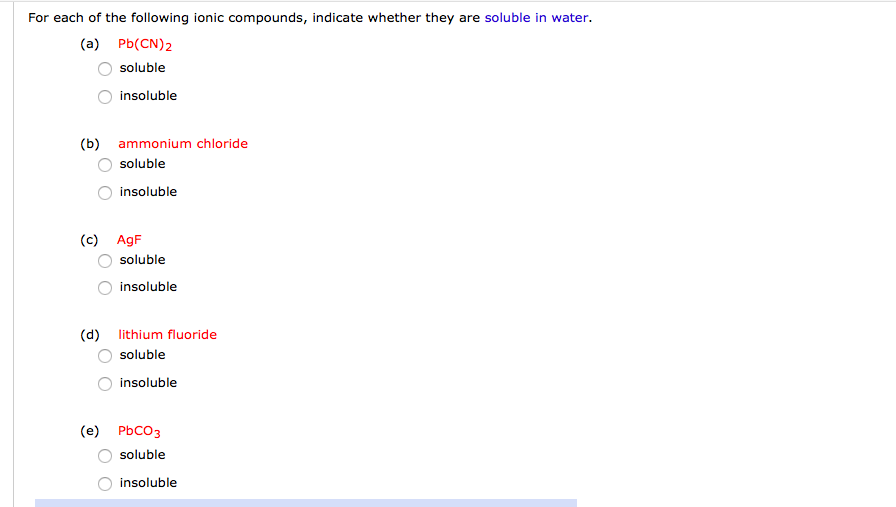
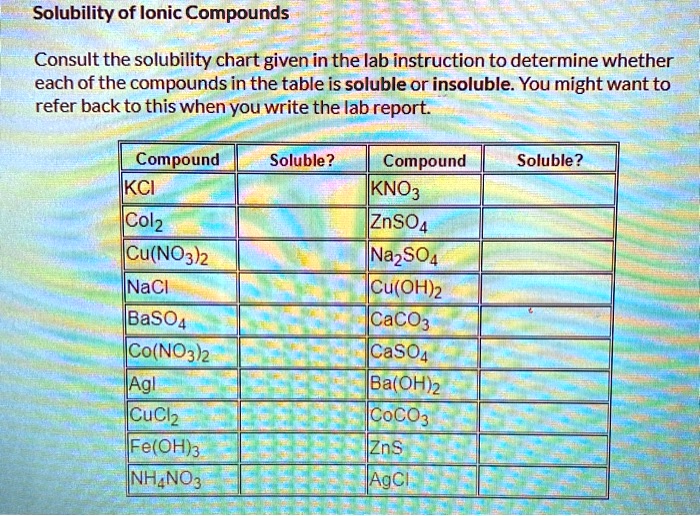
Which Of The Following Compounds Is Soluble

Hey there, coffee buddy! Grab your mug, settle in. We’re gonna chat about something that might sound a little science-y, but trust me, it’s not as scary as it sounds. We’re talking about solubility. Yeah, I know, sounds like something your chemistry teacher droned on about, right? But really, it’s just about what dissolves in what. Like, will that sugar cube disappear in your coffee? Or is it gonna be that sad, grainy lump at the bottom? That’s solubility in a nutshell. Pretty practical, huh?
So, the big question we're pondering today, in our cozy little chat, is: Which of the following compounds is soluble? Now, you might be looking at a list of fancy chemical names and thinking, “Uh, I’m just here for the caffeine, thanks!” But hang in there! We’re going to break it down, no pop quiz, I promise.
Think about it. When you’re making a cuppa, you want that sugar to vanish, right? You want that creamer to swirl in and become one with your dark brew. That’s a successful dissolving act. When something dissolves, it’s basically breaking down into tiny, invisible pieces and spreading out evenly in the liquid. We call that liquid the solvent, and the stuff that dissolves, the solute. Easy peasy, lemon squeezy. Though, if you're trying to dissolve lemon chunks in water, good luck with that – that's a different kind of solubility challenge!
Must Read
Now, the universe of compounds is HUGE. Like, mind-bogglingly enormous. So, how do we even begin to guess which one will play nice with water (which is, you know, our most common solvent, basically the MVP of solvents)? It’s like trying to guess which stranger at a party will become your new best friend. You’ve gotta have some clues, right?
The golden rule, the one that’ll save you from a lot of confusion, is the whole “like dissolves like” thing. Ever heard that before? It’s like the universe’s little motto for solubility. What does it mean? It means polar things like to dissolve in polar things, and nonpolar things like to dissolve in nonpolar things. Simple as that. If you’re feeling a bit lost on the polar/nonpolar thing, don’t sweat it. Think of it like this: polar molecules are a bit like magnets, they have positive and negative ends, and they’re attracted to other things that have opposite charges. Nonpolar molecules? They’re more like… well, they’re just chill. They don’t have those distinct positive and negative bits. They’re more about sharing their electrons equally. No drama, no fuss.
So, What Does This Mean for Our Mystery Compounds?
Water, our trusty solvent, is super polar. It’s like the ultimate social butterfly of molecules. Because water is so polar, it’s really good at dissolving other polar compounds and things that are ionic. Ionic compounds are basically held together by opposite charges, like a magnetic attraction on a molecular level. So, if you see a compound that’s got a lot of positive and negative charges hanging around, or it’s made up of ions, there’s a good chance it’ll be happy to dive into water. Think of salt – NaCl. That’s sodium (Na+) and chloride (Cl-) ions. Boom! Dissolves in water like nobody’s business. You want salt water? Just add salt to water. Done.
