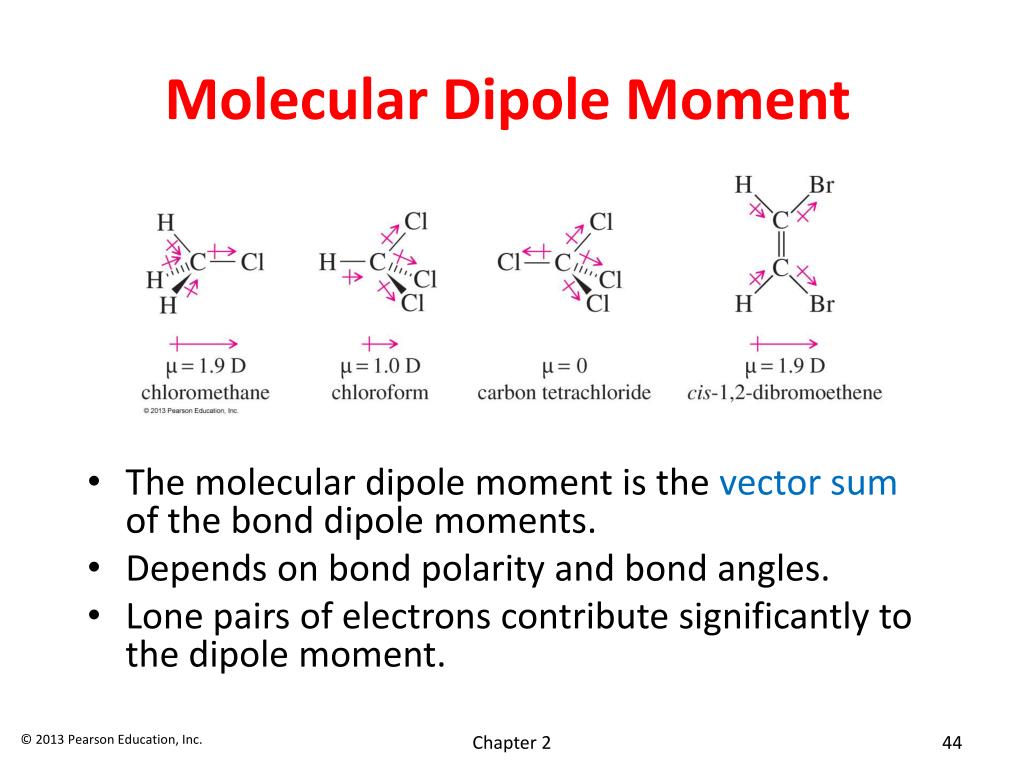
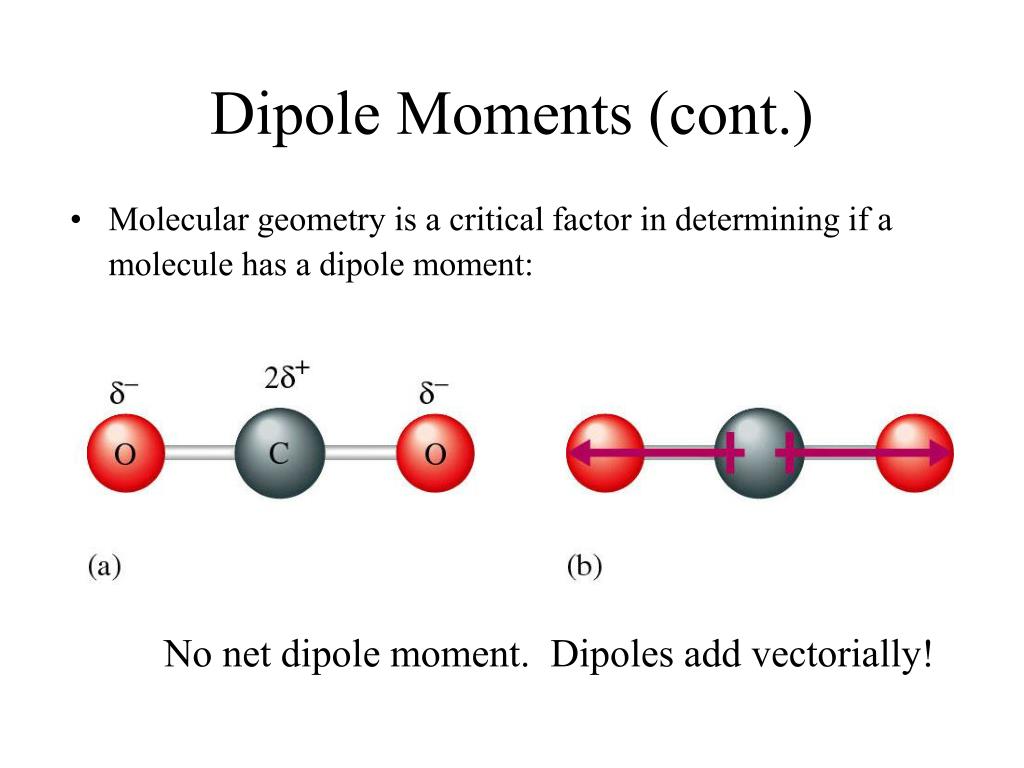
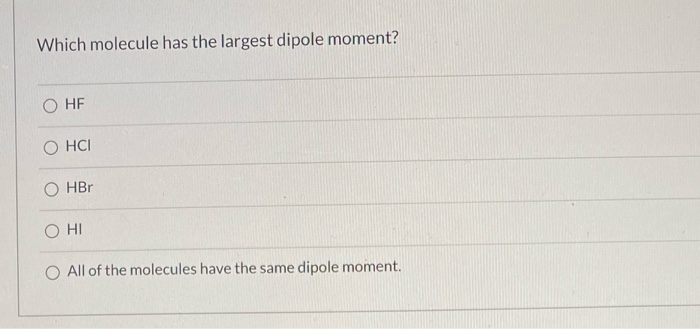
Which Molecule Has The Largest Dipole Moment

We're about to dive into the microscopic world. It's a place full of tiny things doing big jobs. Think of it like a bustling city, but made of atoms and molecules.
Today, we're on a quest. A grand adventure to find the king of the dipole moment. Yes, you heard me right, the dipole moment. It sounds fancy, I know. But stick with me, it's more fun than it sounds.
Imagine a molecule. It's like a tiny LEGO creation. Some of these LEGOs have a little bit of a personality. They're not perfectly balanced. One side might be a bit more "popular" than the other.
Must Read
This "popularity" is what we call a dipole moment. It's like a tiny, invisible magnet. One end is a little bit positive, and the other end is a little bit negative. They're not fully charged like a battery, just a gentle nudge.
So, which molecule wins the crown? The one with the biggest, most enthusiastic nudge? The one that's most definitely not sharing its snacks equally?
Now, you might be thinking, "This is getting a bit technical." And you'd be right. But we're keeping it light. Think of it as gossip for the chemical world.
There are many contenders for this prestigious title. Many molecules strutting their stuff with their unevenly distributed electrons. Some are shy about their differences. Others are quite bold.
One molecule that often gets mentioned is hydrogen fluoride (HF). It's a classic. It's like the "popular kid" of introductory chemistry. Fluorine is a bit of a diva, hogging the electrons. This gives HF a noticeable dipole moment.
But is it the largest? Ah, that's the million-dollar question. Or rather, the tiny fraction of a micro-Debye question.

Then we have molecules with multiple "popular" spots. Think of a molecule with a few atoms that really like to pull electrons their way. This can lead to some interesting dipole situations.
