Which 1 Mole Sample Has The Least Entropy
Hey there! So, imagine you're chilling with a cup of coffee, right? We're gonna talk about something kinda cool, but also a little nerdy. We're diving into the world of entropy. Don't worry, it's not as scary as it sounds, promise! Think of it as a measure of… well, messiness or randomness. Yeah, that’s a good way to put it. More mess, more entropy. Less mess, less entropy. Easy peasy, right?
Now, the question that's been tickling my brain (and hopefully yours too!) is this: out of a mole of different stuff, which one is the least messy? Which one is the most chill, the most ordered? We’re talking about a mole, remember? That's just a super specific number of particles. Like, a dozen eggs is 12 eggs. A mole is… way, way more than that. It’s 6.022 x 10^23 of something. Tiny things, mostly. Like atoms or molecules. So, we’ve got a huge, massive pile of these tiny things, all behaving according to their own rules. And we wanna know which pile is the most… well, tidy.
This whole entropy thing, it's a big deal in science, especially in thermodynamics. It’s all about how energy likes to spread out, how things tend to go from organized to disorganized. Like your desk after a busy week, am I right? It doesn’t just magically tidy itself up, does it? Nope. It becomes a glorious monument to chaos. Entropy is basically that natural pull towards… disorder.
Must Read
But here’s the kicker: we’re looking for the opposite. We want the least entropy. The most order. The most… predictable behavior. So, what kind of substances are we even talking about? We could have a mole of helium gas floating around, all energetic and bumping into each other. Or a mole of solid diamond, all perfectly arranged atoms. Or maybe even a mole of water, H2O, that can be ice, liquid, or gas. Lots of possibilities!
Let’s break it down, shall we? When we talk about entropy, we’re really looking at how many different ways a system can be arranged and still be in the same state. Think of it like shuffling a deck of cards. If the cards are all perfectly ordered by suit and rank, there’s only one way to have that perfect order, right? That's low entropy. But if you shuffle them? Oh boy, there are gazillions of ways those cards can be all jumbled up. High entropy!