On the flip side, if you have a compound that’s mostly nonpolar, it’s not going to be best buds with water. Water will just kind of shrug and say, “Nah, not my scene.” Nonpolar compounds tend to hang out with other nonpolar solvents, like oil or grease. Ever tried to wash greasy hands with just water? It’s a struggle, right? That’s because oil is nonpolar and water is polar. They just don’t mix well. It’s like trying to get a cat and a dog to share a single bed – possible, but there’s usually some tension!
Now, let’s get to the fun part. The actual compounds. Imagine you've got a list. Maybe it looks something like this (don’t worry if you don’t recognize them, that’s what we’re here for!):
- Sodium Chloride (NaCl)
- Methane (CH4)
- Ethanol (C2H5OH)
- Oil (a triglyceride, but let’s just call it oil)
- Sugar (Sucrose, C12H22O11)
Let’s play detective, shall we? We'll put on our imaginary magnifying glasses and squint at each one.
Compound 1: Sodium Chloride (NaCl)
Okay, Sodium Chloride. We already touched on this one. It’s made of a sodium ion (Na+) and a chloride ion (Cl-). Remember what we said about ionic compounds and water? They’re like old friends who haven’t seen each other in a while and are super excited to reunite. Water molecules, with their little positive and negative ends, grab onto those ions and pull them apart. So, is NaCl soluble in water? You bet your sweet bippy it is! It’s practically the poster child for water-soluble ionic compounds. So, if NaCl is on your list, you can probably tick that box with a big, bold, happy checkmark.
Compound 2: Methane (CH4)
Now, Methane. This is the main ingredient in natural gas, which is kind of wild to think about. Chemically speaking, Methane is a simple molecule. It’s got one carbon atom and four hydrogen atoms all bonded together. The thing is, the electrons in those bonds are shared pretty equally. There aren’t those distinct positive and negative poles like in water. It’s a nonpolar molecule. And what do nonpolar molecules like to dissolve in? Yep, you guessed it – other nonpolar things. Water, our polar pal, is not its cup of tea. So, is Methane soluble in water? Not really. You try to mix Methane and water, and they’ll just sort of sit there, politely ignoring each other. They’re not going to blend. So, Methane is a definite no for water solubility.
Compound 3: Ethanol (C2H5OH)
Ethanol. Ah, Ethanol. This is the stuff in alcoholic beverages. Yes, the fun kind of solubility! Now, Ethanol is a bit of an interesting case. It’s got a part of it that’s nonpolar (that C2H5 bit, the ethyl group) and a part that’s polar (the OH group, the hydroxyl group). Because it has that polar OH group, it can actually form hydrogen bonds with water. Hydrogen bonding is like a super-strong attraction between certain atoms, and water loves to hydrogen bond. So, even though it has a nonpolar bit, the polar OH group is enough to make it pretty soluble in water. Think about how liquor mixes with water – they combine pretty easily, right? So, is Ethanol soluble in water? Absolutely! It’s a great example of a molecule that has a bit of both worlds, but the polar part wins out for water solubility.
Compound 4: Oil (Triglyceride)
Oil. We’ve all dealt with oil, haven’t we? Whether it’s cooking oil, motor oil, or that oily film on your skin after a long day. Oil molecules are generally very large and have long chains of carbon and hydrogen atoms. These chains are almost entirely nonpolar. They’re the epitome of “like dissolves like” when it comes to nonpolar solvents. Water, on the other hand, is polar. Remember our greasy hands analogy? Oil and water just don’t mix. They separate. You can shake them up all you want, but eventually, they’ll go back to their own little corners. So, is Oil soluble in water? Nope, not at all. Definitely a no.
Compound 5: Sugar (Sucrose, C12H22O11)
Sugar. Ah, sweet, sweet sugar! We already mentioned it with the coffee. Sugar, like sucrose, is a type of carbohydrate. Its chemical formula is C12H22O11. Now, sugar molecules are quite large, but they are packed with oxygen and hydrogen atoms bonded in a way that creates many polar hydroxyl (-OH) groups. These polar groups are like little magnets that can form strong attractions with water molecules. Water molecules can surround each sugar molecule, break the bonds holding the sugar together, and pull it apart. That’s why when you stir sugar into your tea or coffee, it just… disappears! It dissolves. So, is Sugar soluble in water? Oh yeah, totally! It’s another classic example of a polar compound dissolving beautifully in water.
Putting It All Together
So, if our hypothetical list was: Sodium Chloride, Methane, Ethanol, Oil, and Sugar, and the question was "Which of the following compounds is soluble (in water)?", our winners would be:
- Sodium Chloride (NaCl) - Because it's ionic.
- Ethanol (C2H5OH) - Because it has a polar OH group and can hydrogen bond.
- Sugar (Sucrose, C12H22O11) - Because it has many polar OH groups.
Methane and Oil would be the ones sitting on the sidelines, not playing ball with water. They’re nonpolar and prefer their own kind.

It’s a bit like a party, isn’t it? Water is the host, and it loves to invite polar and ionic guests. Nonpolar molecules are like the ones who prefer a quieter night in with their own nonpolar friends. And that’s perfectly fine!
Sometimes, there are things called partially soluble compounds, too. They’re a little bit of this and a little bit of that, so they might dissolve a bit, but not completely. It’s like that one friend who comes to the party but leaves after an hour. They were there, but not for the whole shindig.
The real trick is remembering that “like dissolves like” rule. It’s your superpower for understanding solubility. Polar dissolves polar. Nonpolar dissolves nonpolar. Ionic compounds usually dissolve in polar solvents like water. Easy, right?
So, next time you’re faced with a list of chemicals and asked about solubility, just channel your inner chemistry guru. Look for those polar groups, those charges, those ionic bonds. And if all else fails, think about your coffee. Does it dissolve? Great! Likely polar or ionic. Does it just float there looking sad? Probably nonpolar. You're basically a solubility ninja now! Keep that coffee mug handy, you’ve earned another sip.