What Set Of Quantum Numbers Is Not Allowed

Imagine you're playing a super cool game, but with tiny, invisible things called electrons. These electrons are like little characters in a cosmic play, and they have their own special ways of behaving. To keep track of them, we use something called quantum numbers. Think of them as the electron's unique ID badge, telling us where it is and how it's acting!
Now, not just any combination of these quantum numbers is allowed. It's like having a secret handshake – you have to get the moves just right for it to work! Some sets of quantum numbers are like a cosmic faux pas, a big no-no in the quantum world. And honestly, figuring out these forbidden sets is kind of like solving a fun puzzle. It's where the magic of quantum mechanics really shines, and it’s honestly pretty entertaining.
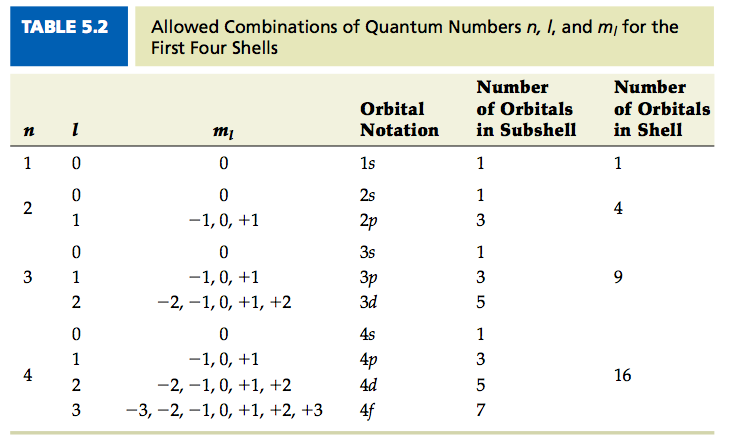
So, what are these mystical quantum numbers? We have four main ones, each telling us something different about our little electron friend. First, we have the principal quantum number, usually shown as a big 'n'. This one tells us the electron's main energy level, sort of like its general neighborhood. Think of it as telling you if the electron lives in a tiny studio apartment (n=1), a cozy one-bedroom (n=2), or a sprawling mansion (n=3). The bigger the 'n', the further away from the atom's nucleus the electron is, and the more energy it has.
Must Read
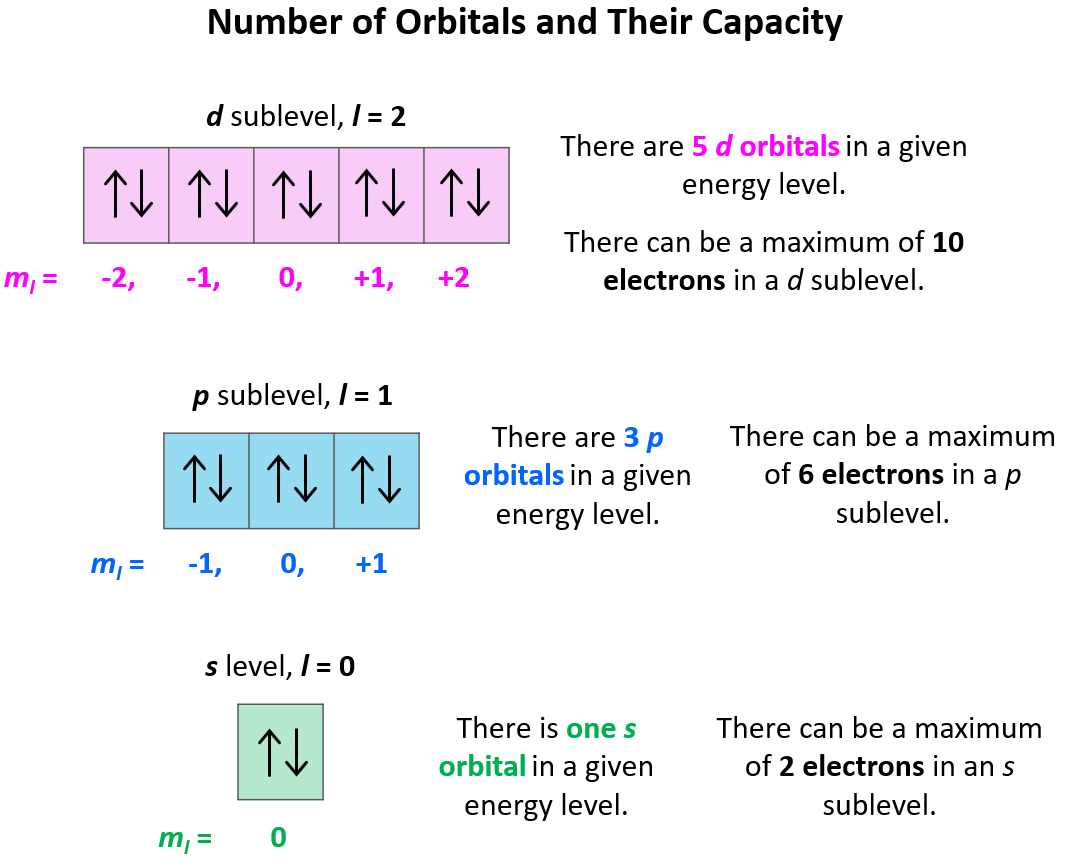
Next up is the angular momentum quantum number, symbolized by a lowercase 'l'. This little guy tells us the shape of the electron's orbital. Orbitals aren't just simple circles; they can be spheres, dumbbells, or even more complex shapes! 'l' determines these shapes. For n=1, 'l' can only be 0, meaning the orbital is a perfect sphere. For n=2, 'l' can be 0 (a sphere) or 1 (which makes a dumbbell shape). It’s like choosing between a round balloon or a fancier, more elaborate balloon for your party!
Then comes the magnetic quantum number, or 'ml'. This number tells us the orientation of that orbital in three-dimensional space. Remember those dumbbell shapes for 'l=1'? Well, 'ml' tells you if that dumbbell is pointing along the x-axis, the y-axis, or the z-axis. It’s like saying your balloon isn't just a dumbbell, it’s a dumbbell pointing this way or pointing that way. It's all about how it sits in space.
And finally, we have the spin quantum number, 'ms'. Electrons are like tiny spinning tops! They can spin in one of two directions, which we call "spin up" (represented by +1/2) and "spin down" (represented by -1/2). This is a fundamental property, and it's super important for how electrons pair up in orbitals.
Now, here’s where the fun really begins. There are strict rules about which combinations of these numbers are allowed. It’s like a cosmic dating service where only certain pairings are allowed to go on a date! The most famous rule, the one that truly sets the stage for forbidden sets, is the Pauli Exclusion Principle. It's a bit like saying, "No two electrons in an atom can have the exact same set of all four quantum numbers." Basically, each electron needs its own unique identity!
So, what kind of quantum number sets are we talking about when we say "not allowed"? Let's dive into some examples, and you'll see how the rules get broken, leading to these impossible scenarios.

