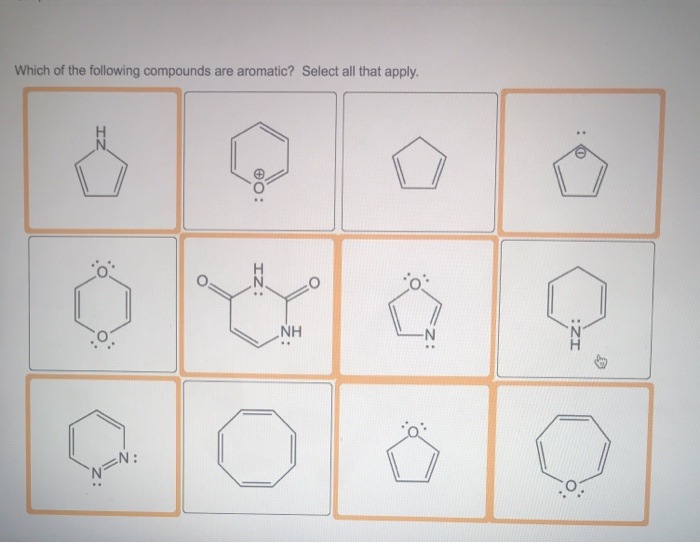
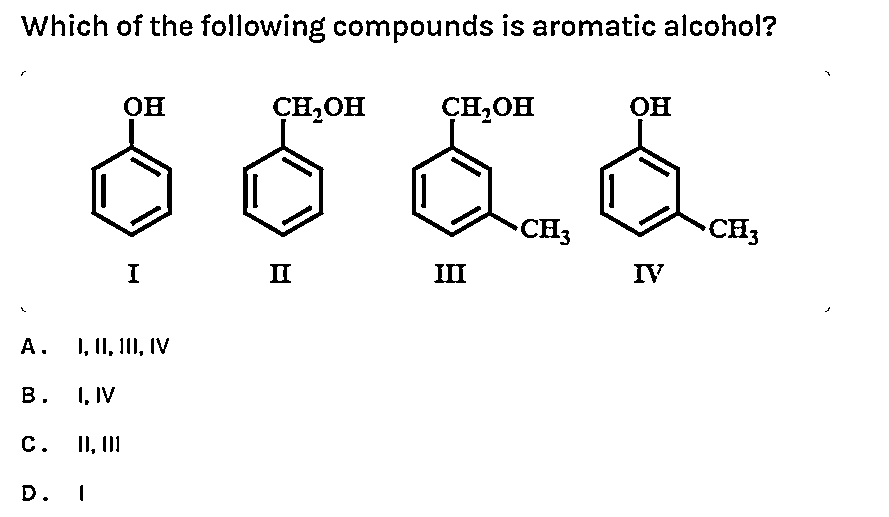
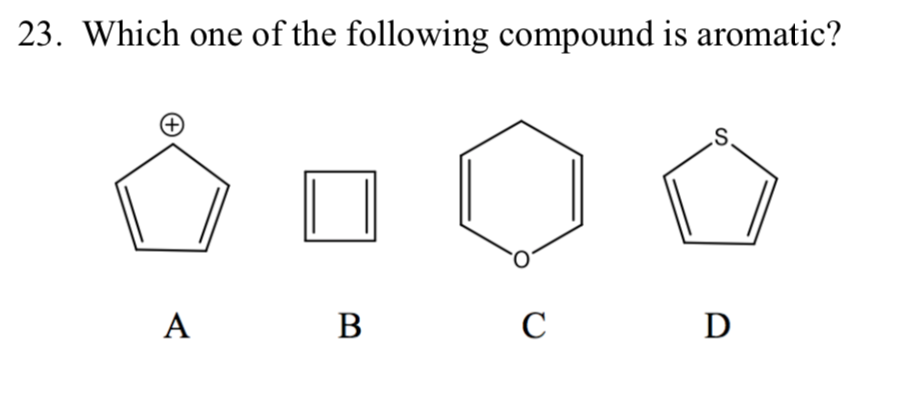
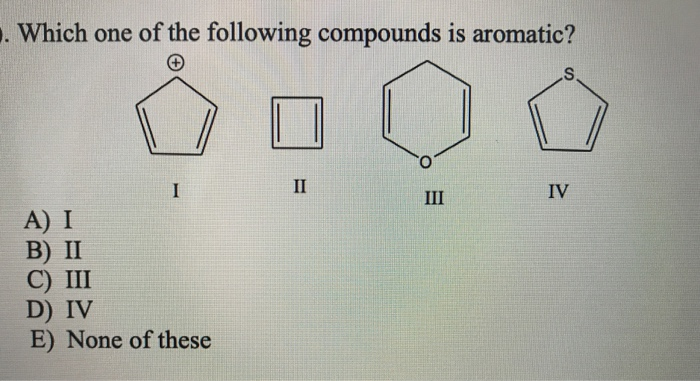
Which Of The Following Compounds Is Aromatic
Okay, so, picture this: I’m rummaging through my old college chem lab notes, you know, the ones that smell vaguely of formaldehyde and desperation. And I stumble upon this doodle. It’s this weird, perfect hexagon with little squiggly lines inside. My roommate, bless his perpetually confused soul, walks by and asks, “Uh, what’s that supposed to be? A really fancy pizza?”
I just sighed. He knew I was deep into organic chemistry at the time, and he still had no clue what any of it meant. It was like trying to explain quantum physics to a goldfish. But that hexagon, that beautiful hexagon, it got me thinking. It was the symbol for something special, something… aromatic. And that, my friends, is where our little adventure today begins. Forget fancy pizza; we’re talking about the true VIPs of the organic chemistry world.
So, the big question that plagues us all, especially after a long night of studying (or, let’s be honest, staring blankly at a textbook), is: which of the following compounds is aromatic? It sounds like a quiz show question, doesn’t it? Like we’re all huddled around, clutching our answer sheets, hoping for that sweet, sweet chemical enlightenment. But it’s more than just a trivia night challenge; it’s about understanding a fundamental concept that shapes so many of the molecules we encounter, from the medicines we take to the flavors we taste.
Must Read
What’s the Big Deal About Aromaticity Anyway?
