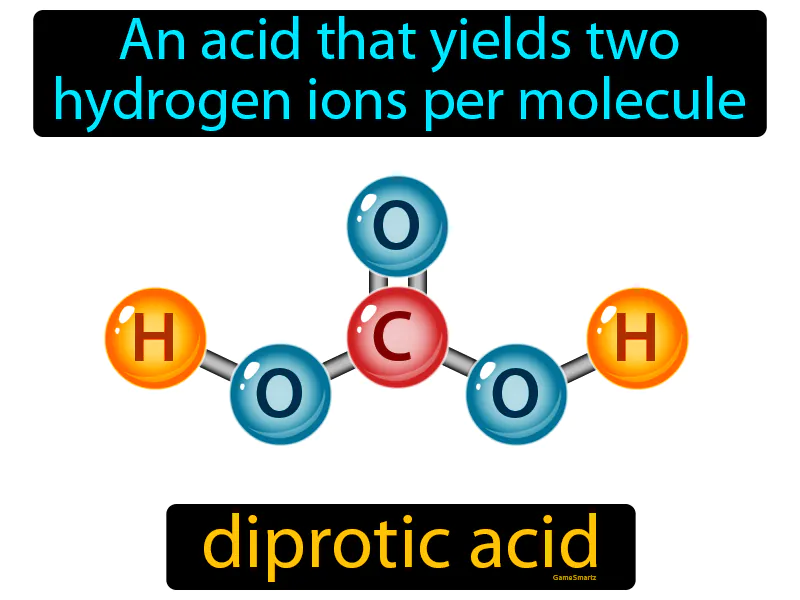
So, the next time you encounter this dazzling question, just picture the snack party. Picture the generous soul offering double handfuls of chips. That, my friends, is your diprotic acid. It’s not scary, it’s not complicated. It’s just a particularly giving kind of molecule, ready to share its proton bounty with the world. And honestly, who doesn't love a good giveaway?
So, next time you're presented with a list, just remember the double-scoop delight, the two-for-one deal, the proton-sharing champion. That's your diprotic acid, out there making the chemical world a more generous and exciting place, one proton at a time!
Embrace the diprotic! It's a concept that's as satisfying as finding an extra fry at the bottom of your bag. You got this!