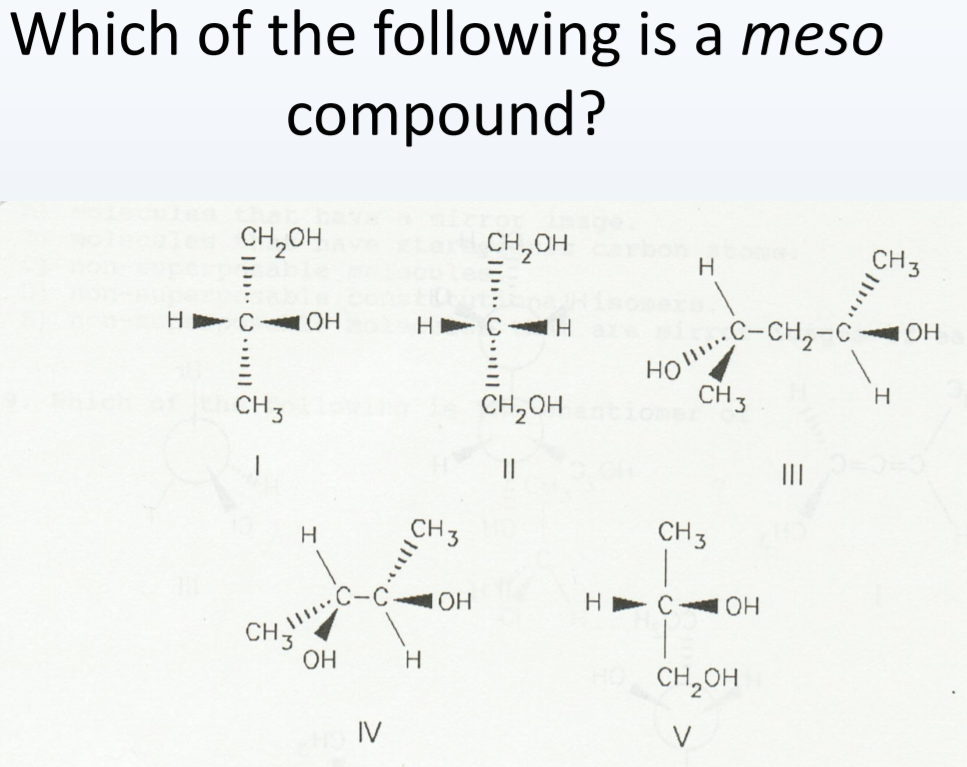
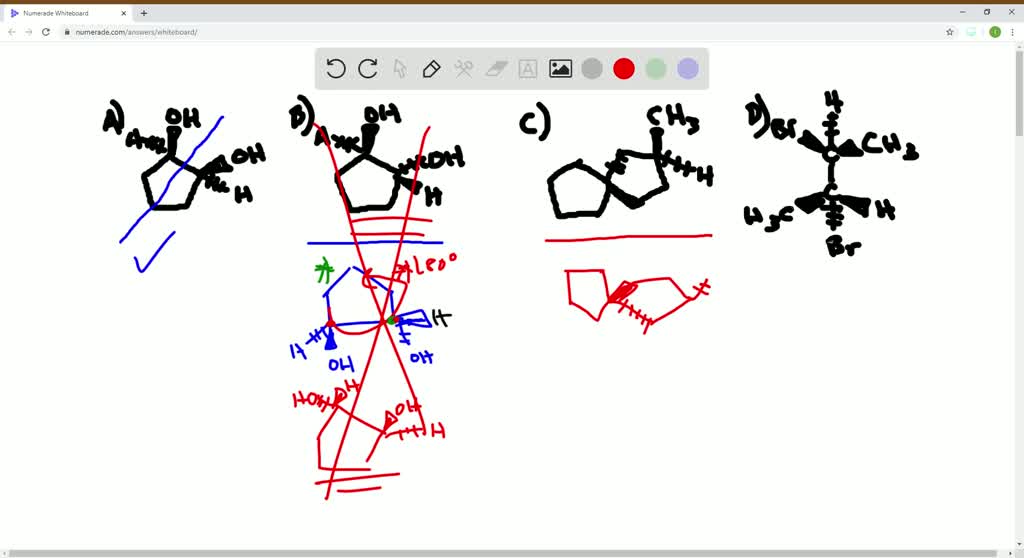
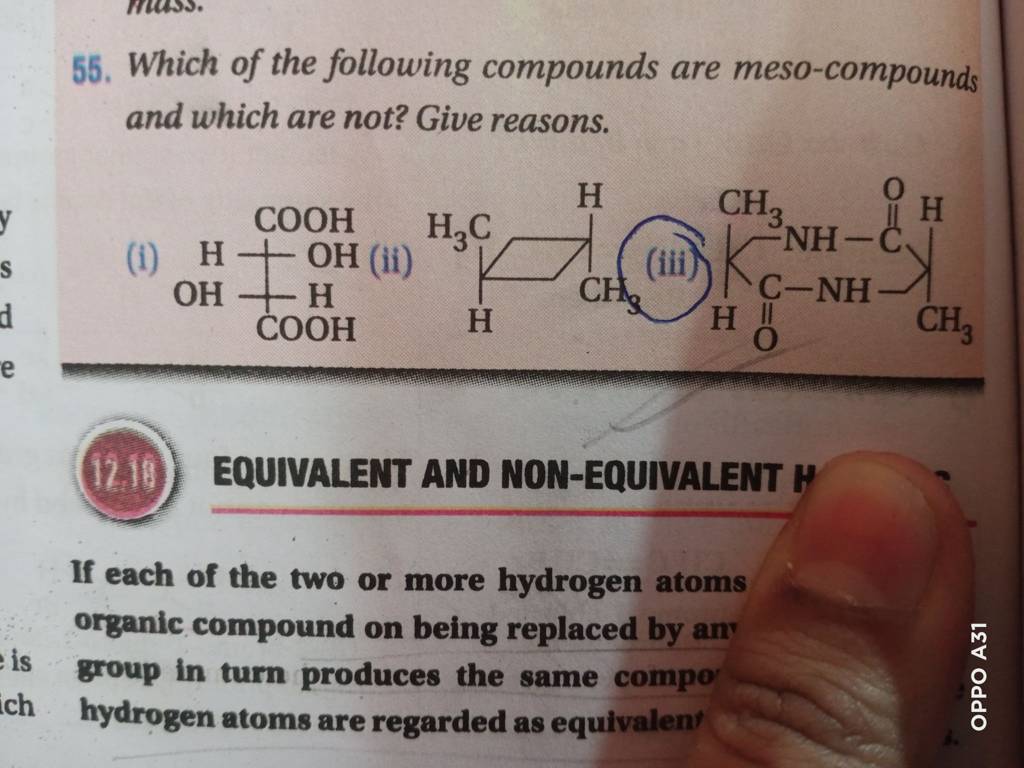
Which Of The Following Are Meso Compounds
Hey there, science explorers! Ever find yourself staring at a molecule and thinking, "What's its deal?" Well, today we're diving into a super cool corner of chemistry: meso compounds. Don't let the fancy name scare you off; it's actually pretty neat, and we're going to break it down in a way that's as relaxed as your favorite comfy chair.
So, what exactly are these meso things? Imagine you've got a molecule, and it's got these special points called chiral centers. Think of them like little hands – they can be "left" or "right," and they're not superimposable on each other. Kind of like your actual hands, right? You can't perfectly stack one on top of the other. Molecules with these chiral centers are called chiral molecules, and they often come in pairs called enantiomers – like mirror images.
Now, here's where it gets interesting. Most of the time, if a molecule has chiral centers, it's going to be chiral itself. It's going to have that whole "left hand" or "right hand" vibe. But, surprise! Sometimes, a molecule can have chiral centers and still be not chiral. How is that even possible? This is the magic of meso compounds!