You see, the word "aromatic" in chemistry doesn't always mean it smells good. I know, right? Big letdown. My roommate was definitely onto something with the pizza comparison in terms of expectations. But in chemistry, aromaticity is all about stability. These molecules are like the calm, collected ones at a party, totally unfazed by the chaos around them. They’ve got this special kind of electron arrangement that makes them remarkably unreactive in certain ways, but incredibly interesting in others.
Think of it as a super-powered, organized electron dance. In these aromatic compounds, the electrons are delocalized, meaning they’re not stuck in one place between two atoms. Instead, they form this continuous cloud of electron density that’s spread out across a ring. This delocalization lowers the overall energy of the molecule, making it much more stable than it would be otherwise. It’s like a well-organized orchestra playing a beautiful symphony, as opposed to a bunch of musicians randomly banging their instruments. You feel the difference, right?
So, when chemists talk about "aromatic," they're talking about this specific electronic structure that confers this extra stability. It’s a property that dictates how these molecules behave in reactions. They tend to undergo substitution reactions rather than addition reactions, which is a whole other can of worms we won’t open today, but just know it’s a big deal!
The Golden Rules of Aromaticity: Hückel’s Magic Number
Now, how do we spot these elusive aromatic creatures? It’s not like they wear a little "A+" badge. Thankfully, there are a set of rules, famously codified by Erich Hückel, that act as our detective guide. If a molecule checks all these boxes, congratulations, you’ve found an aromatic compound! It’s like a secret handshake for molecules.