So, for our mole of stuff, we're essentially asking: how many different microstates correspond to the same macrostate? Don't let those big words scare you! Macrostate is just what we observe on a big scale – like the temperature, pressure, and volume. Microstate is the nitty-gritty, the exact position and momentum of every single little particle. The more ways you can arrange those tiny particles to get the same overall result, the higher the entropy.
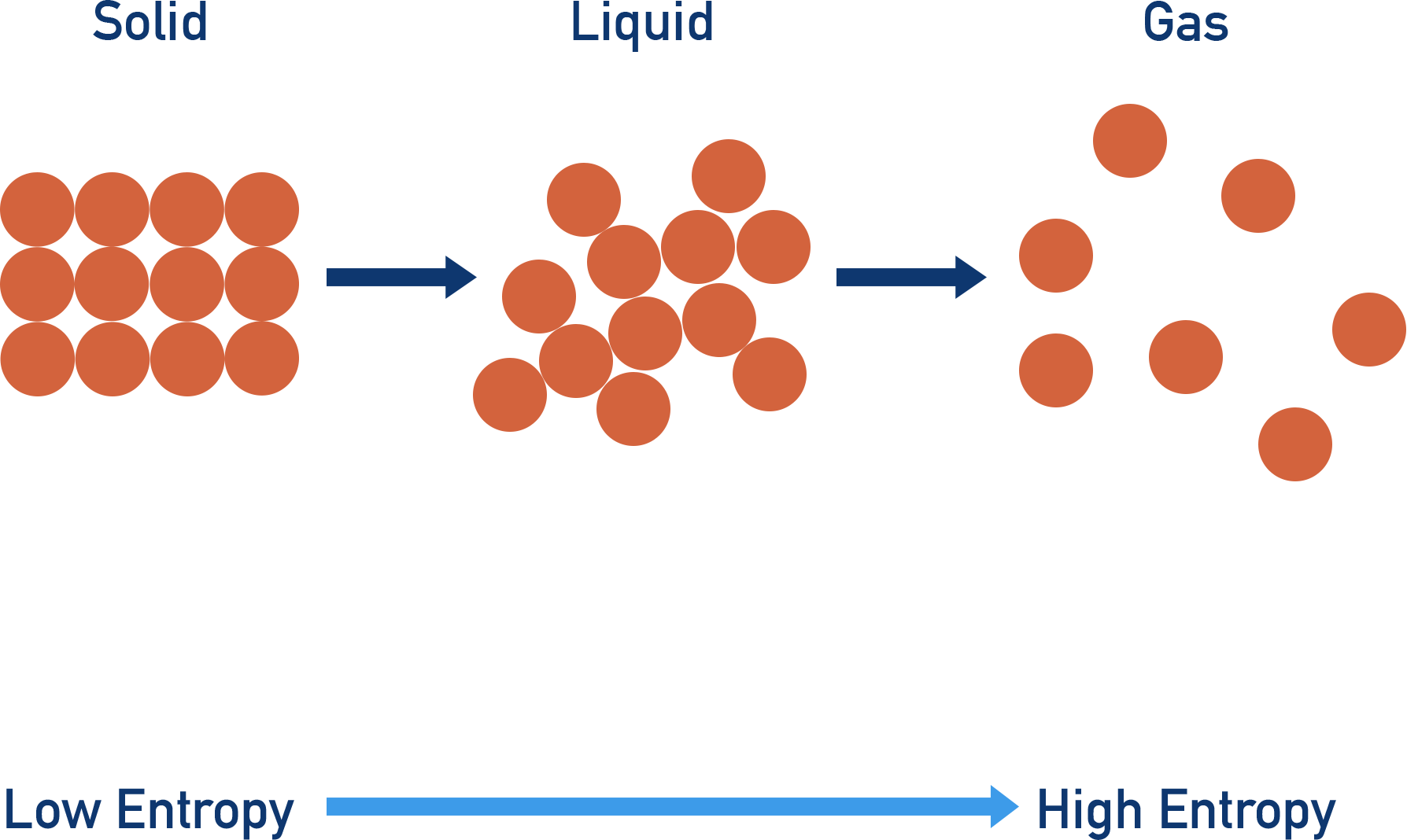
Now, what are the contenders for the least entropy? We need something that's naturally very ordered. Something that doesn't have a lot of wiggle room for its particles to move around randomly. Think about the different states of matter, yeah? We've got solids, liquids, and gases. Which one sounds the most… stuck in place?
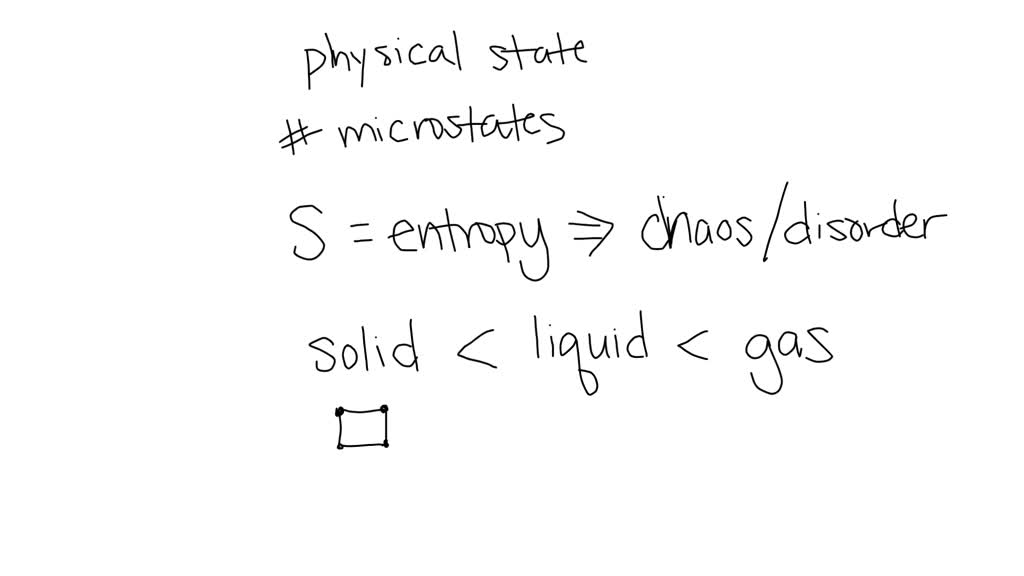
Gases, obviously, are the kings and queens of chaos. Their particles are zipping around everywhere, bouncing off walls, generally having a wild party. That’s high entropy, for sure. Liquids are a bit better, they can slide past each other, but they're still not super organized. Solids, though? Ah, solids are where the magic happens for low entropy. In a solid, the atoms or molecules are usually locked into a pretty rigid structure. They might vibrate a bit, sure, but they're not free to roam all over the place.
The Solid State: A Champion of Order
So, right off the bat, we know we're probably looking at a solid. But not all solids are created equal, are they? Think about a pile of sand versus a perfectly cut gemstone. Both are technically solids, but one is way more ordered than the other. That's where we need to get a little more specific.
We're talking about a mole of particles. So, we need to consider the structure of that solid. What kind of arrangement are we talking about? Are the particles neatly stacked in a predictable pattern, like in a crystal? Or are they more randomly packed, like in an amorphous solid? You guessed it: crystalline solids are the winners when it comes to order.
Think about a beautiful salt crystal, NaCl. The sodium ions and chloride ions are arranged in a very precise, repeating lattice structure. Every sodium is surrounded by chlorides, and every chloride by sodiums. It’s like a perfectly built Lego castle, where every brick has its designated spot. There's very little room for variation, very little chance for things to get mixed up in a way that changes the overall structure.
