Which Metal Dissolves In Hno3 But Not In Hcl

Hey there, fellow curious minds and home experiment enthusiasts! Ever found yourself staring at a pile of shiny metal scraps, maybe from that DIY project that went slightly sideways, and wondered, "What's the deal with these guys and acids?" It’s a question that might pop up while you're sketching out your next masterpiece or even just tidying up the garage. We're diving into the fascinating world of metals and their reactions, specifically focusing on a little chemical puzzle: which metal loves nitric acid but gives hydrochloric acid the cold shoulder?
This isn't your typical, stuffy science lecture. Think of it as a chill chat over a virtual cup of coffee, exploring how different elements behave. We’ll be sprinkling in some real-world connections, a dash of historical intrigue, and maybe even a hint of why this stuff matters beyond the lab coat.
The Unlikely Suspects: A Tale of Two Acids
So, let’s set the scene. We’ve got two heavyweight acids in our corner: nitric acid (HNO₃) and hydrochloric acid (HCl). Both are known for their corrosive properties, but they have very different personalities when it comes to interacting with metals. It's like choosing between a fiery, passionate artist (nitric acid) and a more reserved, steady friend (hydrochloric acid).
Must Read
Hydrochloric acid, bless its heart, is pretty straightforward. It’s a common workhorse, found in everything from stomach acid (yes, your own body makes it!) to industrial cleaning agents. It tends to react with metals that are more reactive than hydrogen. Think of it as a metal selector: if you’re not on its list, it just walks on by.
Nitric acid, on the other hand, is a bit more dramatic. It’s a powerful oxidizing agent, meaning it’s really good at stealing electrons. This makes it capable of dissolving metals that hydrochloric acid can't even budge. It’s the metallurgist's drama queen, ready to tackle the tough jobs. But, and this is a big "but," its reaction can get a bit complicated, sometimes producing different byproducts depending on the concentration and the metal involved. It’s like a complex perfume, with layers of scent that reveal themselves over time.
Enter the Star: The Metal with a Preference
Now, the million-dollar question: which metal fits this particular bill? The answer, my friends, lies with a group of metals known as the noble metals. These are the divas of the periodic table, characterized by their resistance to corrosion and oxidation. Think of them as the ultimate trendsetters, refusing to tarnish or degrade easily.
Within this noble clique, one stands out for this specific acid-versus-acid showdown: copper (Cu). That’s right, good ol’ copper! It’s everywhere, from your electrical wiring and plumbing to the beautiful patina on old statues like the Statue of Liberty.
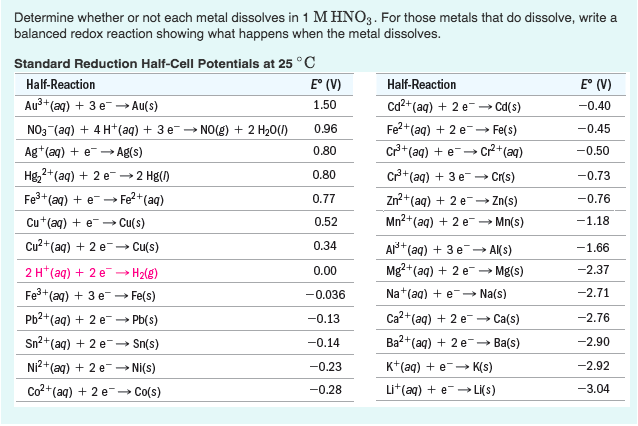
So, why copper? Let's get a little granular. When you expose copper to hydrochloric acid, not much happens. HCl just isn't strong enough, nor does it have the right chemical "oomph," to break down the strong bonds within the copper. It’s like trying to convince a seasoned minimalist to embrace clutter – it’s just not their style.
However, introduce copper to nitric acid, especially a concentrated solution, and bam! A reaction occurs. The nitric acid’s oxidizing power is the key. It attacks the copper, dissolving it and transforming it into copper ions. This is where things get visually interesting: you’ll often see a distinct color change, typically a beautiful blue or green solution, and sometimes the release of brown fumes of nitrogen dioxide (NO₂). It’s a mini-fireworks display in a beaker!
Beyond the Beaker: Copper's Place in Our World
This isn't just some abstract chemistry fact. Understanding how copper reacts with acids has played a significant role in history and continues to be relevant today. Think about the ancient world. The ability to extract and work with metals like copper was a game-changer, marking the beginning of the Copper Age. Imagine early humans discovering that certain rocks, when heated, yielded this malleable, useful metal.
And the artistic applications! That gorgeous verdigris patina that forms on copper roofs and sculptures? That’s often a result of slow reactions with atmospheric elements, including acidic rain. It's nature's way of decorating with copper, adding character and a sense of history. It's a look that interior designers and architects are still trying to replicate!
A Little Fun Fact:
Did you know that the distinctive green color of the Statue of Liberty is due to the oxidation of its copper skin? It took about 20 years for the statue to develop its full patina. So, when you see that iconic landmark, you're looking at a testament to copper's enduring nature and its fascinating chemical transformations!
In the realm of art conservation, knowing these reactions is crucial. If a bronze sculpture (an alloy primarily of copper and tin) needs cleaning or restoration, conservators need to be extremely careful about which chemicals they use. Using the wrong acid could damage the artwork irreparably, turning a priceless artifact into a chemical experiment gone wrong.
The "Why It Matters" Section (Even If You're Not a Chemist)
Okay, so you might be thinking, "This is all well and good, but how does this affect my Netflix binge or my grocery run?" Fair question! This seemingly niche bit of chemistry is actually a foundational concept that underpins many technologies and processes we rely on.
Material Science: Understanding metal reactivity is key to developing new alloys with specific properties. Need a metal that won't corrode in saltwater? Or one that's incredibly strong yet lightweight? Chemists and material scientists are constantly experimenting with how different elements interact, including their behavior in acidic environments. This is how we get everything from corrosion-resistant pipes in your home to the super-strong alloys used in aircraft.
Environmental Chemistry: Acid rain, a phenomenon caused by pollution, can have detrimental effects on metals in our environment, from bridges to historical monuments. Knowing how different metals react to acidic conditions helps us predict and mitigate damage. It also informs how we manage industrial waste and prevent environmental contamination.
![Chemical Properties of Metals [with Reaction Examples] - Teachoo](https://cdn.teachoo.com/9e7bb7eb-ed68-4326-90ae-3382b68b6008/reaction-of-metals-with-dil.-hno3---teachoo.png)
Everyday "Oops" Moments: Ever accidentally mixed cleaning products and gotten a weird smell or a bubbling reaction? While we strongly advise against intentionally mixing household chemicals (seriously, don't do it!), understanding basic chemical principles can make you more aware of potential hazards. For example, many drain cleaners contain acids, and if you've used one, you wouldn't want to pour another type of cleaner on top without knowing what might happen.
A Practical Tip for the Curious Homebody:
If you're genuinely interested in seeing these reactions in a safe and controlled manner, consider getting a starter chemistry kit. Many kits come with diluted acids (like diluted acetic acid, the kind in vinegar!) and safe metal samples. Always, always follow the instructions, wear safety goggles, and work in a well-ventilated area. This is a great way to observe the principles we're discussing firsthand, turning abstract knowledge into a tangible experience. And who knows, you might discover a hidden passion for chemistry!
Cultural Connection: Alchemy and the Quest for Gold
For centuries, alchemists were obsessed with transforming base metals into gold. While they never succeeded in that particular goal, their experiments laid the groundwork for modern chemistry. They were essentially exploring the very reactivity we're talking about! Imagine an alchemist, hunched over a retort, trying to dissolve lead (a base metal) in various concoctions, hoping for a glimmer of golden transformation. The distinction between metals that dissolved in one acid but not another would have been a crucial clue in their complex, often mystical, research.
Their pursuit, though often shrouded in esotericism, was driven by a fundamental question: can we change one element into another? While transmutation as they envisioned it is not possible, their relentless experimentation pushed the boundaries of knowledge about chemical reactions and the properties of matter.