Consider water (H2O). It's a bendy molecule. The oxygen atom is like the popular kid in the middle, attracting all the attention. The hydrogens are a bit further out. This V-shape gives water a significant dipole moment.
Water is pretty amazing. It’s involved in so many things. Its dipole moment is a big reason why. It helps water dissolve all sorts of other things. It's the ultimate social butterfly of solvents.
But is water the absolute champion? The undisputed heavyweight of the dipole world? We're still searching.
Now, let's talk about some outliers. Some molecules that really push the boundaries. They're not afraid to be different. They embrace their unevenness.
What about molecules with very electronegative atoms? These are the "electron hoarders." They just can't help but grab more than their fair share. Fluorine is the prime example. Chlorine and oxygen are also pretty good at it.

When these electron hoarders are paired with atoms that are more generous with their electrons, you get a big difference. A huge tug-of-war for those tiny electrons.
So, what about molecules that are really, really asymmetric? Molecules that have one end that's practically screaming "I'm negative!" and the other end is just a whisper of "I'm slightly less positive."
This is where things get exciting. This is where we might find our winner.
Let's consider some of the heavy hitters in the world of electronegativity. We've already mentioned fluorine. It's the undisputed queen of electron-grabbing.
What happens when you have a molecule where fluorine is on one end, and something much less electronegative is on the other? Something like a simple hydrogen atom, but in a more complex arrangement?
The answer, my friends, might surprise you. Or maybe it won't. We're going on a gut feeling here, an "unpopular opinion" kind of vibe.