Here are the key conditions:
- It must be cyclic. Yep, it has to be a ring structure. No open chains allowed to join this exclusive club. Think of a perfectly formed Ouroboros, eating its own tail, but way less dramatic.
- It must be planar. This means all the atoms in the ring lie in the same plane, like a perfectly flat frisbee. If it’s all wobbly and out of shape, it can’t achieve that beautiful electron delocalization. The electrons need a clear path to flow!
- It must have continuous p-orbital overlap. This is the technical bit. Each atom in the ring must have a p-orbital that can overlap with its neighbors, forming that delocalized electron system. This is where the magic happens!
- It must have (4n + 2) pi electrons. Ah, Hückel’s rule! This is the golden number. Here, 'n' can be any non-negative integer (0, 1, 2, 3, and so on). So, the number of pi electrons must be 2, 6, 10, 14, and so on. If it’s 4, 8, 12, etc., then it's probably not aromatic. This is the key differentiator!
It’s a bit like a molecular scavenger hunt, and these four conditions are your clues. If a molecule fits the bill, it’s in the aromatic club. If it misses even one, it’s out. Harsh, I know, but that’s chemistry for you – sometimes it’s all about the strict criteria.
Let’s Meet Some Suspects (and Some Non-Suspects!)
To really drive this home, let’s look at some examples. Imagine you’re presented with a list of compounds. How do you pick out the aromatic one? Let’s put on our detective hats and analyze some common suspects.
Suspect #1: Benzene (The Quintessential Aromatic!)
Ah, benzene. The poster child, the rockstar, the… well, the archetypal aromatic compound. This is the one you’ll see drawn as that hexagon with the circle inside, or alternating double and single bonds. Let’s run it through our Hückel checklist:
- Cyclic? Absolutely. It’s a six-membered ring. Check!
- Planar? Yes, benzene is a perfectly flat molecule. Check!
- Continuous p-orbital overlap? Each carbon atom in benzene has one p-orbital perpendicular to the plane of the ring, and these overlap beautifully to form a delocalized pi system above and below the ring. Check!
- (4n + 2) pi electrons? Benzene has three double bonds. Each double bond contributes two pi electrons to the delocalized system. So, that’s 3 * 2 = 6 pi electrons. Now, let’s plug this into Hückel’s rule: 4n + 2 = 6. If we solve for n, we get 4n = 4, so n = 1. Since n=1 is a valid non-negative integer, benzene fits! Double-check!
So, there you have it. Benzene is indeed aromatic. It’s the standard by which all other aromatic compounds are judged. It’s so stable, it practically smiles at you. (Okay, maybe not smiles, but you get the idea.)
Suspect #2: Cyclooctatetraene (Looks Like it Might Be, But Isn’t!)
This one is a bit of a trickster. It’s a ring, and it has double bonds. You might be tempted to say, “Aha! Aromatic!” Let’s see:
- Cyclic? Yes, an eight-membered ring. Check!
- Planar? Here’s where it stumbles. Cyclooctatetraene is not planar. To minimize electron repulsion and achieve a more stable (though still strained) conformation, it adopts a tub-like shape. So, it fails the planarity test. Fail!
- Continuous p-orbital overlap? Because it's not planar, the p-orbitals don't overlap continuously. Fail!
- (4n + 2) pi electrons? It has four double bonds, so 4 * 2 = 8 pi electrons. If we try to fit this into 4n + 2, we get 4n + 2 = 8, so 4n = 6, and n = 1.5. That's not an integer! So, it fails Hückel's rule too, even if it were planar. Double fail!
Cyclooctatetraene is actually an example of an antiaromatic compound if it could be forced into a planar conformation (which it can't easily do). Antiaromatic compounds are unstable and highly reactive. They have 4n pi electrons in a cyclic, planar system with continuous p-orbital overlap. So, it’s the opposite of aromatic. Not what we’re looking for today!
This is why checking all the criteria is so crucial. Don’t get fooled by just one or two checks!