Must Read
The Secret Symmetry of Meso Compounds
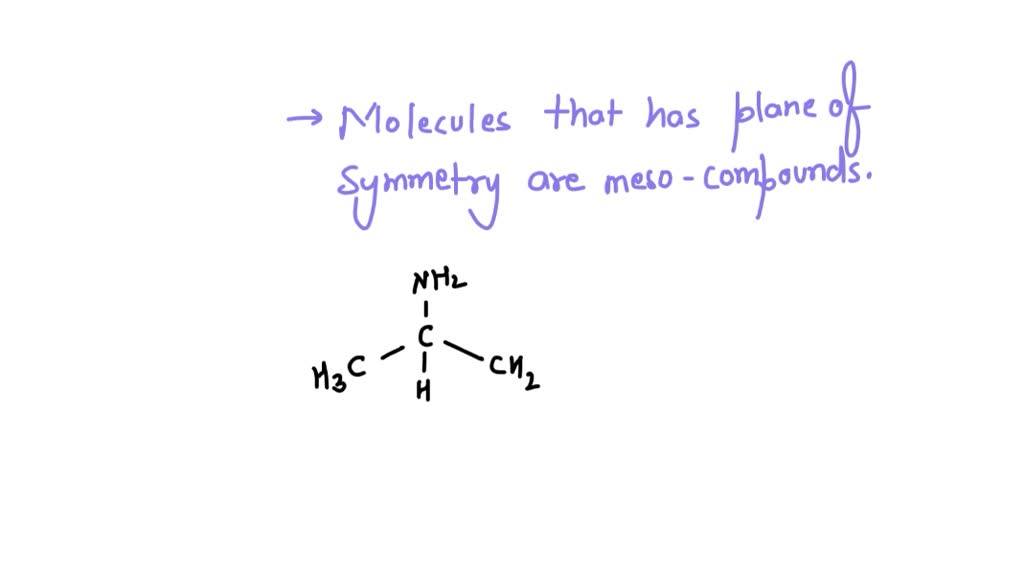
So, how do these sneaky molecules pull off this trick? It all comes down to symmetry. While a meso compound might have multiple chiral centers (those "left" and "right" bits), it also has something special: an internal plane of symmetry. Picture it like a mirror running right through the middle of the molecule. If you can slice it perfectly in half with a mirror, and one half is the exact mirror image of the other, then BAM! You've likely got yourself a meso compound.
Think of it like this: Imagine you have two identical twins, but one is a mirror image of the other. If they stand next to each other in a perfectly symmetrical room, the whole setup might look the same whether you look at it from the left or the right. That's a bit of an oversimplification, of course, but it gives you the general idea. The molecule, despite having these "handed" parts, has a balanced, symmetrical arrangement that makes the whole thing achiral (not chiral).
Why is this a big deal?
You might be wondering, "Okay, cool symmetry, but why should I care?" Well, in the world of chemistry, especially when it comes to how molecules interact with other molecules (like in our bodies!), chirality is a huge deal. Many biological processes are super specific about which "handedness" of a molecule they interact with. Think of a lock and key – the key has to have the right shape to fit. If a drug molecule has a chiral center, one enantiomer might be a super effective medicine, while the other might do nothing, or even worse, have harmful side effects!
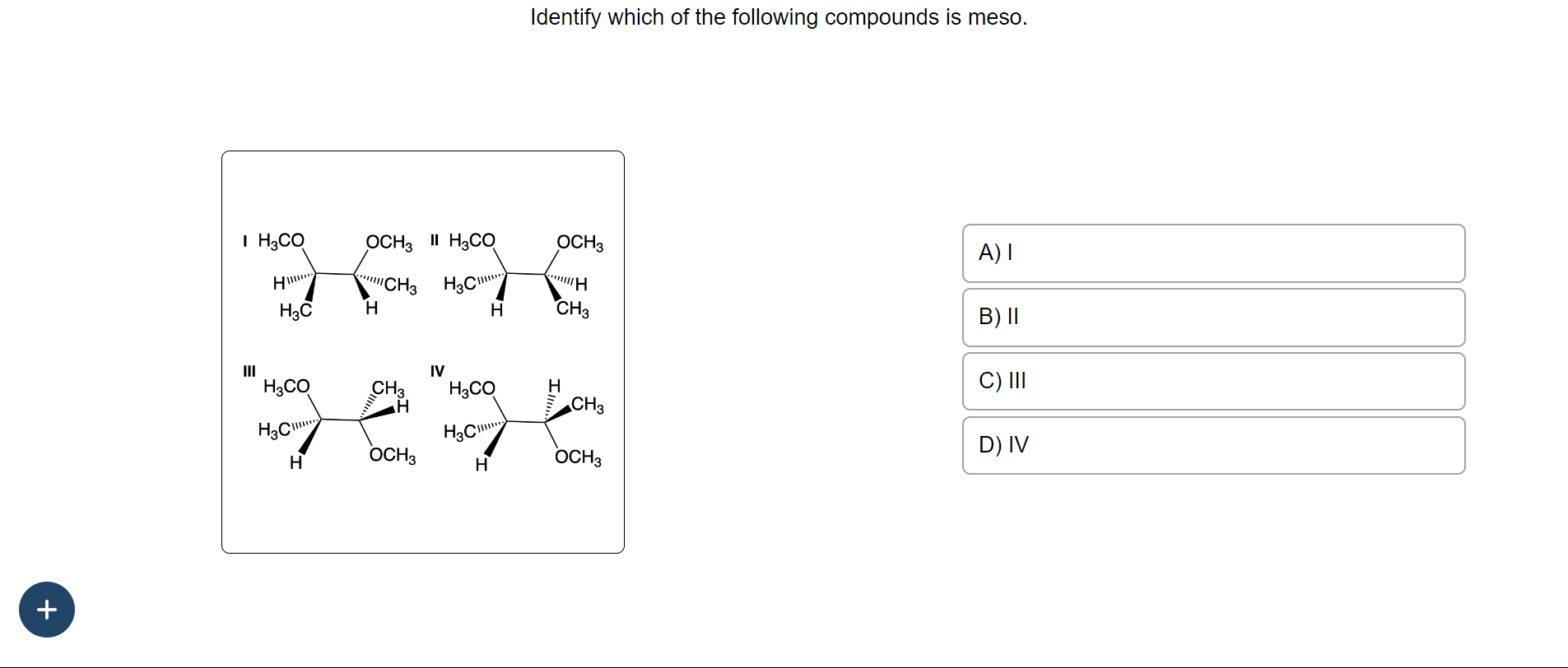
Meso compounds, being achiral, don't have this "handedness" problem. They're like a universal key that can fit into many locks, or rather, they don't have a specific handedness that causes issues. This can be super important in pharmaceuticals and other areas where precise molecular interactions are key.
Spotting a Meso Compound: Your Detective Toolkit
So, how do you become a meso compound detective? Here are a few clues:
First, you need to identify the chiral centers. These are usually carbon atoms that are bonded to four different groups. Keep an eye out for those!

Next, and this is the crucial part, you need to check for that internal plane of symmetry. This can be a bit trickier. You'll often find it in molecules that have a symmetrical backbone. Imagine a symmetrical shape, like a rectangle or a perfectly balanced dumbbell, with those chiral centers attached in a mirrored fashion.
