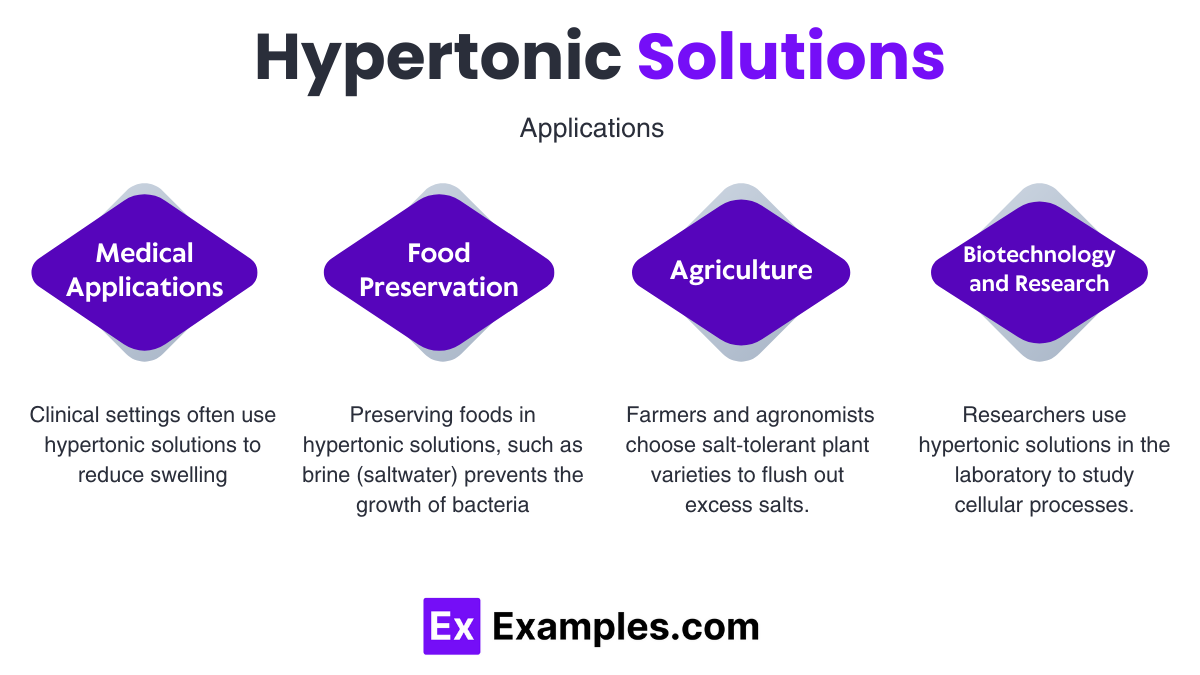
Which Field Contains A Hypertonic Solution
Ever feel like your body's playing a wacky game of tug-of-war with water? Well, get ready to dive into the wonderfully weird world of hypertonic solutions, because it’s a concept that’s much more fun (and less… science-textbook-y) than it sounds!
Imagine you're at a picnic, and someone brings out a ginormous bowl of popcorn. But this isn't just any popcorn; it's been sprinkled with, like, a mountain of salt. That salty popcorn, my friends, is your first clue into the world of the hypertonic. It’s all about things being super, duper concentrated!