The Humble Penny Test (A Quick and Easy Illustration)
Here’s a simple, albeit not perfectly controlled, experiment you might even be able to relate to. Old copper pennies minted before 1982 (in the US) are mostly copper. Newer ones are mostly zinc with a copper plating. If you were to carefully expose a pre-1982 penny to a dilute nitric acid solution (again, safely and with proper precautions!), you'd see it react and dissolve. However, trying that with a dilute hydrochloric acid solution would likely yield very little, if any, reaction.
It’s a small illustration of the principle: different acids have different strengths and reactivities, and not all metals are created equal in their chemical resistance.
A Moment of Reflection: The Beauty of Nuance
Isn't it fascinating how even something as seemingly simple as two liquids reacting with a solid can reveal such depth and complexity? This specific chemical dance between copper, nitric acid, and hydrochloric acid is a perfect metaphor for life. We all encounter different "acids" – challenges, relationships, opportunities – and our reactions, like those of the metals, depend on our inherent nature and the specific "environment" we're in.
Some situations might dissolve us, while others leave us fundamentally unchanged, perhaps even stronger. Some experiences might create beautiful, new patinas on our character, while others are simply shrugged off. The world of chemistry, in its intricate dance of elements and compounds, often mirrors the subtle yet profound nuances of our own daily lives.
So, the next time you see a copper pipe, a bronze statue, or even just a shiny penny, take a moment to appreciate the invisible forces at play. It’s a reminder that there’s always more going on beneath the surface, a constant interplay of reactions shaping the world around us, one atom at a time.

![Chemical Properties of Metals [with Reaction Examples] - Teachoo](https://cdn.teachoo.com/12cc88d3-864a-4dac-9690-e6fe9427b830/reaction-of-magnesium-with-dil.-hno3---teachoo.png)



