Which Formula Is An Ionic Compound Brainly

Hey there, fellow curious minds! Ever found yourself staring at a jumble of letters and numbers, like NaCl or H₂O, and wondered, "What in the world is this stuff, and more importantly, which one is the ionic compound?" If your brain feels like a scrambled egg trying to decode chemistry formulas, you're in the right place! Think of me as your friendly neighborhood chemistry guide, ditching the dusty textbooks for a chat over coffee.
We're going to unravel the mystery of ionic compounds. No need to break out your calculator or memorize the periodic table (yet!). We'll keep it light, breezy, and hopefully, a little bit fun. Because, believe it or not, understanding this stuff is actually pretty neat and pops up in our everyday lives more than you might think.
So, What's the Big Deal with Ionic Compounds?
Imagine you're at a party, and you've got two friends who just really like each other. They're practically inseparable. That's kind of like an ionic compound! It's formed when one atom is a bit of a giver and the other is a bit of a taker. They're so attracted to each other that they stick together, forming a bond. In chemistry terms, one atom gives away an electron, becoming positively charged (like a happy, generous friend), and the other atom takes that electron, becoming negatively charged (like someone who gratefully accepts a gift). These opposite charges create a super strong attraction, like magnets!
Must Read
Why should you care? Well, these sticky-together atoms create all sorts of cool stuff we use every single day. Think about it: the salt on your fries? That's an ionic compound! The baking soda you use to make your cookies fluffy? Yep, ionic compound! Even the bleach that whitens your clothes? You guessed it – ionic compound!
Let's Play "Spot the Ionic Compound"
So, how do we spot these ionic compounds in the wild, or rather, in the world of chemical formulas? It usually comes down to the types of atoms involved. Ionic compounds typically form between a metal and a nonmetal. Think of metals as the shiny, happy givers and nonmetals as the electron-loving takers.
Let's break it down with some everyday comparisons:
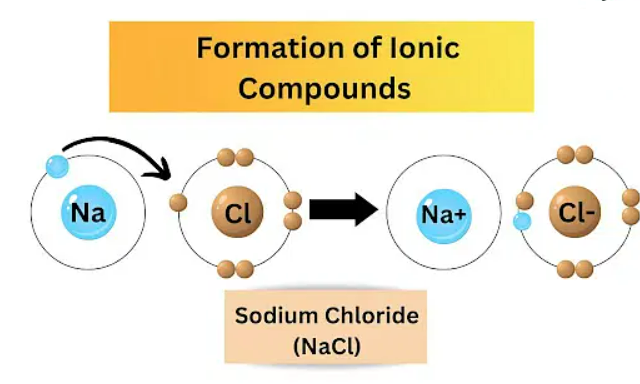
The "Salt and Pepper" Analogy
Take our classic friend, NaCl. This is sodium chloride, otherwise known as table salt. Sodium (Na) is a metal, and chlorine (Cl) is a nonmetal. See that pairing? Metal and nonmetal! Sodium is the generous friend who says, "Here, have an electron!" and chlorine happily accepts. They become charged and stick together like perfect dance partners. It's a classic ionic compound.
Imagine salt as a perfectly balanced meal. You've got your savory protein (the metal, willing to share) and your zesty spice (the nonmetal, eager to receive). They complement each other perfectly, and you get that satisfying flavor – or in chemistry, a stable compound!
The "Give and Take" Dance
Another common one is MgO, magnesium oxide. Magnesium (Mg) is a metal, and oxygen (O) is a nonmetal. Again, metal and nonmetal! Magnesium is so generous it gives away two electrons, and oxygen is so enthusiastic it takes them both! This dance of giving and taking creates a super strong ionic bond. This is actually what makes up the white powdery stuff you might see on some rocks – it's a mineral called periclase.
Think of it like a really generous person (magnesium) finding someone who needs a lot of help (oxygen). They make a pact, and they're bound together by that act of extreme generosity and need.
What About the Other Guys? (The Covalent Compounds)
Now, not all chemical formulas are ionic. Some are what we call covalent compounds. These are like friendships where everyone shares nicely. Instead of one atom giving an electron away and the other taking, they share electrons. It's more like a potluck where everyone brings a dish to share, rather than one person buying all the food.
A perfect example is water, H₂O. Here, we have hydrogen (H) and oxygen (O). Both are nonmetals. They don't have that strong "give and take" vibe of metals and nonmetals. Instead, they decide to share their electrons. It’s a collaborative effort, and they form a covalent bond. This is why water is so amazing and can dissolve so many things – it's a fantastic sharer!

