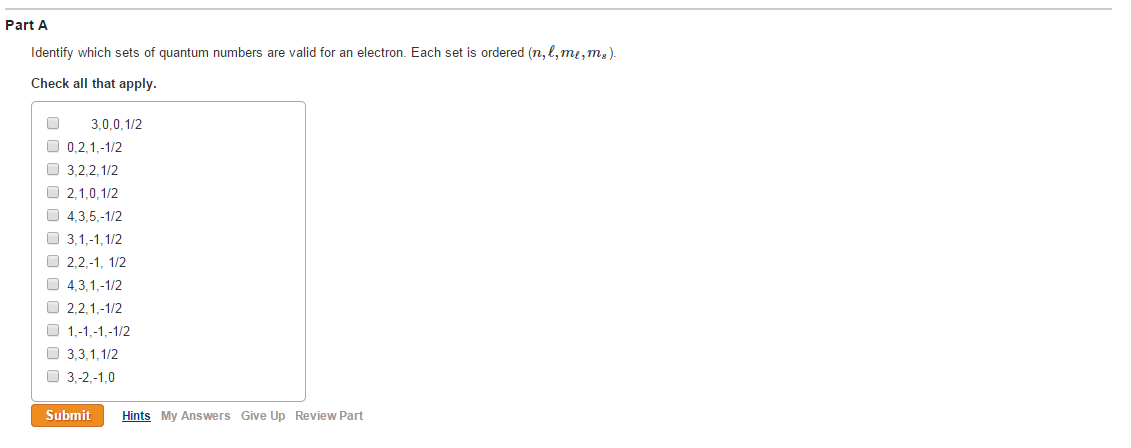
Which Is Not A Valid Set Of Quantum Numbers
Quantum numbers. They sound like something out of a sci-fi movie, don't they? Like the secret code to unlock a secret dimension or maybe the password to get into a super exclusive club. But nope, they're actually about tiny, itty-bitty things called electrons.
Think of electrons like little tenants living in an apartment building called an atom. This building has floors, rooms, and even specific spots where each tenant likes to hang out. And the quantum numbers? They're basically the address of each electron.
You've got your principal quantum number, which is like the floor number. It tells you how much energy the electron has. Higher floor, more energy. Simple enough, right? Like living on the penthouse versus the basement.
Must Read
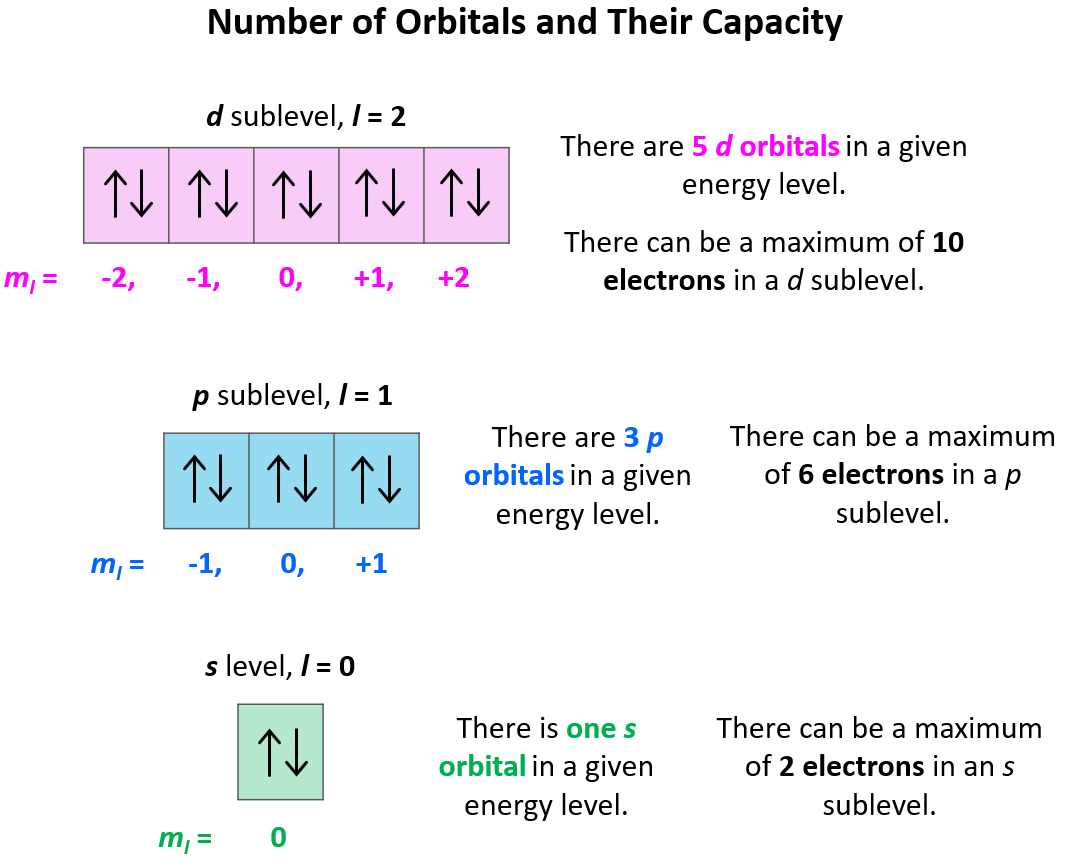
Then there's the angular momentum quantum number. This one's a bit trickier. It describes the shape of the electron's "room" or orbital. Some are round like a ball, others look like dumbbells. It’s like saying your apartment is a studio or a two-bedroom.