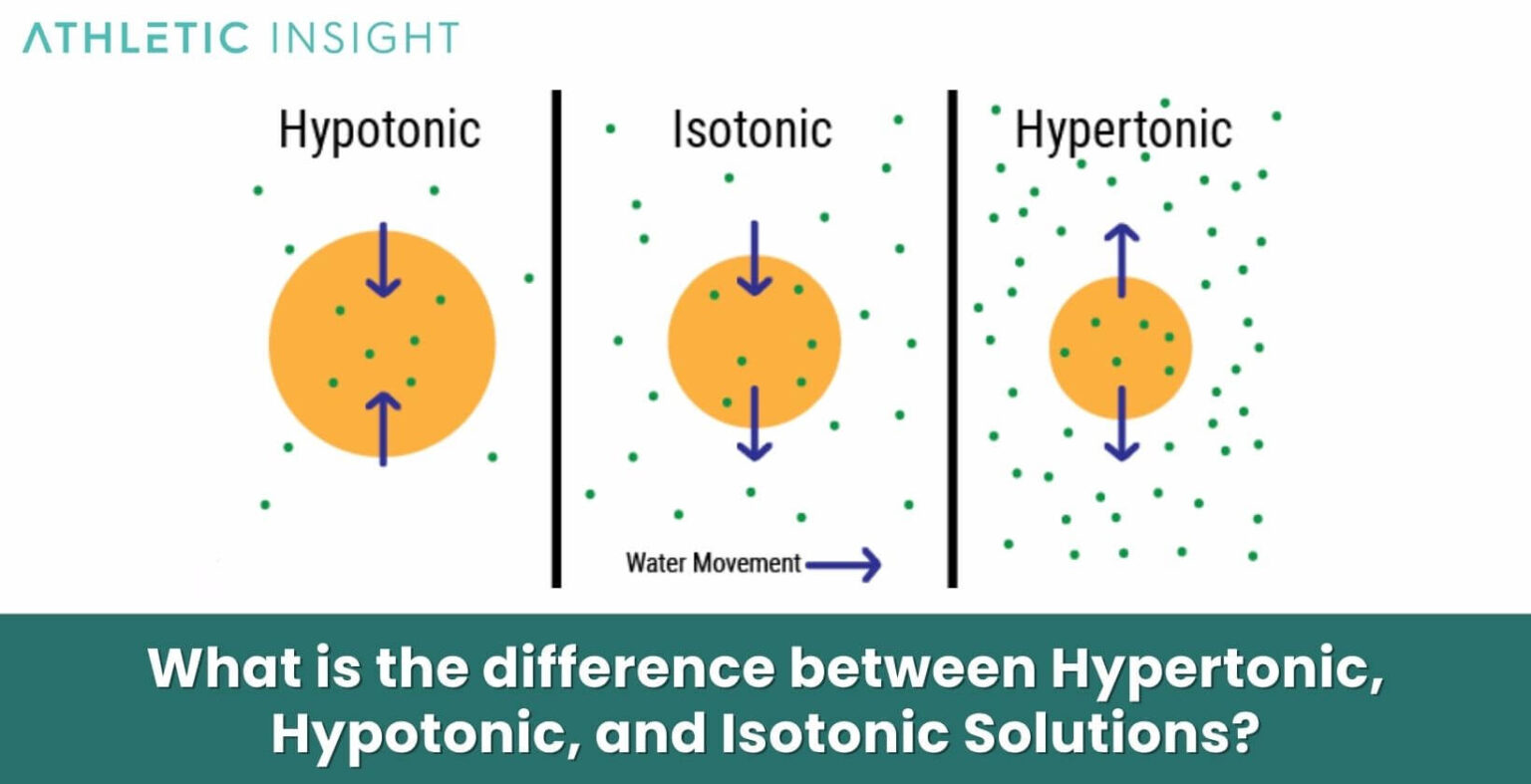
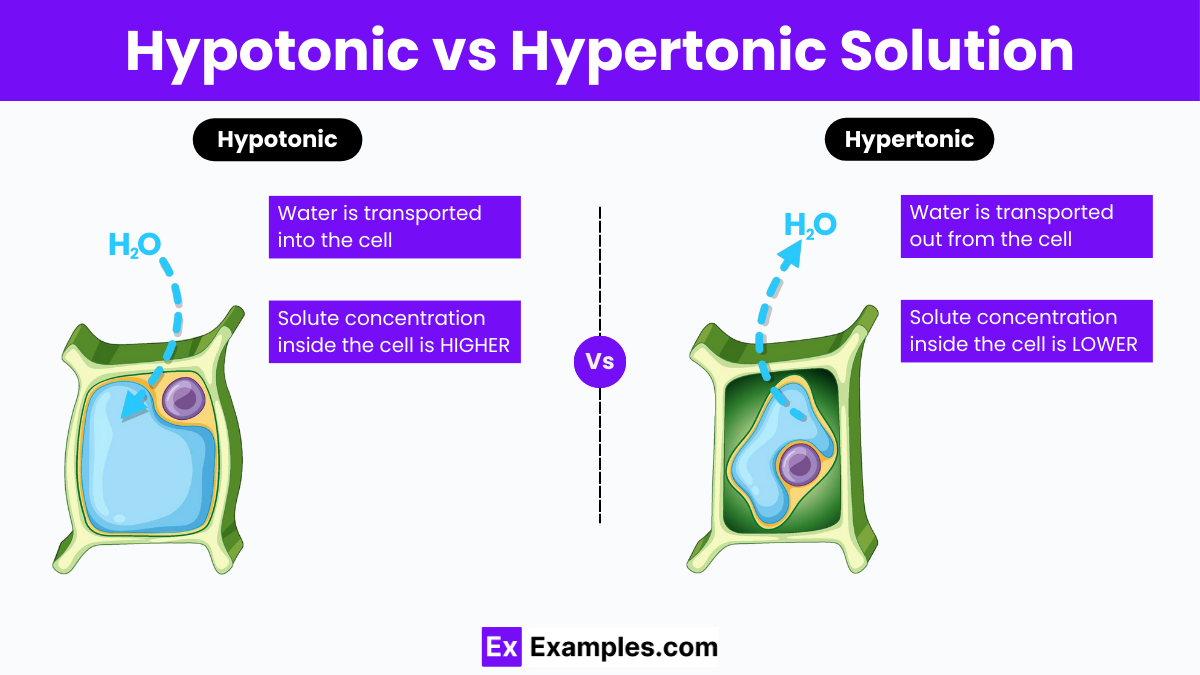
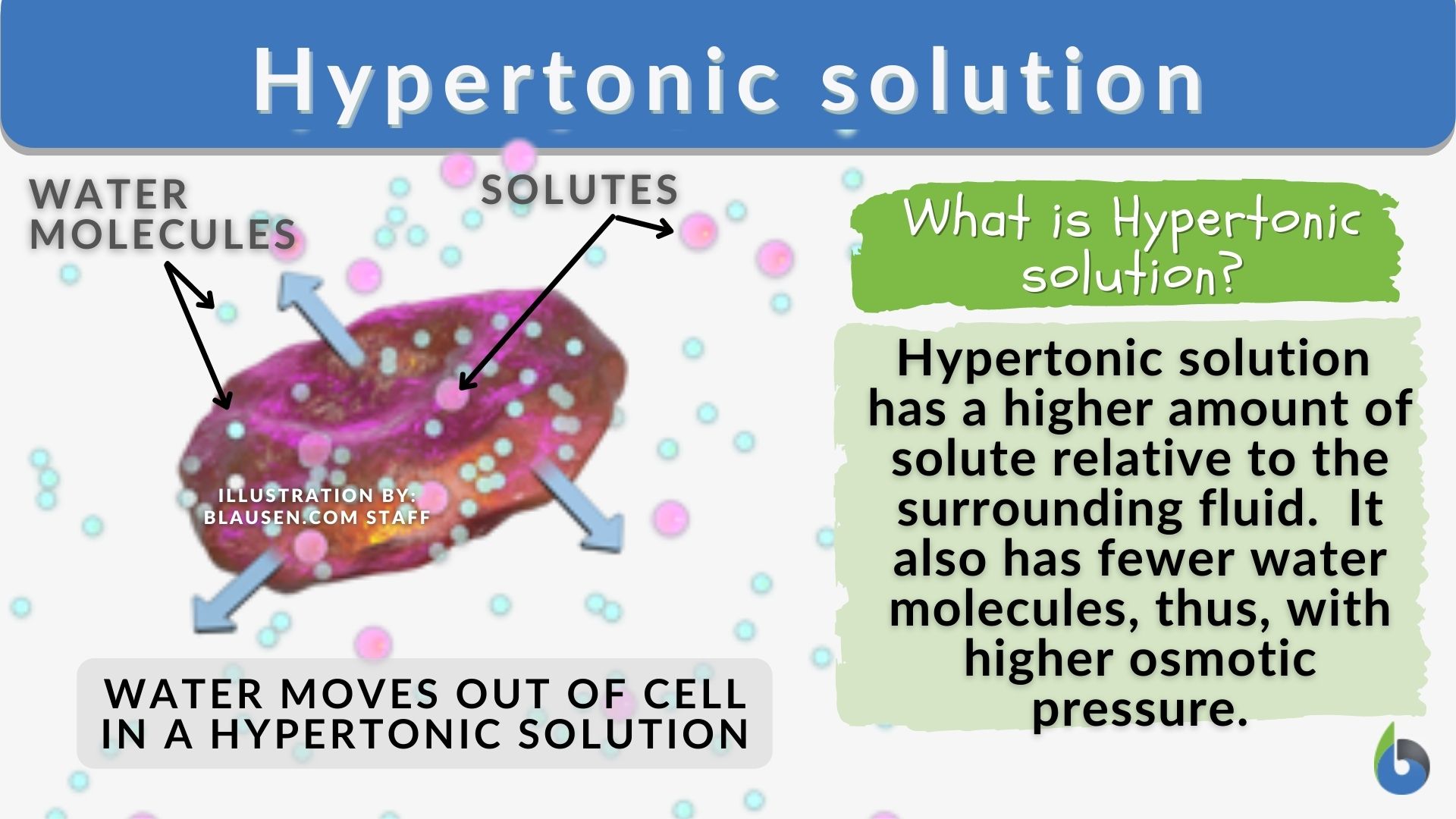
So, what's the deal with a hypertonic solution? Think of it as a very thirsty friend. This solution has way more stuff dissolved in it compared to the stuff inside the tiny little cells floating around. This “stuff” could be anything – salts, sugars, you name it!