Let's look at a classic example: tartaric acid. It's a common compound found in wine and grapes. Pure, naturally occurring tartaric acid is actually chiral. But, there's a specific form, often referred to as meso-tartaric acid, that has two chiral centers but possesses an internal plane of symmetry. Because of this symmetry, the molecule as a whole is achiral, making it a meso compound. It's like having two hands that are mirror images, but they're held together in such a way that the entire structure is symmetrical.
Another way to think about it is that if you have a pair of enantiomers (those mirror-image chiral molecules) and you can somehow "flip" one to become identical to the other without breaking any bonds, and that flipping involves a symmetrical arrangement, then you're probably dealing with a meso situation. It's a bit like how some optical illusions work – your brain sees something, but there's an underlying symmetry that makes it all balance out.

What about the opposite?
On the flip side, if a molecule has chiral centers and no internal plane of symmetry, it's going to be chiral. It'll have those distinct "left" and "right" forms (enantiomers), and those are the ones that can behave differently in biological systems. For instance, something like lactic acid can exist as two enantiomers, L-lactic acid (the one produced in your muscles during exercise) and D-lactic acid. They're mirror images, and our bodies interact with them differently.
So, the key takeaway is this: meso compounds are the "symmetry superheroes" of the chiral world. They've got the potential for chirality within their structure (those chiral centers), but their overall symmetry cancels out that handedness, making them achiral. It’s a bit like having a complicated dance routine with two dancers who are mirror images, but the overall choreography is perfectly balanced and symmetrical.
The "Which of the Following" Puzzle
Now, if you were presented with a question like "Which of the following are meso compounds?", you'd be looking for molecules that fit this description. You'd examine each option, identify any chiral centers, and then meticulously check for that internal plane of symmetry. It's like a fun puzzle where you're the detective, and symmetry is your main clue!