And don't forget the magnetic quantum number. This one specifies the orientation of that "room" in space. Imagine those dumbbell shapes: they can point up and down, or side to side. It's like having a view of the park or just a brick wall.
Finally, we have the spin quantum number. This is where things get a little wild. Electrons have this thing called "spin," which is like a tiny internal magnet that can point either up or down. It's like saying whether the tenant likes their cereal with milk or dry.
Now, here's the fun part. Just like you can't have a rent of negative $500 or an apartment number that's a purple unicorn, there are rules for these quantum numbers. They have to follow a specific set of principles. And when they don't? Well, that's where the magic (or the confusion) happens.
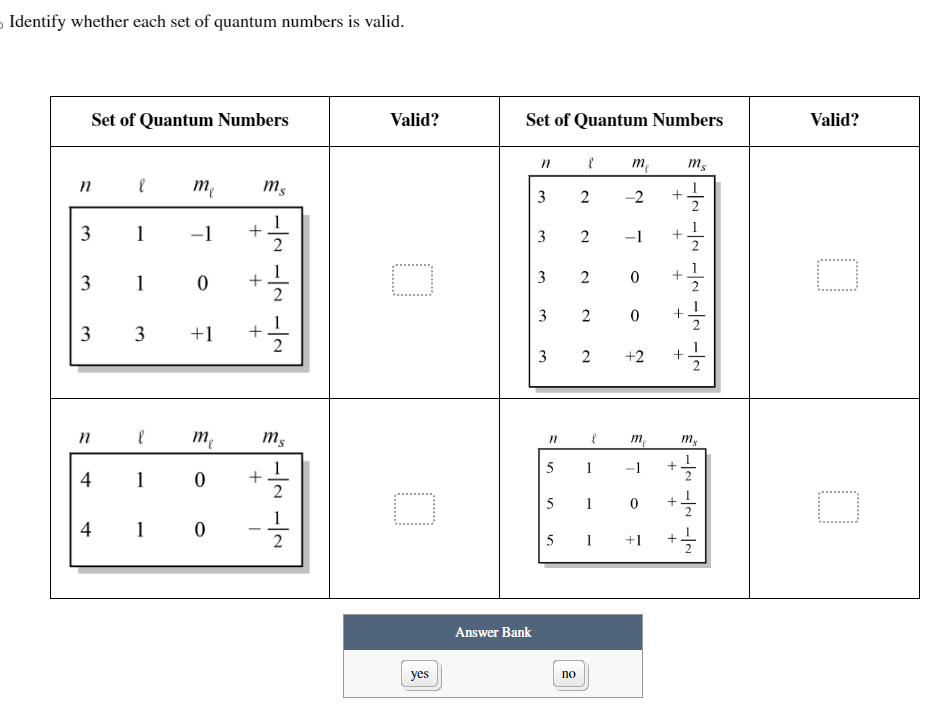
Imagine you're trying to book a hotel room. You need a room number, right? And maybe a floor number. But what if you ask for "Room 7, Floor Banana"? That doesn't quite make sense in the hotel universe.
