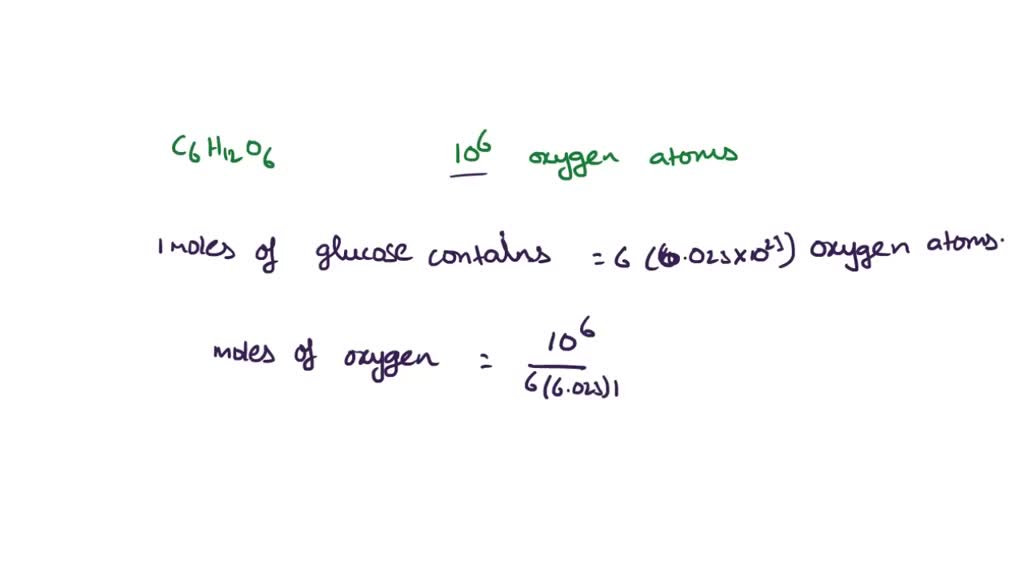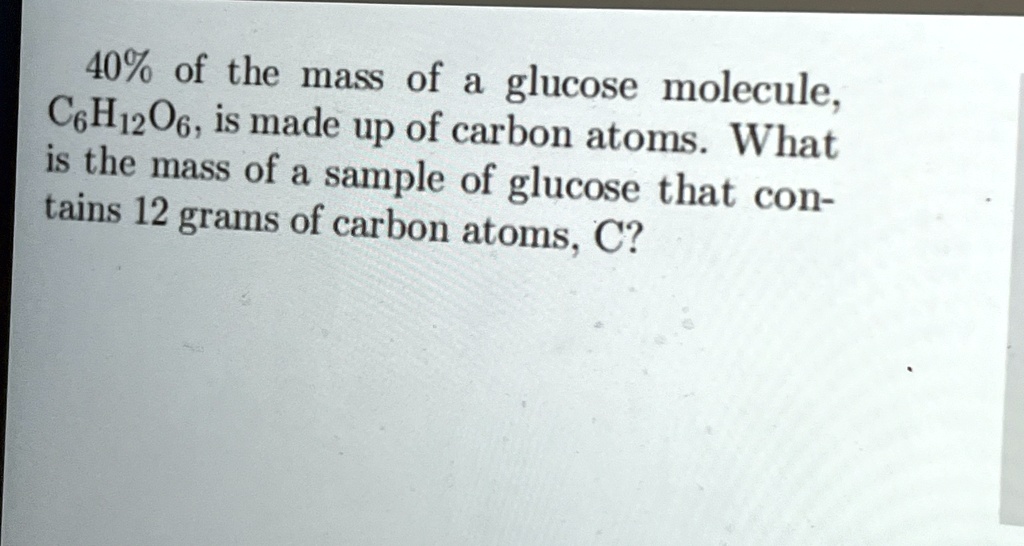What Percent Of Glucose C6h12o6 Is Carbon
Hey there, curious minds and kitchen adventurers! Ever find yourself staring at a bag of sugar, or maybe a perfectly ripe piece of fruit, and wonder about the tiny, invisible building blocks that make them so sweet and satisfying? Today, we're diving into the fascinating world of glucose, also known as dextrose or the simple sugar that fuels our bodies and sweetens our lives. It’s a topic that might sound a little scientific, but trust me, understanding it is like unlocking a secret superpower for appreciating the food we eat and even how our bodies work! It's a fundamental part of so many things we love, from baking a batch of cookies to powering our afternoon jog.
Glucose (with its chemical formula C6H12O6) is incredibly important for our everyday lives. Think of it as the primary energy source for our cells. When we eat carbohydrates, our digestive system breaks them down into glucose, which then travels through our bloodstream to give our brain, muscles, and every other organ the fuel they need to function. Without glucose, we'd feel sluggish, unfocused, and generally pretty terrible. It's also a key ingredient in many foods, contributing not just sweetness but also texture and color during cooking and baking. From the energy that helps you power through a workout to the sweetness that makes your morning coffee just right, glucose is a true unsung hero.
You encounter glucose in countless forms. It's the natural sugar in fruits like apples and bananas, the star player in honey, and the foundational sweetener in table sugar (sucrose, which is made of glucose and fructose). Bakers know it as corn syrup or dextrose, essential for achieving that perfect chewy cookie or glossy frosting. Even our bodies create and store glucose as glycogen to have a readily available energy reserve. It’s everywhere, from the fuel powering your muscles during a brisk walk to the sweetness that makes a celebratory cake so delightful.
Must Read
So, let's get to the core of our curiosity: what percent of glucose is carbon? To figure this out, we need to look at its chemical formula: C6H12O6. This tells us that one molecule of glucose contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms. To find the percentage of carbon by mass, we need the atomic masses of each element. The approximate atomic mass of carbon (C) is 12.01 g/mol, hydrogen (H) is 1.01 g/mol, and oxygen (O) is 16.00 g/mol.
Let's calculate the molar mass of glucose:
- Carbon: 6 atoms * 12.01 g/mol = 72.06 g/mol
- Hydrogen: 12 atoms * 1.01 g/mol = 12.12 g/mol
- Oxygen: 6 atoms * 16.00 g/mol = 96.00 g/mol
- Total Molar Mass of Glucose: 72.06 + 12.12 + 96.00 = 180.18 g/mol
Now, to find the percentage of carbon:
(Mass of Carbon / Total Molar Mass of Glucose) * 100%
(72.06 g/mol / 180.18 g/mol) * 100% ≈ 39.99%
So, approximately 40% of glucose by mass is carbon! Isn't that neat? It means that a significant portion of this vital molecule is made up of the element that forms the backbone of organic chemistry.
To enjoy glucose and its role in our lives even more effectively, try to appreciate the balance it provides. When baking, experiment with different types of sugars to see how they affect texture and taste – a little bit of knowledge about their glucose content can go a long way! For those focused on health, understanding that complex carbohydrates are broken down into glucose can help in making informed choices about dietary energy. And next time you enjoy a sweet treat or a power-filled meal, take a moment to marvel at the incredible chemistry happening behind the scenes, with carbon playing such a starring role!