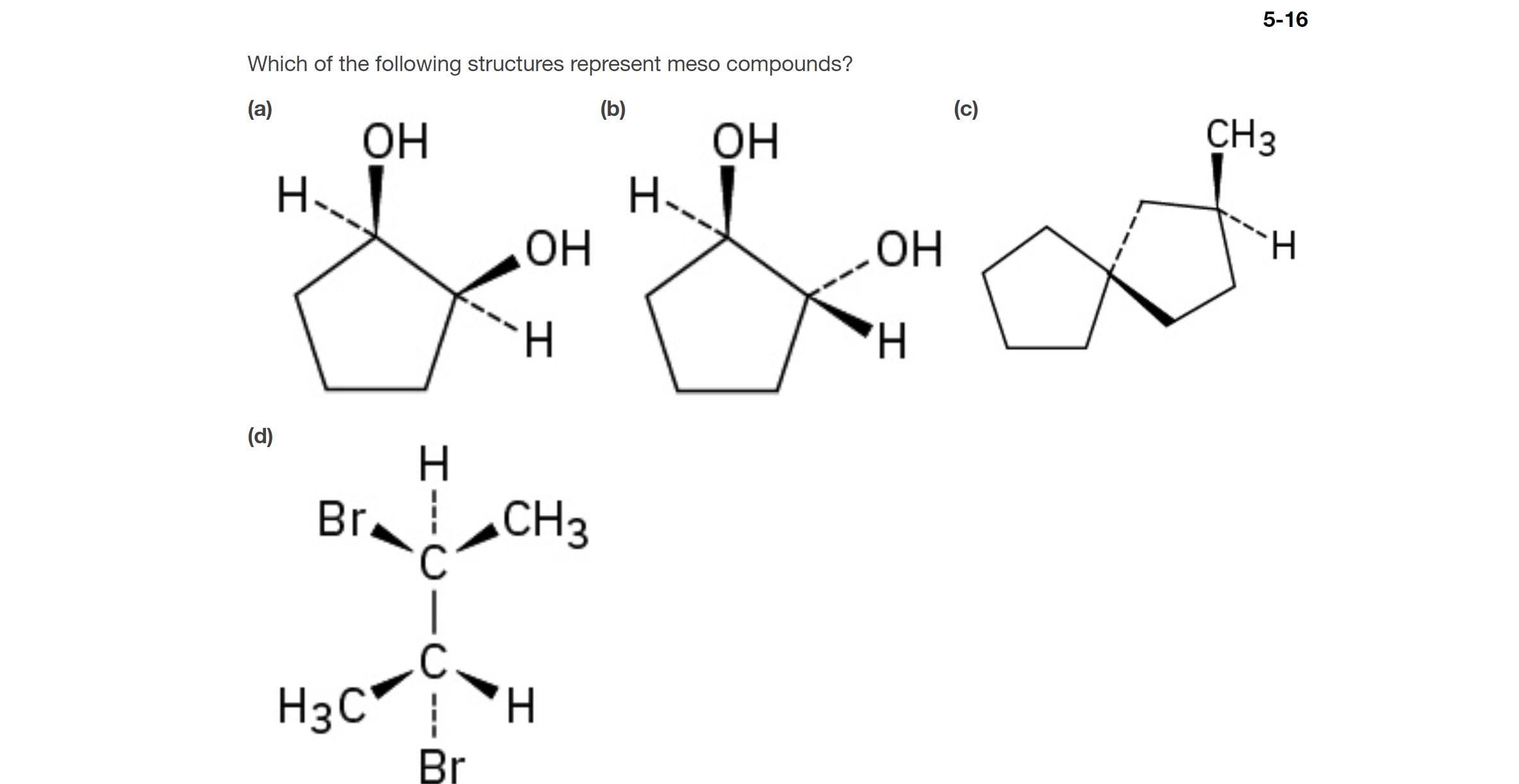
It’s also important to note that sometimes a molecule might have multiple chiral centers, and some of the possible stereoisomers might be meso, while others are chiral. This is why careful examination of each specific molecule's structure is so important. It's not just about counting chiral centers; it's about how those centers are arranged in space.
Understanding meso compounds isn't just about passing a chemistry test; it’s about appreciating the subtle yet significant ways molecular structure dictates function. It highlights how nature, and chemistry, can be incredibly clever and sometimes surprising in its designs. So next time you see a molecule, remember to look beyond just the building blocks and consider the elegant symmetry that might be hiding within!
Keep exploring, keep questioning, and embrace the cool complexities of the molecular world. Happy molecule hunting!