Now, compare that to something like a piece of glass. Glass is also a solid, but it's amorphous. The atoms are sort of frozen in a disordered arrangement. It’s like someone tried to build that Lego castle but just threw the bricks down randomly before they hardened. Still solid, but much, much messier at the molecular level. So, a mole of salt is going to have less entropy than a mole of glass, assuming they're at the same temperature, of course. Gotta keep our variables consistent, right?

But Wait, There's More! The Element of Surprise (or Lack Thereof)
Okay, so crystalline solids are the frontrunners. But what if we're comparing different elements in their solid crystalline form? Let's say we have a mole of diamond versus a mole of graphite. Both are pure carbon, right? But they have different crystal structures, and therefore, different entropies.
Diamond is known for its incredible hardness and its tetrahedral structure. Each carbon atom is bonded to four other carbon atoms in a very strong, rigid network. It's like the ultimate super-organized structure. Super stable, super ordered. You can just feel the lack of chaos radiating from it, right? (Okay, maybe not, but you get the idea.)
Graphite, on the other hand, is made of sheets of carbon atoms arranged in hexagonal rings. These sheets are held together by weaker forces, and they can slide past each other. Think of it like a stack of pancakes. The pancakes themselves are ordered, but the stack as a whole can be a bit wobbly. So, while graphite is still a solid and more ordered than a gas or liquid, it has more ways for its particles to arrange themselves in a stable, but still slightly flexible, manner compared to diamond.
This means that, at the same temperature, a mole of diamond will have less entropy than a mole of graphite. Diamond is just that much more tightly locked into its perfect, rigid structure. It’s the ultimate picture of atomic tidiness.
What about comparing different elements entirely? Let's think about things like helium versus neon versus argon. These are all noble gases, right? They're notoriously unreactive, which hints at some inherent stability and order. But we’re talking about them in their solid form, assuming we can even get them there. At extremely low temperatures, they do solidify.

When we look at the absolute entropy (that’s the entropy at 0 Kelvin, where everything is theoretically perfectly ordered), we find that simpler atoms tend to have lower entropies. This is because they have fewer electrons, fewer ways to arrange those electrons, and generally less internal complexity. So, if we were to compare solid helium to solid neon, solid neon would have a slightly higher entropy because it has more electrons and a larger electron cloud, giving it more subtle ways to arrange itself.
However, the question is about least entropy. And we've already established that crystalline solids are the way to go. Among the elements that form simple crystalline structures, we need to think about the simplest, most compact arrangements. The elements at the very top of the periodic table, in their solid states, are good candidates.
The Simplest Structures Win!
So, what's the ultimate winner? The substance with the absolute least entropy for a mole of particles? We need something that is a solid, preferably a highly ordered crystalline solid, and ideally made up of the simplest possible atoms.
Think about it. What’s the simplest atom we have? Helium. It’s just two protons and two neutrons in the nucleus, and two electrons whizzing around. It’s incredibly small and has very little going on, electronically speaking. When you get helium down to a temperature cold enough to become a solid (which is super cold, like almost absolute zero!), its atoms arrange themselves in a pretty simple, close-packed structure.
Compare that to, say, a mole of iron. Iron atoms are much bigger, they have more electrons, and they can arrange themselves in different crystal structures depending on the temperature. This gives them more potential for different arrangements, hence more entropy. Even a mole of water, as ice, is more complex. Water molecules have a specific shape and can form hydrogen bonds, which introduce more possibilities for arrangement.

So, the general rule of thumb here is: the simpler the atom or molecule, the more ordered its solid form tends to be, and therefore the lower its entropy. And when we talk about the absolute simplest, it's hard to beat solid helium. It’s the poster child for order at the atomic level. Its particles are tightly packed, they don’t have much internal structure to vary, and they’re in the most rigid state possible.
It's like comparing a perfectly folded origami crane to a crumpled up piece of paper. The origami crane is the epitome of order, and it represents a very specific, low-entropy state. The crumpled paper? That's chaos in its purest form, maximum entropy!
So, if you were forced to pick one substance that, as a mole, would have the absolute least entropy, you'd be looking at something incredibly simple, in its most ordered state. That points us towards solid helium. It’s the rockstar of low entropy, the undisputed champion of atomic tidiness. It’s just… so well-behaved!
Of course, in reality, achieving perfect order, even at 0 Kelvin, is a theoretical concept. Quantum mechanics throws in some quirks. But for our purposes, thinking about the general principles, solid helium is a fantastic example of minimal entropy. It’s a testament to how much nature prefers things to be neat and tidy, even if it takes a whole lot of cold to get there!
Isn’t that neat? We went from talking about a messy desk to the fundamental order of the universe, all over a hypothetical cup of coffee. So next time you see a gas doing its thing, remember its high entropy. And then, perhaps, picture a tiny, frozen speck of helium, the epitome of atomic tranquility. It’s a little bit of science wonder, right there.