Similarly, in the quantum world, certain combinations of quantum numbers are just… not allowed. They're like the forbidden fruit of electron addresses. They break the fundamental laws of electron housing.
So, which combinations are the troublemakers? Which ones make the universe say, "Whoa there, buddy, that's not how it works!" It's like trying to fit a square peg in a round hole, or wearing socks with sandals to a formal event. It just feels wrong.
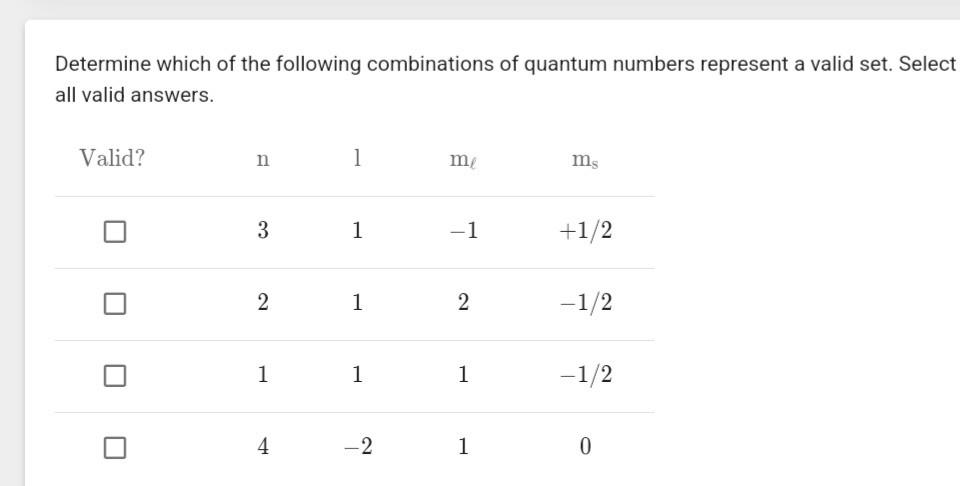
Let's consider the principal quantum number, symbolized by 'n'. This guy can be 1, 2, 3, and so on. No decimals, no negative numbers, and definitely no fractions. It's like saying you can only have whole floors in your building. You can't have a 2.5 floor, can you? It’s just not a thing.
Then we have the angular momentum quantum number, 'l'. This one has a relationship with 'n'. It can range from 0 up to 'n-1'. So, if you're on floor 1 (n=1), 'l' can only be 0. If you're on floor 2 (n=2), 'l' can be 0 or 1. It's like saying on the first floor, you only have basic rooms (l=0). On the second floor, you get basic rooms and some slightly fancier ones (l=1).

Now, the magnetic quantum number, 'ml'. This one depends on 'l'. It can range from -l up to +l, including 0. So, if 'l' is 0, 'ml' can only be 0. If 'l' is 1, 'ml' can be -1, 0, or 1. This is like saying if you have a basic room (l=0), there's only one way it can be oriented. But if you have a fancier room (l=1), it can be oriented in a few different directions.
And finally, the spin quantum number, 'ms'. This one's the easiest to please. It can only be +1/2 or -1/2. It's like the electron having two moods: happy or grumpy. Or perhaps: awake or asleep. Always one of two options.