Must Read
Now, here’s where the magic (and a little bit of drama) happens. Water, being the incredibly polite and helpful molecule it is, absolutely hates it when things are uneven. Water is like that friend who always tries to balance out the snacks at a party.
So, when water encounters a hypertonic solution, it’s like it’s saying, "Whoa there, buddy! You're looking a little… concentrated! Let me even things out!" And how does water do this? By doing what it does best: moving!
Water will naturally flow from an area where there’s less stuff dissolved to an area where there’s more stuff dissolved. It's like a water-based migration, all in the name of fairness and balance. It's a natural instinct for water to try and dilute the really concentrated areas.
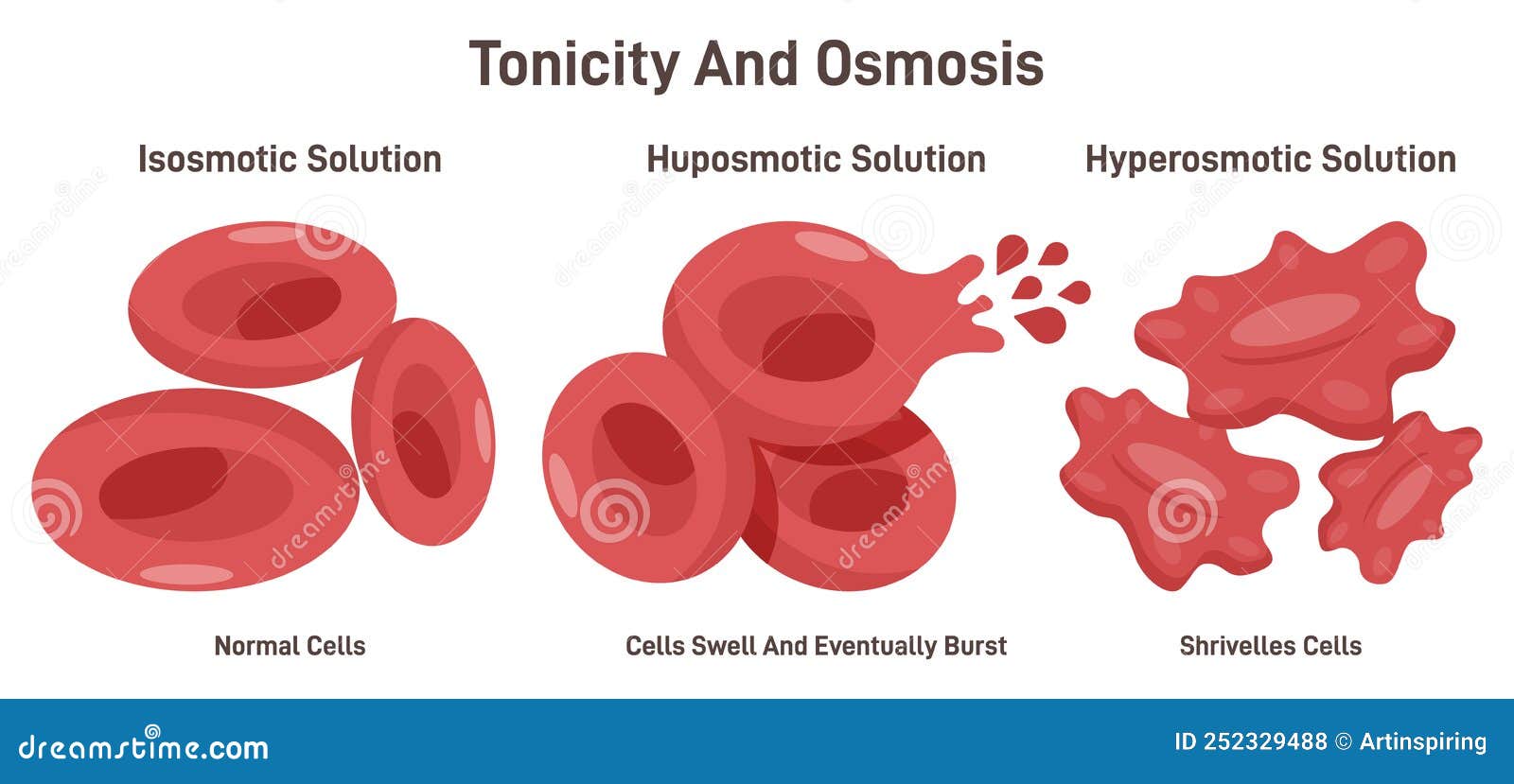
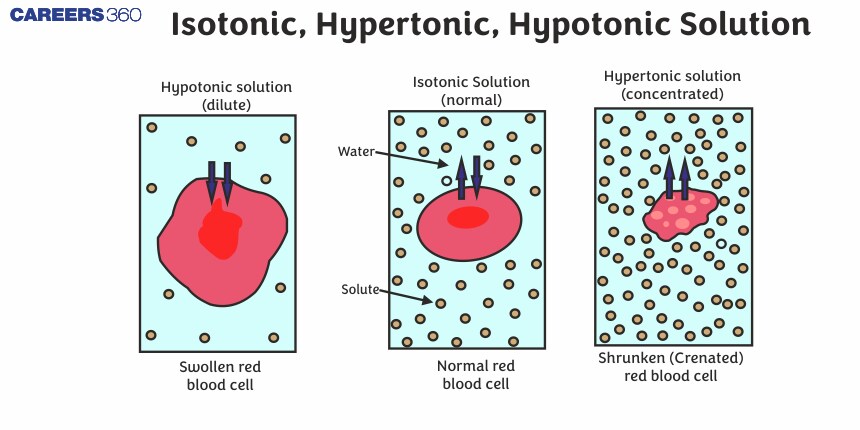
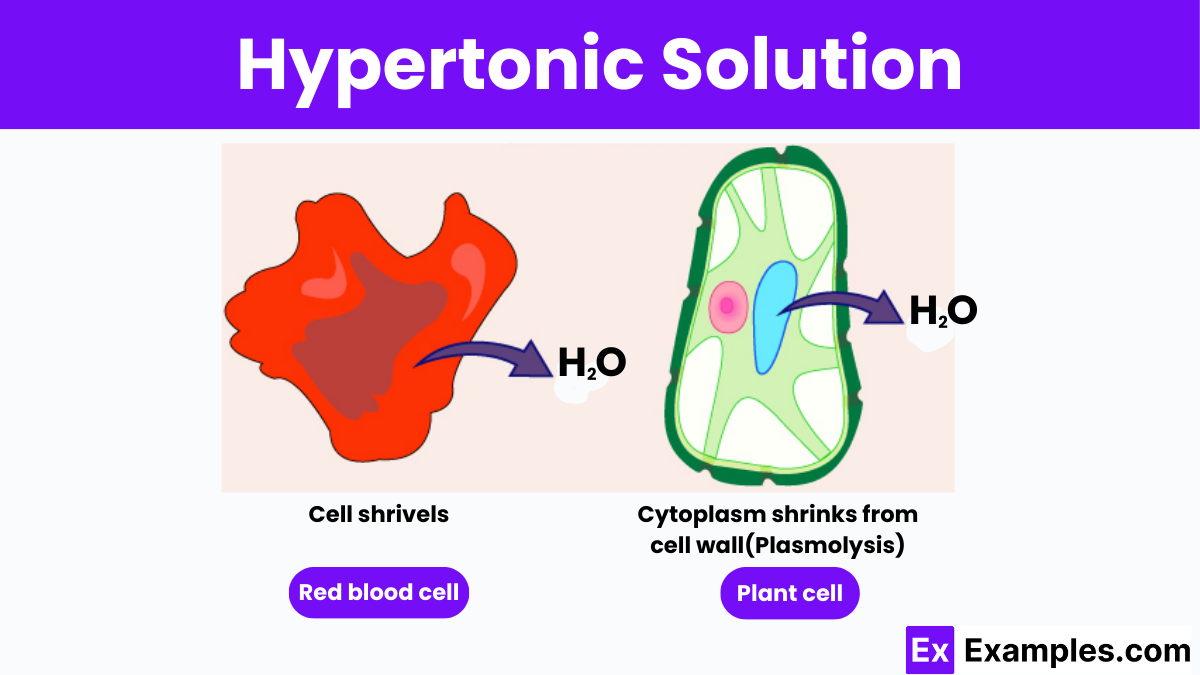
Let’s bring in our trusty friend, the red blood cell. These little guys are like tiny, plump water balloons, and they're constantly surrounded by different fluids. If a red blood cell finds itself swimming in a hypertonic