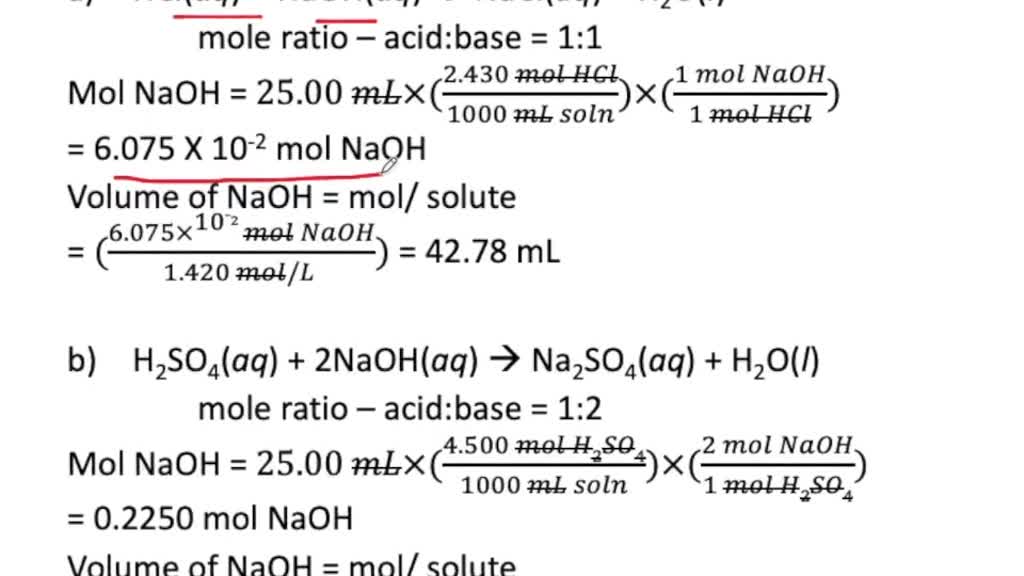
What Volume Of 0.125 M Hno3 In Milliliters

Ever found yourself staring at a science problem and feeling like you're trying to herd cats in a hurricane? Yeah, me too! Especially when it involves things like nitric acid, which sounds super fancy and a little bit like something you'd find in a mad scientist's lab. But guess what? We're going to tackle a tiny little piece of that acid-y puzzle today, and I promise, it's going to be easier than finding matching socks in the laundry abyss!
We're talking about 0.125 M HNO3. Now, if that looks like a secret code, don't sweat it! Think of 'M' as meaning 'Molarity,' which is just a fancy way of saying how much stuff is packed into a certain amount of liquid. It's like saying "a really full cup" versus "a barely wet cup." And HNO3? That's just the scientific nickname for nitric acid. Nothing to fear there, just a common chemical that’s actually pretty useful!
So, the big question that might be buzzing around your brain like a confused bumblebee is: "What volume of this 0.125 M HNO3 are we talking about?" And that, my friends, is where the fun really begins. We're about to dive into the world of figuring out exactly how much of this solution you'd need if you had a specific amount of something else you wanted to mix it with. It's like playing a super important game of "how much goes where?"
Must Read
Imagine you're baking the most epic batch of cookies ever. You need a specific amount of, say, vanilla extract. Too much, and your cookies taste like a perfume factory exploded. Too little, and they're just…sad. This is kind of like that, but with science! We need to know our measurements so everything reacts just right.
Now, the article prompt has a little bit of a twist. It's asking "What Volume Of 0.125 M HNO3 In Milliliters." This sounds like a question where the answer should be a number, right? Like, "Oh, you need 50 milliliters!" But here's the wonderfully quirky thing: the question itself doesn't give us enough information to pick a single, definitive number. It's like asking, "How much flour do I need?" without saying how many cookies you're baking!
So, instead of giving you a single, boring number (which would be impossible anyway!), we're going to celebrate the idea of figuring out that volume. We're going to imagine all the exciting scenarios where you would need to calculate that volume. Think of this article as a treasure map, not to a specific pot of gold, but to the understanding of how to find that gold!