Or think about the air you breathe, O₂. That's just two oxygen atoms, both nonmetals, deciding to be best buddies and share their electrons. They're not giving or taking; they're just hanging out and sharing.
The Brainly Question: How Do You Know?
So, when you see a formula on Brainly or anywhere else, and you're asked, "Which one is an ionic compound?", here’s your easy-peasy shortcut:
- Look for a Metal and a Nonmetal: If you see a metal atom bonded with a nonmetal atom, there's a very high chance it's an ionic compound. Think of the periodic table as your cheat sheet. Metals are generally on the left side, and nonmetals are on the right (excluding the noble gases, which are loners!).
- Look for Two Nonmetals: If you see two or more nonmetal atoms bonded together, it's almost certainly a covalent compound.
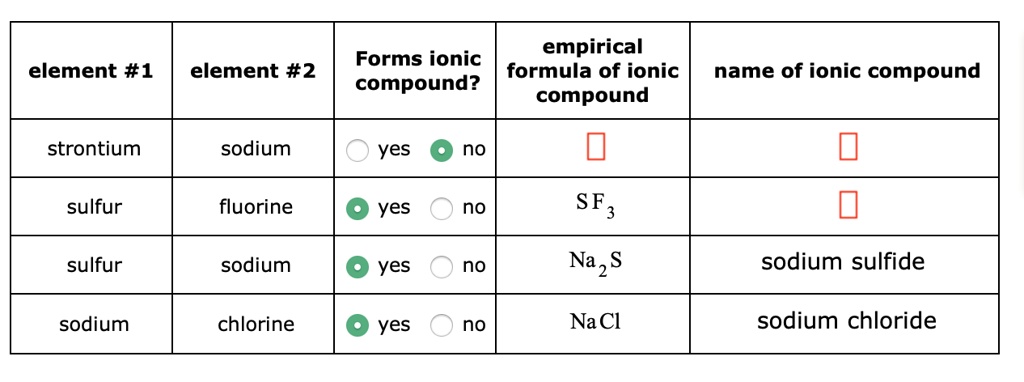
For example, if you saw CaCl₂, you'd spot calcium (Ca), which is a metal, and chlorine (Cl), which is a nonmetal. Metal + Nonmetal = Ionic Compound! Your Brainly answer is ready!
.PNG)
If you saw CO₂ (carbon dioxide), you'd see carbon (C) and oxygen (O), both nonmetals. Nonmetal + Nonmetal = Covalent Compound!
Why This Matters (Beyond the Chemistry Quiz)
Understanding this difference helps us understand why things behave the way they do. Ionic compounds, because of their strong attraction, often form solids with high melting points. Think about how hard it is to melt salt! Covalent compounds, on the other hand, can be gases, liquids, or solids with lower melting points. Water boils relatively easily, right?
It's like the difference between a tightly knit family that sticks together through thick and thin (ionic) and a group of good friends who are there for each other but can also go their separate ways (covalent). Both are important, but their structures and behaviors are different.
So, next time you're looking at a chemical formula, don't be intimidated! Just remember the party friends, the sharing buddies, and the trusty metal-nonmetal rule. You've got this! And who knows, you might even start to find it as fascinating as finding the perfect topping for your pizza. Happy chemically-exploring!


![[ANSWERED] 4 Ionic compounds In the chart below write the ionic formula](https://media.kunduz.com/media/sug-question-candidate/20231215163733250409-5651308.jpg?h=512)