solution, oh boy, things are about to get interesting!
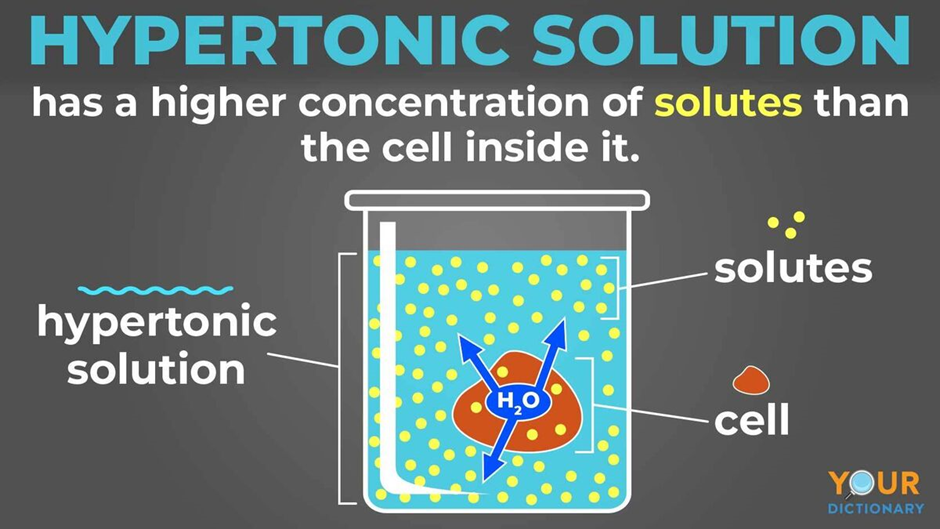
Remember our super-salty popcorn? Imagine that popcorn is now the fluid outside the red blood cell. The inside of the red blood cell has a certain amount of dissolved stuff, but the outside is way more concentrated. The red blood cell is like, "Uh oh, this is not good!"
