Which Of Dalton's Postulates Are Incorrect

Hey science fans and curious minds! Ever heard of John Dalton? This guy was like the OG of atomic theory, dropping his amazing ideas way back in the early 1800s. He basically told us what matter was made of, and for a long time, everyone was like, "Wow, Dalton, you're a genius!"
Think of it like this: Dalton was the first person to say, "Hey, everything is made of tiny, tiny balls called atoms!" It was a revolutionary idea that explained so much about how stuff worked. He was the Sherlock Holmes of the microscopic world, piecing together clues about the fundamental nature of reality.
So, Dalton, being the super-smart dude he was, came up with a list of these brilliant postulates, or rules, about atoms. These were like the Ten Commandments for the atomic age, guiding scientists for decades. They were so good, they practically became gospel.
Must Read
But you know how things go, right? Even the best ideas can get a little… updated. As science zoomed forward with faster microscopes and cooler experiments, we started noticing a few little snags in Dalton's perfect picture. It turns out, the atom isn't quite the un-dividable little billiard ball he imagined.
Let's dive into the fun parts, the bits where Dalton's brainy postulates took a little detour. Prepare for some atomic eyebrow-raising!
The Atom Isn't Quite So "Un-dividable"!
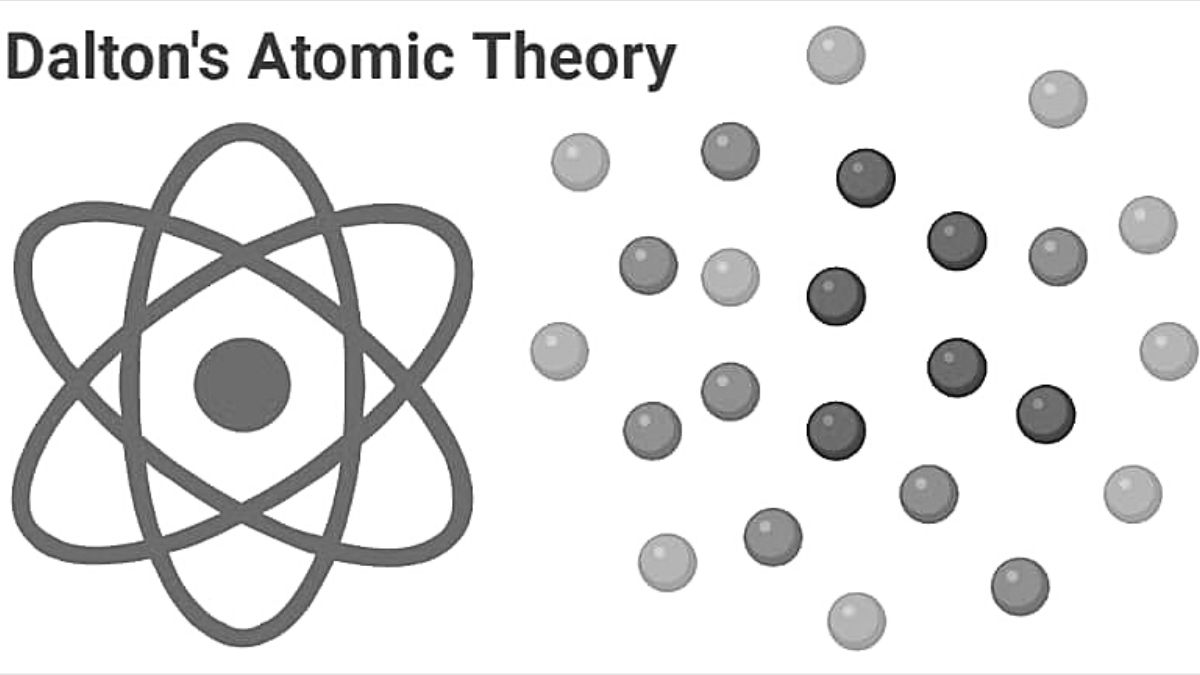
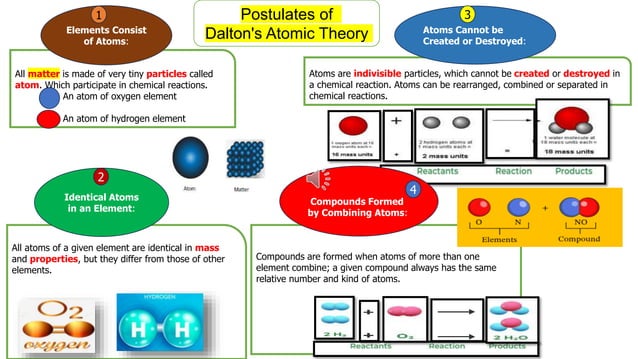
Okay, so Dalton's first big pronouncement was that atoms are indivisible. Like a tiny LEGO brick that you absolutely, positively cannot break down any further. He probably imagined them as these solid, indestructible little spheres.
Imagine trying to break a diamond with your bare hands. That's kind of the vibe Dalton was going for with his atoms! Nothing, not even the most powerful force, could split them. It was a bold statement, and for its time, it made perfect sense.