Let's pretend you're a chemist who's just discovered a new superhero power. This power allows you to neutralize a villain's evil bubbling potion! And this potion, oh boy, it's a doozy. It's made with a super-concentrated version of something nasty, let's call it the "Gooey Glob of Grime." You know that to make your superhero fizz-bomb effective, you need exactly 0.02 moles of nitric acid to react with the Gooey Glob.
Aha! Now we're getting somewhere! We know our target: 0.02 moles of HNO3. We also know our friendly neighborhood acid has a concentration of 0.125 M. Remember what 'M' means? It means moles per liter. So, 0.125 M HNO3 is the same as saying there are 0.125 moles of HNO3 in every single liter of this solution.
Our goal is to find the volume in milliliters. Milliliters are the tiny little cousins of liters. There are 1000 milliliters in 1 liter. So, if we figure out our answer in liters first, we can easily zoom over to milliliters!
Now, let's get our trusty calculator (or our super-powered brain) out. We need 0.02 moles. We have 0.125 moles for every liter. To find out how many liters we need, we can do a little division dance. We divide the moles we need by the moles we have per liter.
So, 0.02 moles divided by 0.125 moles/liter.
Ta-da! The math wizards amongst us will instantly see that this equals 0.16 liters. Isn't that just the coolest? You need 0.16 liters of your 0.125 M HNO3 to stop that Gooey Glob!

But wait! The question specifically asked for the volume in milliliters. And as we said, there are 1000 milliliters in a liter. So, to convert our 0.16 liters into milliliters, we just do a quick multiplication. We multiply our liter amount by 1000.
0.16 liters multiplied by 1000 milliliters/liter.
And the answer? A glorious 160 milliliters! You need 160 milliliters of your 0.125 M HNO3 to save the day! See? Not so scary after all. It’s like unlocking a secret level in your favorite game!
Let's try another super fun scenario. Imagine you're a master perfumer, creating the world's most exquisite scent. You're using a special ingredient, a delightful floral essence, and you know that to get the perfect balance, you need a precise amount of nitric acid to help stabilize it. You’ve determined, through extensive (and probably very fragrant) experimentation, that you need exactly 0.05 moles of HNO3.
And, of course, you have your trusty bottle of 0.125 M HNO3 ready and waiting. The concentration is the same, that lovely 0.125 moles per liter. So, we're doing the same calculation dance!
We need 0.05 moles. Our solution has 0.125 moles in every liter. We divide the moles we need by the moles per liter.
0.05 moles / 0.125 moles/liter.
This time, our calculation gives us 0.4 liters. So, you'd need 0.4 liters of that 0.125 M HNO3 to make your perfume smell absolutely divine. Can you imagine a perfume that smells of nitric acid? Probably not the best marketing angle, but you get the idea!
And again, let's convert to milliliters because that's usually how we measure smaller liquid amounts. So, 0.4 liters times 1000 milliliters/liter gives us a whopping 400 milliliters! Four hundred milliliters of our awesome acid!
The beauty of this is that the 0.125 M part is like a constant sidekick. It's always there, ready to tell us how much 'stuff' (moles) is packed into each liter. The only thing that changes, and therefore dictates the volume we need, is the amount of 'stuff' (moles) you require for your particular scientific adventure.
So, when you see a question like "What Volume Of 0.125 M HNO3 In Milliliters," it’s really an invitation to play a game of "if you need X moles, how much of this concentration do you grab?" It’s about understanding the relationship between moles, molarity, and volume. It’s about knowing your ingredients!
Whether you're neutralizing evil potions, crafting magnificent perfumes, or even doing a simple experiment for school, the principle is the same. You figure out how many moles you need, and then you use the molarity (0.125 moles/liter in this case) to find out the volume. And then, a quick hop, skip, and a jump to milliliters!
Don't let the scientific jargon intimidate you. Think of it as a fun code to crack. HNO3 is just a name, 'M' is just a unit, and 'milliliters' are just a way of measuring. You’ve got this!
So, the next time you encounter such a question, remember this. It’s not about a single, fixed answer. It’s about the journey of calculation, the thrill of figuring out the perfect amount, and the satisfaction of knowing you can conquer any measurement challenge that comes your way. You're basically a chemistry ninja!
Keep that enthusiasm buzzing! You're not just reading about science; you're doing science, one calculation at a time. And that, my friends, is incredibly cool!





