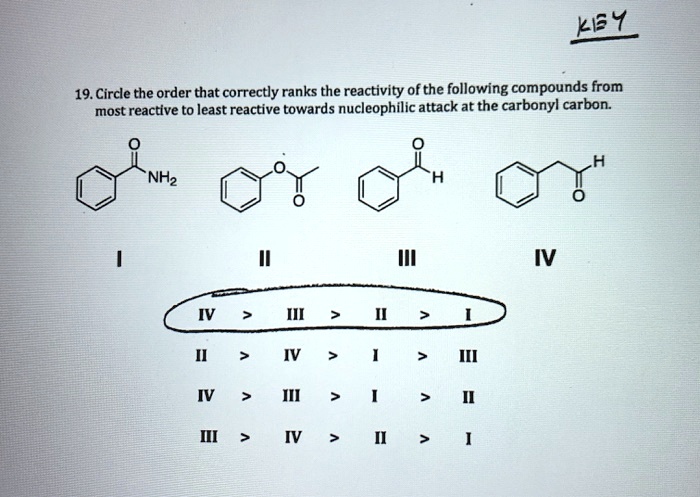
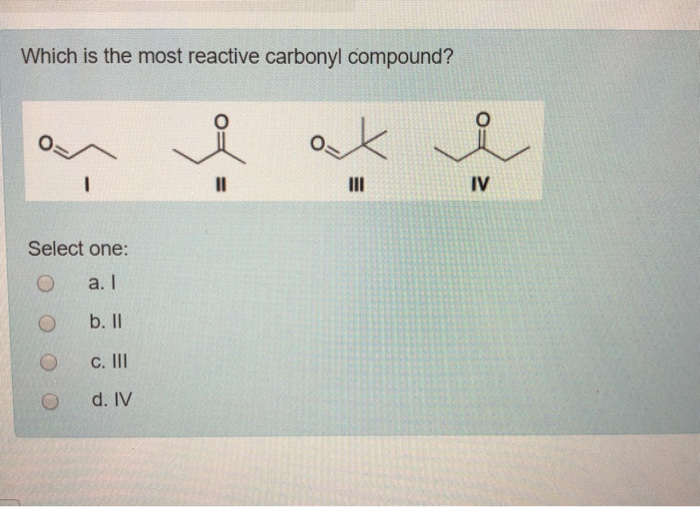
Which Is The Most Reactive Carbonyl Compound
Let's talk about molecules. Not just any molecules, but the ones that have a certain sparkle, a certain something. We're diving into the world of carbonyl compounds. Think of them as the life of the party in chemistry class.
These guys have a carbon-oxygen double bond. It's like a handshake, but way more dramatic. This bond is pretty special. It makes them super interesting to chemists.
We're on a quest today. A quest for the most reactive. The speed demon. The one that jumps into reactions the fastest. It's a bit like picking your favorite flavor of ice cream. So many choices, but one reigns supreme.
Must Read
Now, you might think you know the answer. Maybe you've heard whispers in the lab. Or perhaps you've seen some dramatic experiments. But I'm here to throw a little curveball. An opinion that might ruffle some feathers.
We've got a whole family of these carbonyl stars. There are aldehydes, and then there are ketones. They're like siblings, but with different personalities. Some are shy, others are bold.
And then we have the ones with the extra flair. The carboxylic acids. They bring their own acidity to the party. And let's not forget their derivatives: esters, amides, acid halides, and acid anhydrides. They're like the cousins, all related but distinct.
The question is, who’s the fastest? Who’s the most eager to react? Who can’t wait to get involved? It’s a tough call, I know. They all have their moments.
We often hear about the usual suspects. People point fingers at the acid halides. They're like the hotshots, always ready to go. They have leaving groups that are just begging to be replaced.

And then there are the acid anhydrides. They’re pretty speedy too. They can donate their acyl group with gusto. They’re like that friend who always brings a plus-one.
Esters are a bit more mellow. They’re polite. They react, but they take their time. They’re not in a huge rush.
Amides? Oh, they’re the most chill of the bunch. They are incredibly stable. It takes a lot to get them going. They’re like that one person who always says, "Let's just relax."
So, where does that leave our aldehydes and ketones? They’re somewhere in the middle. They react with nucleophiles, those electron-rich guests. They’re happy to oblige.
But here’s where my unpopular opinion might land. While the acid halides are undeniably reactive, and the anhydrides are close behind, I have a soft spot. A very, very soft spot.

I think, in spirit, the simplest aldehyde takes the cake. Yes, you heard me. Formaldehyde.
Now, I know what you’re thinking. "Formaldehyde? The stuff that preserves things?" Exactly! But think about its structure. It's got two little hydrogens attached to that carbonyl carbon.
This means there’s nothing bulky in its way. No big groups to get in the middle of a reaction. It's wide open. A welcoming committee for any incoming nucleophile.
Compare it to, say, acetone. Acetone has two methyl groups. They're a bit like little traffic cones. They can slow things down just a tad. They create a bit of steric hindrance.
Formaldehyde has zero steric hindrance. It’s like a perfectly smooth runway. Reactions just happen. It’s so ready, it’s practically vibrating with anticipation.
And the electron-withdrawing effect of the oxygen in the carbonyl is felt most acutely in formaldehyde. With no other electron-donating alkyl groups, the carbonyl carbon is even more positive, making it a prime target.

Think of it like this: If reactivity were a dance, formaldehyde would be the first one on the dance floor. No hesitation. Just pure, unadulterated action.
The acid halides have super reactive leaving groups, sure. They’re like a champagne cork that’s already loose. But formaldehyde is like a perfectly tempered spring, ready to release its energy at the slightest touch.
It’s the purity of its structure. The lack of any "attitude" from bulky side groups. It’s just carbon, oxygen, and two tiny hydrogens. The essence of carbonyl reactivity.
So, while the textbooks might tell you to look at the leaving groups, I urge you to consider the unsung hero. The molecule that’s always ready. The one that’s so pure in its intention to react.
It’s the simplest aldehyde, the humble, yet mighty, formaldehyde. It’s not about the fancy substituents; it’s about the fundamental attraction. The pure, unadulterated pull of the carbonyl.

Of course, there are nuances. Specific reactions can favor different carbonyls. But if we're talking about general eagerness, about being the most open and inviting to a wide range of nucleophiles, I stand by formaldehyde.
It’s a bit like judging the best singer. Do you go for the one with the most impressive vocal range, or the one who sings with the most heartfelt emotion? I'm leaning towards emotion here. And formaldehyde has that in spades.
So next time you see a carbonyl compound, remember the little guy. The one that’s often overlooked for its sheer simplicity. It’s a testament to how even the smallest things can have the biggest impact.
It’s the molecule that says, "Come on, let’s do this!" without any fuss or fanfare. Just pure, unadulterated chemical enthusiasm. And that, my friends, is a kind of reactivity we can all appreciate.
So, while acid halides might get all the press for their lightning speed, I’m giving a standing ovation to formaldehyde. It's the underdog, the quiet achiever, the true heartthrob of the carbonyl world.
It's the molecule that shows us that sometimes, less is more. Less clutter, less obstruction, more pure, vibrant reactivity. It’s a beautiful thing, really.
And that, my friends, is my slightly controversial, but deeply felt, take on the most reactive carbonyl compound. It’s a matter of perspective, and my perspective is firmly rooted in the elegant simplicity of formaldehyde.