Because the outside solution is hypertonic, it has a higher concentration of solutes. The water inside the red blood cell is thinking, "There’s so much more 'stuff' out there! I gotta go help!" And with that, water starts to trickle out of the red blood cell and into the surrounding hypertonic solution.
It’s like the red blood cell is being gently squeezed. The water is leaving, trying to dilute that super-concentrated environment. This makes the red blood cell start to shrink. It loses its plumpness and becomes all shriveled up, looking like a tiny, sad raisin. Poor little guy!
The Salty Sea Scourge
Think about being at the beach. The ocean is, well, salty! If you were to somehow (don't do this, it's a bad idea!) drink a big gulp of seawater, your body would be in a bit of a pickle. The seawater is a hypertonic solution compared to the fluids inside your cells.

Your body would have to work overtime to get rid of that extra salt. Water would actually be pulled out of your cells to help dilute the ingested seawater. This is why drinking saltwater is a terrible idea for survival. It dehydrates you more!
The Sweet Surprise
Now, let’s switch gears to something a little more… delicious. Imagine you're making homemade jam. You’ve got loads of fruit, but then you add a massive amount of sugar. That sugary concoction, before it gets all gooey and delightful, is a super concentrated, hypertonic solution!
If you were to dip something delicate, like a tiny piece of fruit flesh, into that sugar syrup, the water would start to leave the fruit and join the sugary party. This is actually how jam is preserved – the high sugar concentration makes it hard for bacteria to survive because it draws water out of them!
Pickles and Preserves
Speaking of preserving, have you ever wondered how pickles get so… well, pickle-y? It often involves brine, which is basically a salty water solution. And guess what? That brine is often a hypertonic solution!
When cucumbers hang out in that salty brine, water is drawn out of them. This helps to firm them up and start the pickling process. It’s a classic example of osmosis and hypertonic solutions in action, making our snacks taste amazing!
The Medical Marvel
This isn’t just about snacks and seas; hypertonic solutions play a big role in medicine too! Doctors sometimes use them to help reduce swelling.
For instance, if someone has swelling in their brain, a doctor might administer a hypertonic solution intravenously. This draws excess water out of the swollen brain cells, helping to alleviate pressure. It’s like a gentle, medical tug-of-war to get things back to normal!
So, next time you hear the term hypertonic solution, don't let it intimidate you. Just think of that super-salty popcorn, the vast ocean, or the sweet jam. It’s all about a solution that’s a little too full of dissolved goodies, prompting water to do its thing and move!
It’s a fundamental principle of how water behaves, influencing everything from how our cells function to how we preserve food and even how medical treatments are administered. It’s a small concept with a HUGE impact, and understanding it just makes the world a little more fascinating!
So, the field that contains a hypertonic solution is any place where you find a concentrated mixture, often with a goal to draw water out of surrounding areas. From the biology lab to your kitchen counter, these solutions are quietly doing their work, making the world a more balanced (and sometimes, tastier!) place.
Remember, water is always looking for that sweet spot, that equilibrium. And when it finds a hypertonic solution, it’s like it’s got a mission: to dilute, to balance, and to make things right. It’s a constant, microscopic dance that keeps our world running!
Think of all the tiny cellular parties happening right now, where water is making its choices. In a hypertonic environment, water is the guest who’s always leaving the crowded room to go to the slightly less crowded hallway. It's a fascinating phenomenon, truly!
So, embrace the wonder of the hypertonic solution. It’s a concept that's both simple and profound, and a reminder that even in the smallest of things, there’s a whole universe of activity and purpose. Isn't that just… awesome?