Imagine an electron with n=1. We know for n=1, 'l' can only be 0. So far so good. Then, for l=0, 'ml' can only be 0. Still on track! Now, the spin. 'ms' can be +1/2 or -1/2. So, a valid set for the first energy level could be (n=1, l=0, ml=0, ms=+1/2) or (n=1, l=0, ml=0, ms=-1/2). These are perfectly fine and allowed.
But what if we tried to give an electron in the n=1 shell a different shape? What if we tried to say (n=1, l=1, ml=0, ms=+1/2)? That's an immediate red flag! Remember, for n=1, 'l' must be 0. It can't be 1. It’s like trying to put a square peg in a round hole – it just doesn't fit the rules of this quantum game. This is a classic example of a forbidden set because the 'l' value is incompatible with the 'n' value.
Another way a set can be forbidden is if the 'ml' value doesn't make sense for the given 'l' value. For example, if we have an electron with l=1, the possible 'ml' values are -1, 0, and +1. But what if we tried to assign it ml=2? So, a set like (n=2, l=1, ml=2, ms=-1/2)? Nope! That 'ml=2' is way outside the allowed range for 'l=1'. It's like asking for a parking spot that doesn't exist.

And of course, the big kahuna, the Pauli Exclusion Principle. If we have an orbital (defined by n, l, and ml), we can have at most two electrons in it, and they must have opposite spins. So, if we try to create a situation where two electrons have the exact same set of (n, l, ml, ms), that’s a major violation. For example, if we already have an electron with (n=2, l=1, ml=0, ms=+1/2), we cannot add another electron with the exact same set. That's forbidden!
These forbidden sets are not just arbitrary rules; they are the very foundation of how atoms are structured and how they behave. They explain why elements have the properties they do, why chemical bonds form, and ultimately, why the universe looks the way it does. It’s a testament to the elegance and order of the quantum realm, even in its seeming complexity.
Thinking about these forbidden quantum number sets is like peeking behind the curtain of reality. It shows us that even at the smallest scales, there are fundamental laws that govern everything. It’s a bit like discovering a secret language that the universe speaks. And the more you learn about these "not allowed" combinations, the more you realize how much incredible order there is in what might seem like pure chaos. It's a truly fascinating and entertaining aspect of science that invites you to explore the hidden rules of the cosmos. So, next time you hear about quantum numbers, remember the fun of figuring out what can't happen, because that’s often where the most interesting discoveries lie!