Suspect #3: Pyridine (The Heterocyclic Gentleman)
Now, let’s introduce a heterocycle. That just means a ring that contains atoms other than carbon. Pyridine is a six-membered ring like benzene, but one of the carbon atoms is replaced by a nitrogen atom. Does that throw a wrench in the works?
- Cyclic? Yes, a six-membered ring. Check!
- Planar? Yes, pyridine is planar. Check!
- Continuous p-orbital overlap? The nitrogen atom in pyridine has a p-orbital that participates in the pi system, just like the carbons. The lone pair of electrons on the nitrogen is in a different orbital (an sp2 orbital) and does not participate in the delocalized pi system. This is important! It still allows for continuous overlap. Check!
- (4n + 2) pi electrons? Pyridine has three double bonds, so 3 * 2 = 6 pi electrons. Just like benzene, 4n + 2 = 6 gives us n = 1. Check!
So, even though it has a nitrogen atom, pyridine is also aromatic! This shows that heterocycles can be aromatic too, as long as they meet the rules. The nitrogen just has to play by the rules, not hog all the electrons or mess up the orbital overlap.
Suspect #4: Cyclopentadiene (The Anion Powerhouse)
This one is interesting because the neutral form isn’t aromatic. But if you remove a proton (making it a cyclopentadienyl anion), things get exciting!
- Cyclic? Yes, a five-membered ring. Check!
- Planar? The anion is planar. Check!
- Continuous p-orbital overlap? The anion has 6 electrons in its p-orbitals. This forms a continuous delocalized system. Check!
- (4n + 2) pi electrons? The anion has 6 pi electrons (two from each double bond, and the two from the negative charge which are in a p-orbital). 4n + 2 = 6 gives us n = 1. Check!
So, the cyclopentadienyl anion is aromatic! It’s a classic example of how gaining or losing electrons can drastically change a molecule’s aromaticity and stability. This little anion is super stable and a common building block in organometallic chemistry. Who knew a negative charge could bring so much peace and harmony?

Why Does This Even Matter in the Real World?
Beyond the satisfaction of solving molecular puzzles, understanding aromaticity is HUGE. Think about:
- Pharmaceuticals: Many drugs contain aromatic rings. Their stability and reactivity profile are crucial for how they interact with biological systems. Benzene itself is a building block for all sorts of important molecules.
- Materials Science: From plastics to dyes, aromatic compounds are everywhere. Their electronic properties, conferred by aromaticity, are exploited in various materials.
- Flavors and Fragrances: While not all aromatic compounds smell good, many important natural and synthetic flavor and fragrance compounds are aromatic. Think of the distinct aroma of vanilla (vanillin) or cinnamon.
- Biochemistry: DNA and RNA bases (like adenine, guanine, cytosine, thymine, and uracil) are all aromatic! This aromaticity contributes to their stability and their ability to stack and interact. Life as we know it would be very different without these stable, ring-like structures.
So, the next time you hear the word "aromatic" in a chemistry context, remember it's not just about a pleasant smell. It's about a stable, delocalized electron system that follows a very specific set of rules. It’s about molecules that have found their perfect energetic groove.
The Takeaway for Your Next Quiz (or Just for Fun!)
When you’re faced with the question, "Which of the following compounds is aromatic?", remember your trusty Hückel rules:
- Is it a ring?
- Is it flat?
- Do the p-orbitals overlap all the way around?
- Are there (4n + 2) pi electrons (where n = 0, 1, 2...)?
If the answer to all four is a resounding "YES!", then you’ve got yourself an aromatic compound. If even one is a "NO," then it’s not aromatic. It might be antiaromatic or just a regular, non-aromatic molecule. And that’s perfectly okay too!
It's a bit like a membership application to a very exclusive, very stable club. Not everyone makes it, but those who do are pretty special. So, the next time you see that hexagon with the circle, or a similar ring structure with the right electron count, give it a nod of recognition. You've just identified a molecule that's mastered the art of stability and electron flow. Pretty cool, huh?