But then, BAM! Along came folks like J.J. Thomson, who discovered the electron. This was like finding out that your diamond, the supposedly unbreakable thing, actually had tiny little crumbs falling off it! The electron was a tiny, negatively charged particle.
And then, even more mind-blowing, Ernest Rutherford discovered the nucleus! This showed that the atom had a super-dense, positively charged center. So, not only could atoms be split, but they had whole internal structures! It’s like discovering your LEGO brick has a tiny, buzzing motor inside.
So, Dalton's idea that atoms are indivisible? Turns out, they're actually made of even smaller things: protons, neutrons, and electrons. We can split atoms, especially in things like nuclear power plants or bombs (though let's focus on the power plant for now!). It's a bit like discovering that a perfectly formed cookie can be broken into delicious crumbs, and then those crumbs are made of flour, sugar, and chocolate chips. Who knew?!
Atoms of the Same Element Aren't Identical After All!
Here's another one that got a little twist. Dalton also said that all atoms of a given element are identical. So, every single atom of gold is exactly the same, down to its tiny atomic core. Think of it like perfectly manufactured identical twins for every element.

If you had an atom of oxygen, it was an oxygen atom, and there was no other kind of oxygen atom. Period. End of story. This made chemical reactions super predictable and tidy. It was like having a set of perfectly matched socks for every occasion.
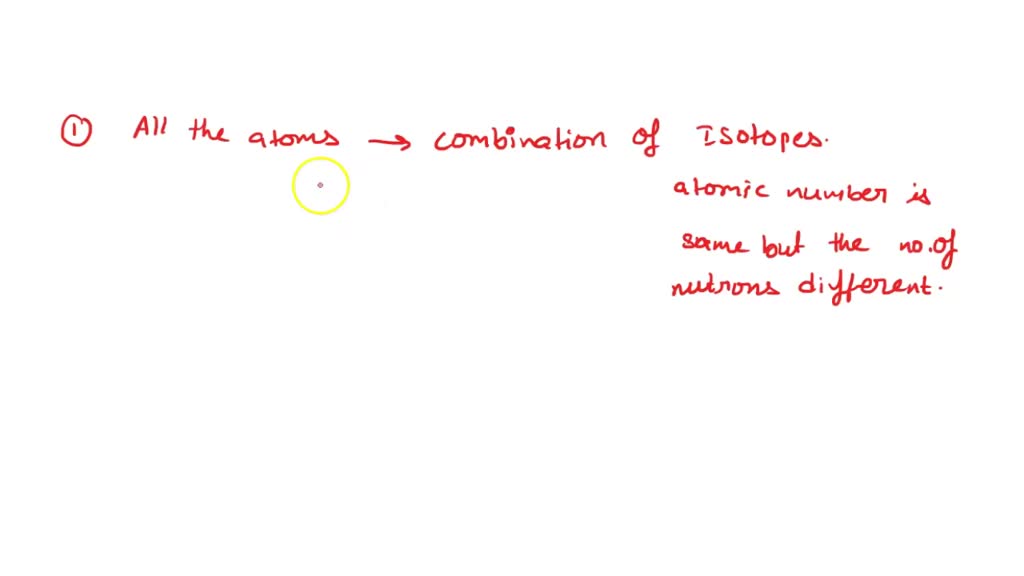
But then, surprise, surprise! Scientists discovered isotopes. These are atoms of the same element that have a different number of neutrons. So, while they have the same number of protons (which defines the element), they weigh a little differently. It's like having identical twins, but one of them had an extra helping at breakfast!
Imagine you have two apples from the same tree. They look the same, right? But maybe one apple is slightly sweeter, or a tiny bit larger. That's kind of like isotopes! They're both oxygen, but they have a little variation.
This discovery meant that while atoms of an element are very similar, they aren't always perfectly identical. It added a layer of complexity and nuance to the atomic world, proving that nature loves a little bit of variety. It’s like finding out that even though all your white socks look the same, some are just a touch softer.

Atoms Can Change into Other Atoms?!
And here’s a doozy! Dalton's fourth postulate stated that atoms cannot be created or destroyed in chemical reactions. They just rearrange themselves, like shuffling a deck of cards. You can't magic new cards into existence, and you can't make them disappear.
This was a cornerstone of his theory, explaining conservation of mass – that in a chemical reaction, the total mass of what you start with is the same as the total mass of what you end up with. It was a very neat and tidy principle. No atoms were harmed, or created, in the making of any chemical reaction.
However, while this is mostly true for everyday chemical reactions, we’ve since discovered things like radioactive decay and nuclear reactions. In these processes, atoms can transform into atoms of different elements! It’s like watching one card in your deck spontaneously turn into a completely different card from another deck. Whoa!
Think about that glowing rock you might see in a science fiction movie. That glow is often from radioactive decay, where an atom is literally changing into a different kind of atom over time. It's a far cry from simply rearranging them!
So, while Dalton's idea holds for typical chemical changes, the universe has shown us that atoms can indeed be… well, transmuted. It's like discovering that a magician can not only shuffle cards but also make them transform into playing cards from a different game entirely. Talk about a plot twist!
Why Does This All Matter?
Now, before you start thinking Dalton was totally wrong, let's give him a huge round of applause! His postulates were brilliant for their time and laid the absolute groundwork for everything we know about chemistry and physics today. He was the spark that ignited the scientific revolution of the atom!
It's not about being wrong; it's about how science evolves. Every great idea becomes a stepping stone for even greater discoveries. We build upon the foundations laid by giants like Dalton. His work was so good, it took scientists generations to even begin to poke holes in it.
So, the next time you hear about Dalton's postulates, remember that science is a journey of discovery. It's about asking questions, testing ideas, and constantly refining our understanding of the universe. And sometimes, those refinements lead to some pretty mind-blowing revelations about the tiny, invisible world that makes up everything around us. Pretty cool, right?