While many textbooks will point to certain common molecules, I have a sneaking suspicion about a particular class of compounds. They're often overlooked in these general discussions.
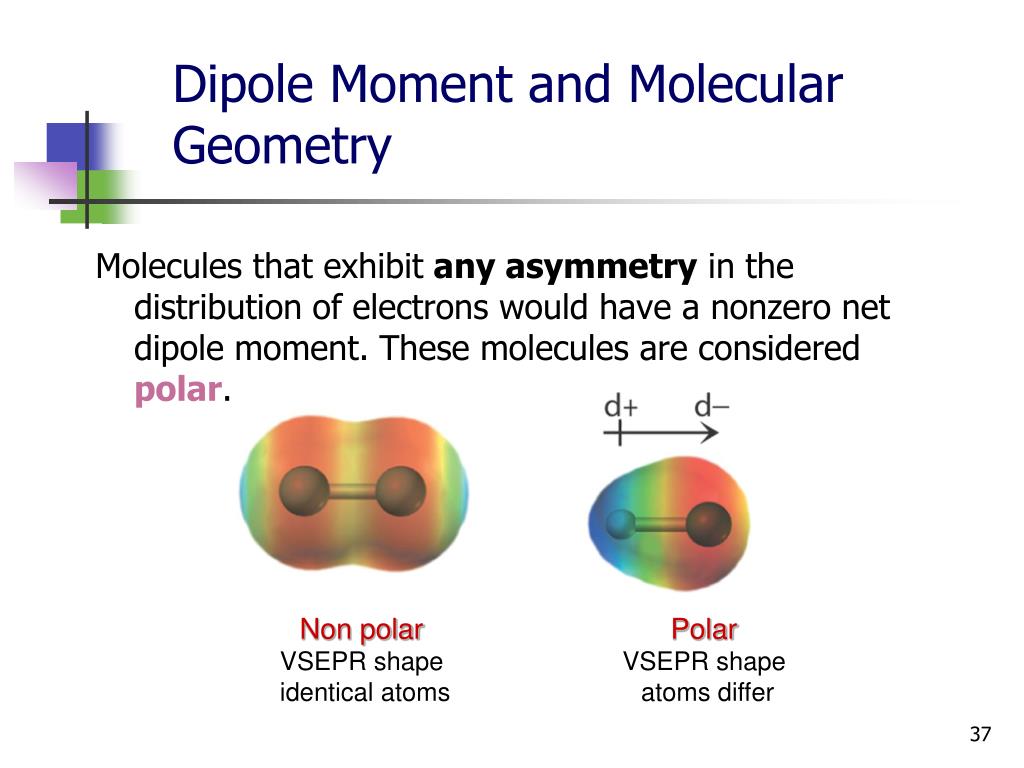
Think about molecules with a very strong central atom that's highly electronegative, and it's surrounded by less electronegative atoms in a way that creates a very lopsided charge distribution. It's all about the geometry, you see.
Here's my personal, slightly controversial pick. Drumroll, please... It's the cyanogen halides! Specifically, molecules like cyanogen bromide (BrCN) or cyanogen iodide (ICN).
Now, why these guys? They're not as cuddly as water, are they? They're a bit more... intense.
In these molecules, you have a carbon atom. It's bonded to a nitrogen atom, which is very electronegative. Then, it's bonded to a halogen like bromine or iodine.
The nitrogen is pulling electrons hard. The carbon is also involved in a triple bond, which is pretty electron-dense. And then you have the halogen, which, while less electronegative than nitrogen, still contributes to the overall pull.
The way these atoms are arranged creates an incredibly strong polarization. The negative end is quite negative, and the positive end is quite positive.

It's like a tiny, incredibly dedicated tug-of-war team. They are really committed to pulling those electrons.
So, while water is a fantastic socialite with a lovely dipole, and hydrogen fluoride is a strong contender, I'm leaning towards the cyanogen halides for their sheer, unadulterated charge separation.
They're the underdogs, the unexpected champions. They're the molecules that say, "Yeah, we have a dipole moment, and it's HUGE!"
It's an unpopular opinion, perhaps. But I stand by it. Sometimes, the most electrifying personalities are found in the most surprising places.
These molecules might not be in your everyday coffee cup, but they are certainly making a splash in the world of chemistry with their impressive dipole moments. They’re the silent, powerful forces shaping how molecules interact.
So, next time you think about molecular personalities, remember the cyanogen halides. They’re not just chemicals; they're tiny powerhouses of polarized potential.
And that, my friends, is my ode to the mightiest dipole moment. A little bit of science, a lot of personality, and a whole lot of fun.