So, what happens when these numbers go rogue? When they try to pull a fast one on the laws of physics? It’s like a glitch in the matrix, but for electrons.
Let's say you have an electron with n=1. Based on the rules, 'l' can only be 0. So, if you try to give it an 'l' value of, say, 2? That's a big nope. It's like trying to put a king-sized bed in a shoebox. It's just not going to fit, and it's definitely not allowed.
Or imagine you have 'l' = 1. Then 'ml' can be -1, 0, or 1. But if someone suggests 'ml' = 3? That’s another red flag. It’s like trying to order a third scoop of ice cream when the machine only offers two flavors. Utterly impossible.
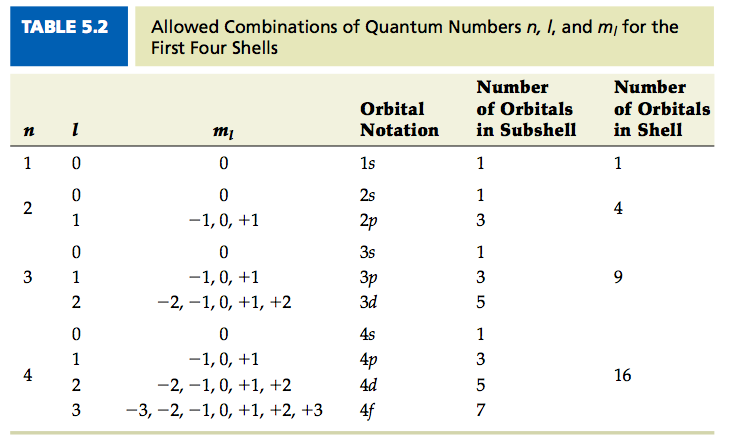
Sometimes, the error might be in the principal quantum number itself. If someone proposes n = -2? Well, you can't have a negative floor in your apartment building, can you? That would be like living in the underworld, and electrons, as cool as they are, don't typically reside there.
Or what about fractional principal quantum numbers? Like n = 1.5. That's just… silly. Floors are whole numbers. Energy levels are whole numbers. It’s the universe’s way of saying, "Keep it simple, folks!"
It's these little inconsistencies, these little rule-breaking combinations, that are not valid sets of quantum numbers. They're the cosmic equivalent of a typo in a very important document. They just don't compute.
Think of it like this: you're trying to send a postcard. You need a street address, a city, a state, and a zip code. But what if you try to send a postcard with a zip code that doesn't exist, or a street name that's a made-up word? The postcard isn't going anywhere. It’s invalid.
And that's precisely how these invalid sets of quantum numbers function. They're like phantom addresses for electrons. They don't correspond to any real state an electron can occupy. They’re theoretical oddities, the scientific equivalent of a unicorn sighting.
So, while the rules might seem a bit finicky, they’re actually what keep the universe in order. They ensure that electrons behave themselves and don't go around causing quantum chaos. It’s all about maintaining that delicate balance in the atomic realm.
It’s kind of like when you’re trying to assemble furniture. If you use the wrong screw, or put a piece on backwards, it just doesn’t work. The whole thing falls apart, or at least looks really wonky. Invalid quantum numbers are like using the wrong screw.
These invalid sets are the ones that violate the fundamental relationships between n, l, ml, and ms. They are the ones that are mathematically and physically impossible for an electron to possess. They are the outliers, the exceptions that prove the rule.
So, the next time you hear about quantum numbers, remember it's all about these tiny addresses. And just like any address, it has to follow certain rules to be valid. Otherwise, the electron just… well, it just can't go there. It's the universe's way of keeping things tidy. And honestly, who can argue with tidy?
The beauty of science, even the quirky parts like quantum numbers, lies in its order. And sometimes, finding what doesn't fit helps us understand what does.
So, while the valid sets are the rockstars of electron addresses, the invalid ones are the background characters that help us appreciate the main performers. They’re the funny mistakes that make the story more interesting. And in the grand scheme of things, that’s pretty cool, wouldn’t you say?